Saved Bookmarks
| 1. |
Predict the major product of the following reactions and explain their formation. H_3C-CH=CH_2 underset"HBr"overset ((Ph-CO-O)_2)to H_3C-CH=CH_2 overset"HBr"to |
Answer» SOLUTION :In presence of peroxides , addition of HBr to propene follows anti-Markovnikov's RULE and HENCE give 1-bromopropane as the major product . The reaction occurs through a free radical mechanism VIA a more stable free radical intermediate as shown below : 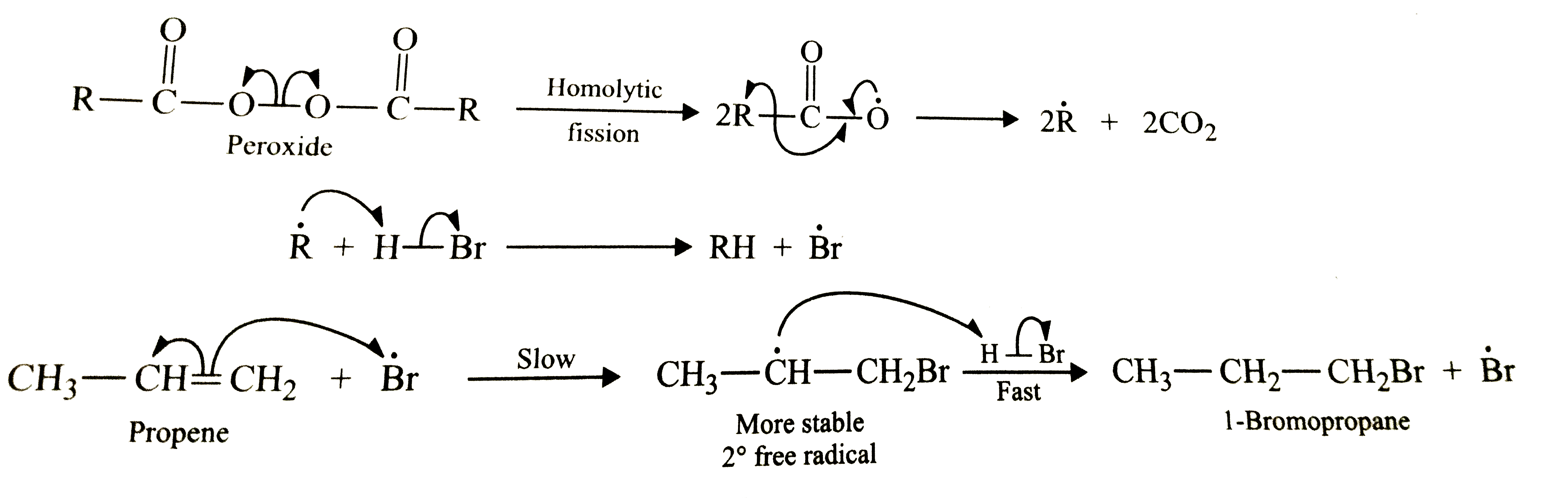 However, in absence of peroxides , addition of HBr to propene follows Markovnikov's rule and hence give 2-bromopropane as the major product. The reaction occurs through IONIC mechanism via a more stable carbocation intermediate as shown below : 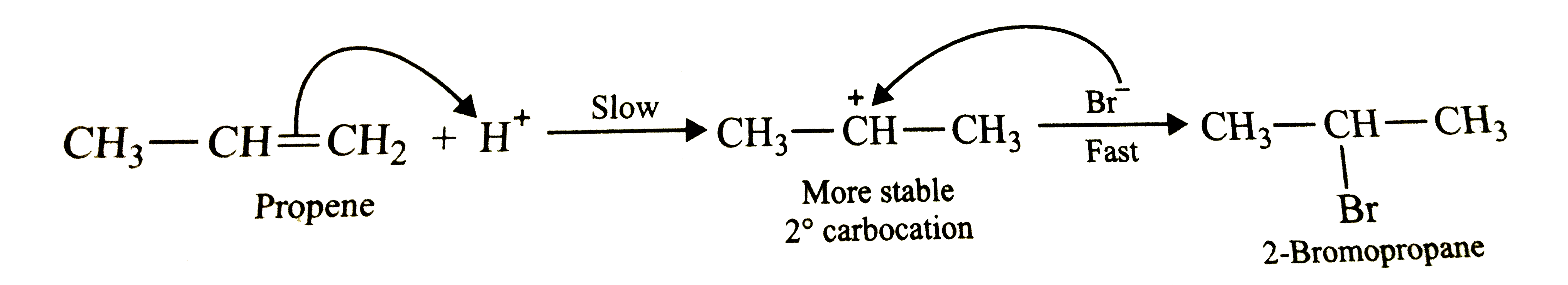
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me