Saved Bookmarks
| 1. |
Represent the potential energy/enthalpy change in the following processes graphically. (a) Throwing a stone from the ground to roof. (b) (1)/(2) H_(2) (g) + (1)/(2) Cl_(2) (g) hArr HCl (g) Delta_(r) H^(Θ) = - 92.32 kJ mol^(-1) In which of the processes potential energy/enthalpy change is contributing factor to the spontaneity ? |
|
Answer» Solution :Representation of potential energylenthalpy change in the following PROCESSES (a) Throwing a stone from the ground to roof. 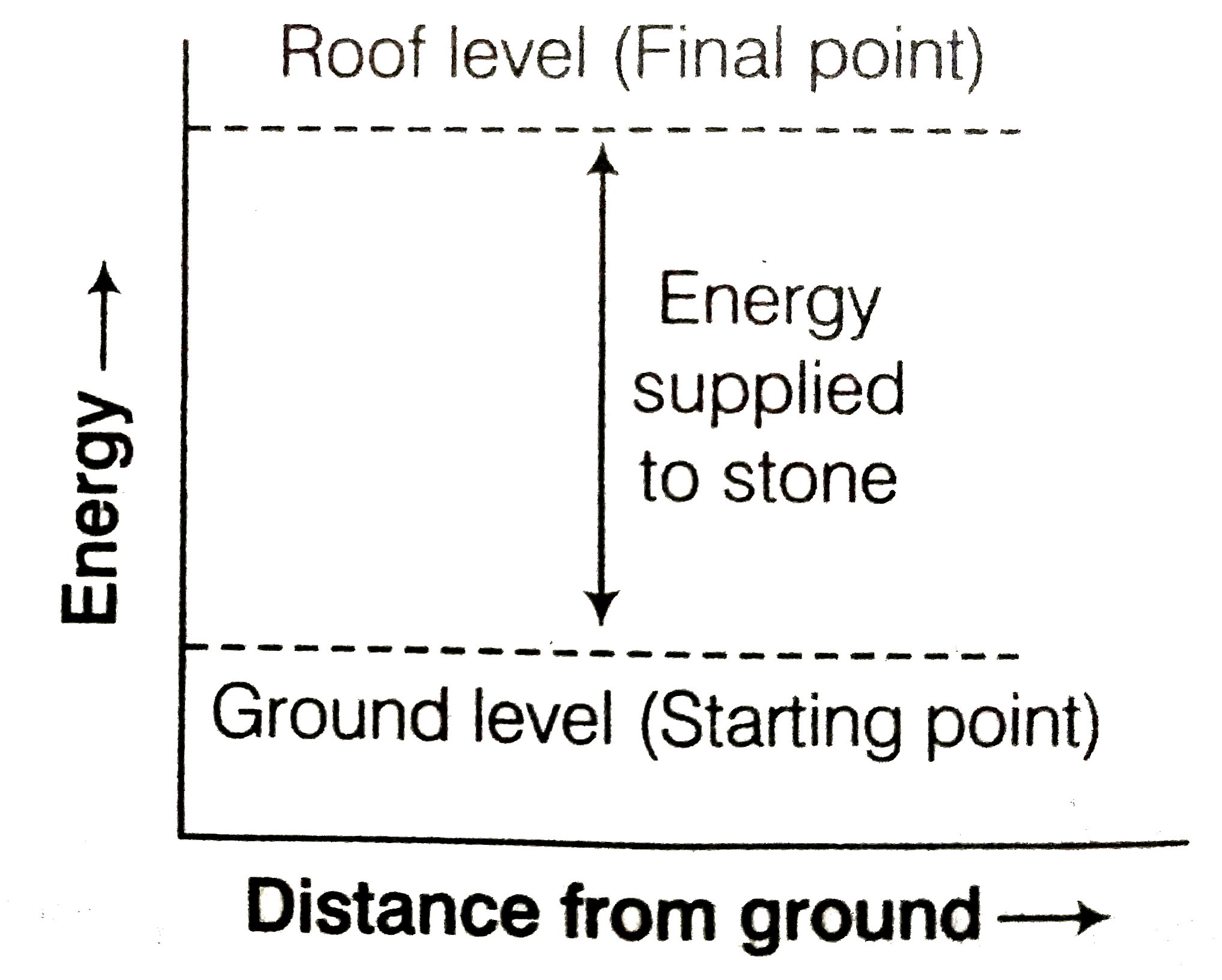 (b) `(1)/(2) H_(2) (g) + (1)/(2) Cl_(2) (g) hArr HCl (g) , Delta_(f) H^(Θ) = - 92.32 kJ MOL^(-1)` 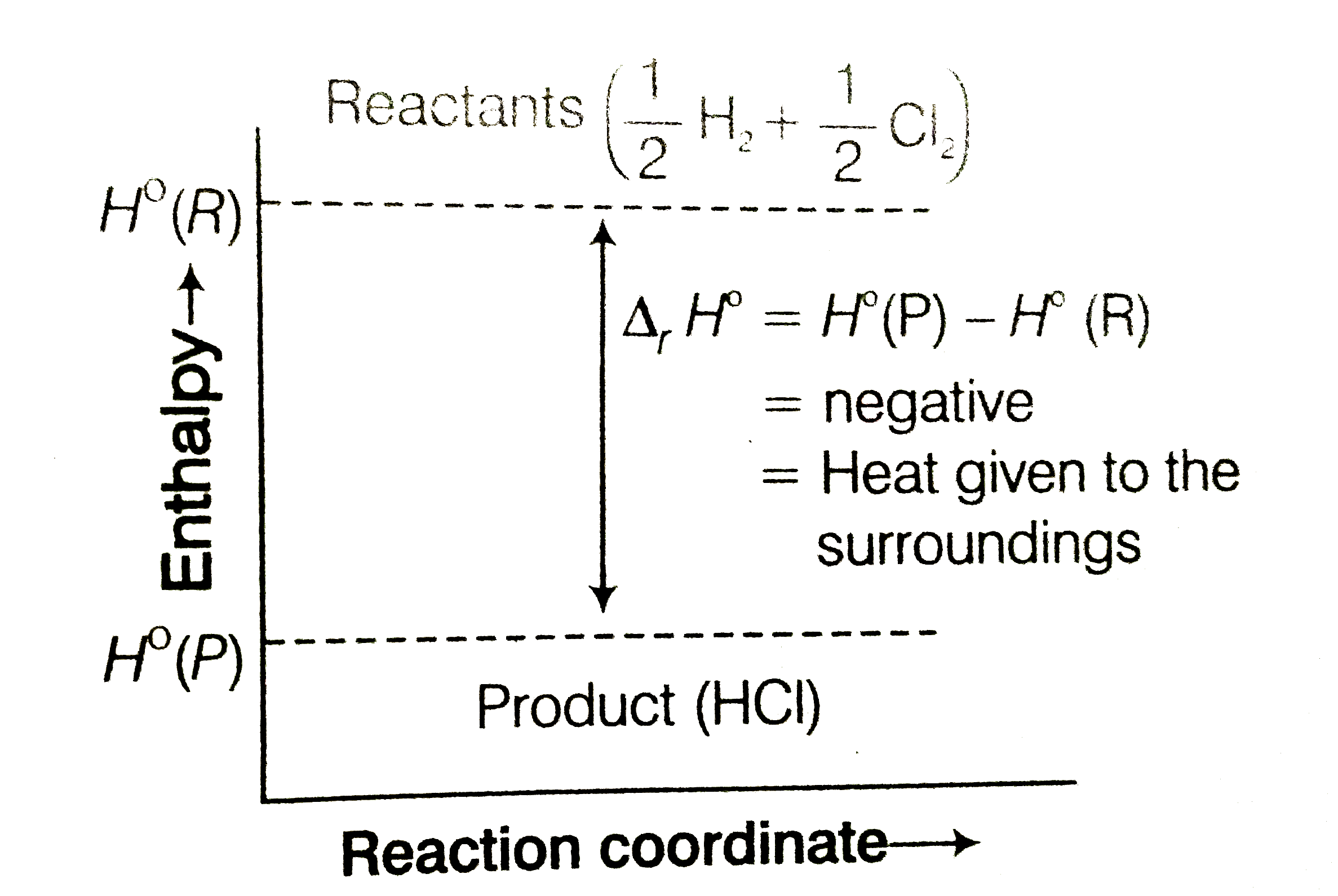 Energy increases in (a) and it decreases in (b) PROCESS. Hence, in process (b), enthalpy change is the contributing FACTOR to the spontaneity |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me