Saved Bookmarks
| 1. |
One mole of a gas changed from its initial state (15L,2 atm) to final state (4L,10 atm) reversibly. If this change can be represented by a straight line in P - V curve maximum temperature (approximate), the gas attained is x xx10^(2) K. Then find the value of x. |
|
Answer» `P-2=(10-2)/(4-15)(V-15)` `P-2=(8)/(11)(V-15)` `P=2-(8V)/(11)+(15xx8)/(11)` `P=((142)/(11)-(8V)/(11))` 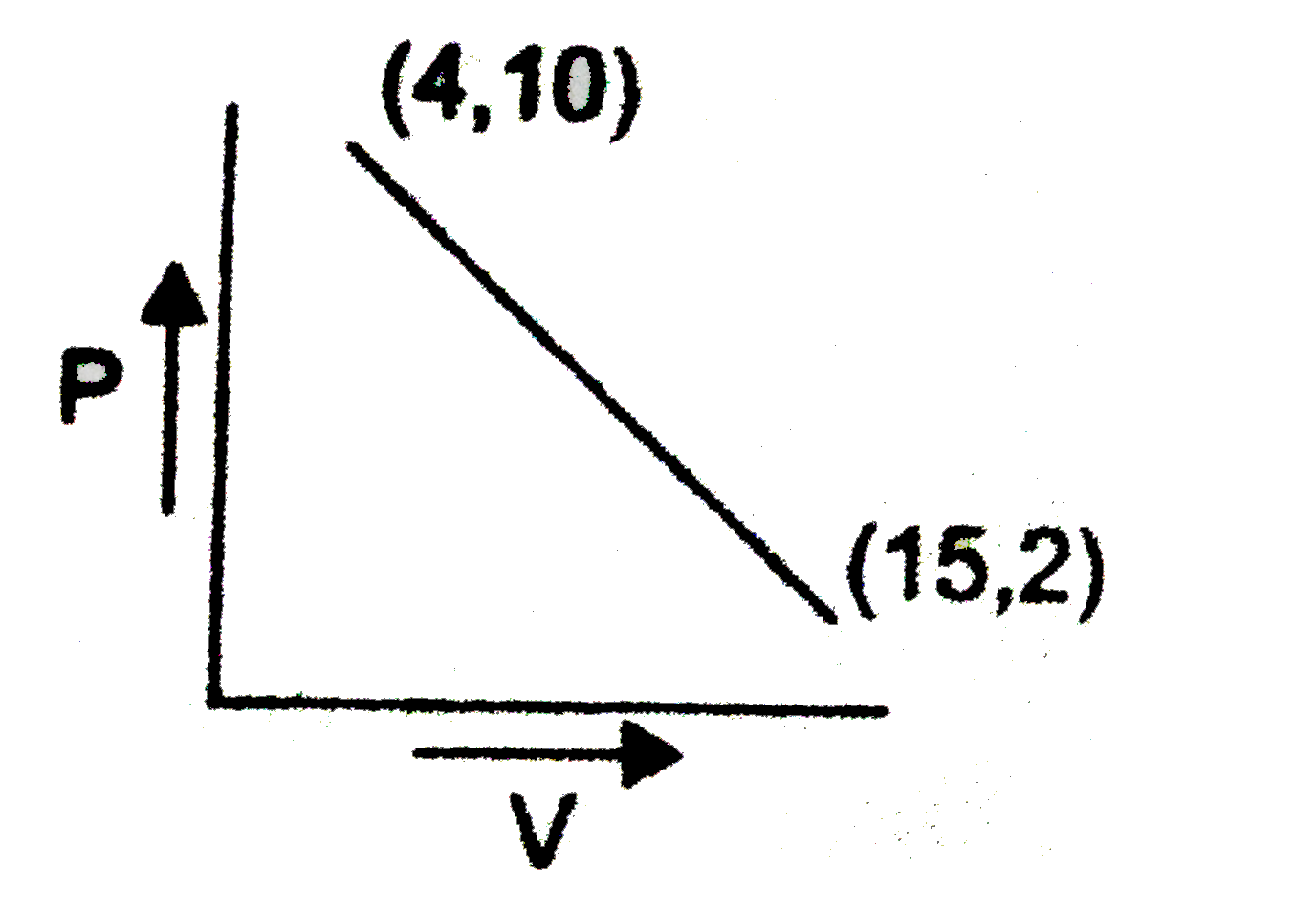 `f(T)=(1)/(nR)((142V)/(11)-(8V^(2))/(11))` `(d(F(T)))/(dV)=(1)/(nR)((142)/(11)-(8V^(2))/(11))=0` `V=(142)/(11xx16)=8.875` `P=(142)/(11)-(8)/(11)xx(8.875)/(16)=(71)/(11)` `T_("Max")=(PV)/(nR)=(71)/(11)xx(142)/(16xx0.0821)=700` `=7XX10^(2)K` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me