Saved Bookmarks
| 1. |
p-Blockelementformacidicbasicand amphotericoxides . Explaineachproperty by givingtwoexamplesand alsowritethe reactions oftheseoxides withwater. |
|
Answer» Solution :Theoxides of p- blockelementshwo acidicbasicand amphotericproperties. Thisis due tothe combinedeffectof numberof factorssome of whichare discussed below, (i)Ionizationenthalpy. Ifthe ionizationenthalpy ofan elementis highit oxidewill beacidicin natureif low , it willbe basicand ifintermediateitsoxidewill beamphotericin nature . Furtherhighertheionizationenthalpystronger will be theacid. (ii)ELECTRONEGATIVITY. Higherthe electronegativity of theelementmoreacidicis itsoxide . Forexampleelectronegativitiesof B . C and Nare -2.0 , 2.5 and3.0respectivelythereforeacidiccharacterof theiroxidesincreasesin the same order`,B_(2)O _(3) ltCO_(2)lt N_(2)O_(3)` (iii) Oxidation state(O.S) . Amongthe oxides of thesameelementhigherthe oxidationstate of theelementstrongeris theacid . Forexample `P_(4)O`(inwhichO.S. of P is + 5)is astrongeracidthant `P_(4) O_(6)` (inwhichO.S.of P is+ 3) . SIMILARLY`SO_(3)` (in whichO.S. of S i s + 6)is a strongeracidthatn `SO_(2)` (inwhichO.S.of S is+ 4) . In thelightof the abovefactorslet us nowdiscus thevariationin acid `//` baseproperties ofoxides ofp-blockelements withina groupand acrossa period. (a)Variationwithin agroup. The oxideof firstelement ofeach groupis acidicnature. Onmovingdownthe groupthe acidiccharacterfirstdecreasesthenbecomesamphotericand finallychangestobasic . For examplethe oxides ofelementsof group13 showthe followingacid - basebehaviour. `underset("Weakly acidic")(B_(2)O_(3))""underset("Amphoteric ")(A1_(2)O_(3))""underset("Amphoteric")(Ga_(2)O_(3))""underset("Basic")(In_(2)O_(3))"" underset("Strongly basic ")(T1_(2)O)` Theionizationenthalpyof B issomewhathigh. Therefore`B_(2)O_(3)` isweakly acidic. It isslightlysolubleinwater andformsorthoboricacid which doesnot ionize itselfin waterbut accepts a pair ofelectronsfrom OH group of water. Thereforeit doesnot act as a protonic acid butacts aweak lewisacid . `underset("Boron trioxide")(B_(2) O_(3) )+3 H_(2) O hArr2 H_(3) BO_(3) underset("Orthoboric acid ")(" or ") 2 B (OH)_(3)` 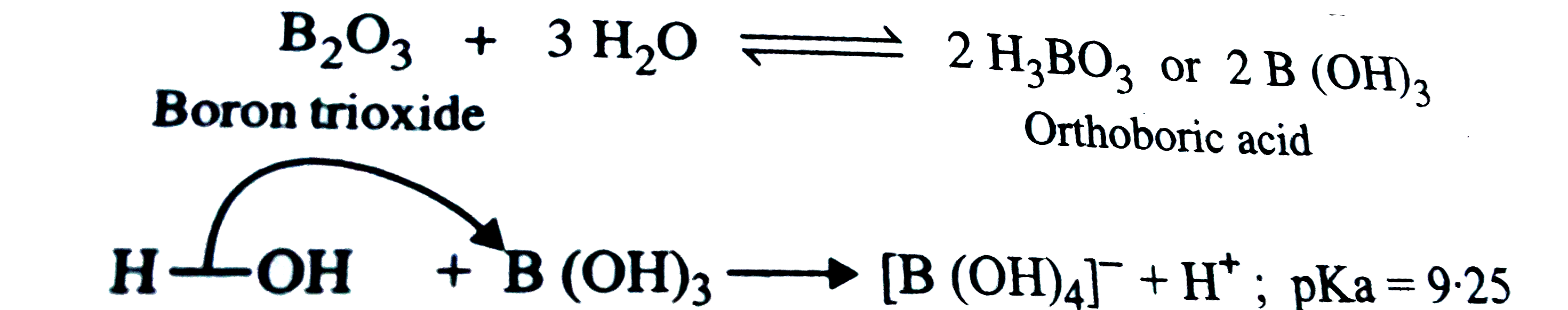 Sinceionizationenthalpyof A1 ismuchlowerthan that of B , therefore`A1_(2)O_(3)`isamphoteric . Itinsolublein waterbut dissolves inalkalies andreacts withacids. `underset("Aluminium oxide ")(A1_(2) O_(3) ) +2 Na OHoverset(Heat) (to) underset( "Sod. meta- aluminate ")(2 NaA1O_(2)) + H_(2)O` `underset("Aluminimum oxide ")(A1_(2)O_(3)) + 6HCIoverset (Heat) (to) underset("Aluminium chloride ")(2 A1C1_(3)) + 3 H_(2)O` Althoughionizationenthalpy of Ti issomewhathgiehr thant thatof A1,its oxidei.e.,`Ti_(2) O` isas basicasNaOHprobablydue toitsloweroxidationstate of + 1aas compapredto + 3or A1. `T1_(2)O_(3)+ 2 HC1 to 2 T1C1 + H_(2)O` Theoxidesof groups14,15and 16elements showsimilarytrendsbut oxidesof group 17 elements are allacids. `{:("Group" 14 "oxides ",,"Group" 15 "oxides",,"Group" 16 "oxides",,"Group" 17 "oxides"),(CO_(2) "Acidic ",,N_(2)O_(5) "Strongly acidic",,-,,-),(SiO_(2) "Less acidic",,P_(4) O_(10)"Moderately acidic",,SO_(2) "Acidic ",,C1_(2)O_(7)"Strongly acidic "),(GeO_(2) "Lesser acidic ",,As_(4)O_(10) "Amphoteric",,SeO_(2)"Weakly acidic ",,BrO_(2)"Acidic "),(SnO_(2) "Amphoteric ",,Sb_(14) O_(10) "Amphoteric ",,TeO_(2)"Amphoteric ",,I_(2)O_(5) "Acidic "),(PbO_(2) "Amphoteric ",,Bi_(2)O_(3) "Basic ",,PoO_(2) "Amphoteric " ,,-):}` ( b)Variation acrossa period. The ionzationenthalpyandelectronegativity ofelements and theirhighest oxidationstatesincreasesfrom leftto rightacrossa period . Due tocombinedeffect of thesefactors , theacidiccharacter of the oxides of elementsincreases.forexampleacrossthe thirdperiodof p-block element, theacidiccharacter of the oxidesincreasesin the order`,A1_(2) O_(3)` (amphoteric ) `lt SiO_(2)` (weakly acidic) `lt P_(4) O_(10)`(moderately acidic ) `lt SO_(3)` (strongly acidic ) `lt C1_(2) O_(7)` (verystronglyacidic) . These oxides dissovle in water toformtheircorrespondingacids : `underset("Phosphoruspentoxide ") (P_(4) O_(10))+6 H_(2) O tounderset( " Orthophosphoricacid ")(4 H_(3)PO_(4))` `underset("Dichlorine heptoxide ")(C1_(2)O_(7)) +H_(2) O tounderset( "Perchloric acid ")(2 HC1O_(4))` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me