Saved Bookmarks
| 1. |
Show by an arrow the preferred product of reaction with E^(+) of each of the three isomeric, (a) nitrotoluenes, (b) methoxy toluenes and ( c) methoxy acetanilides. Explain your choices in each case. |
Answer» Solution :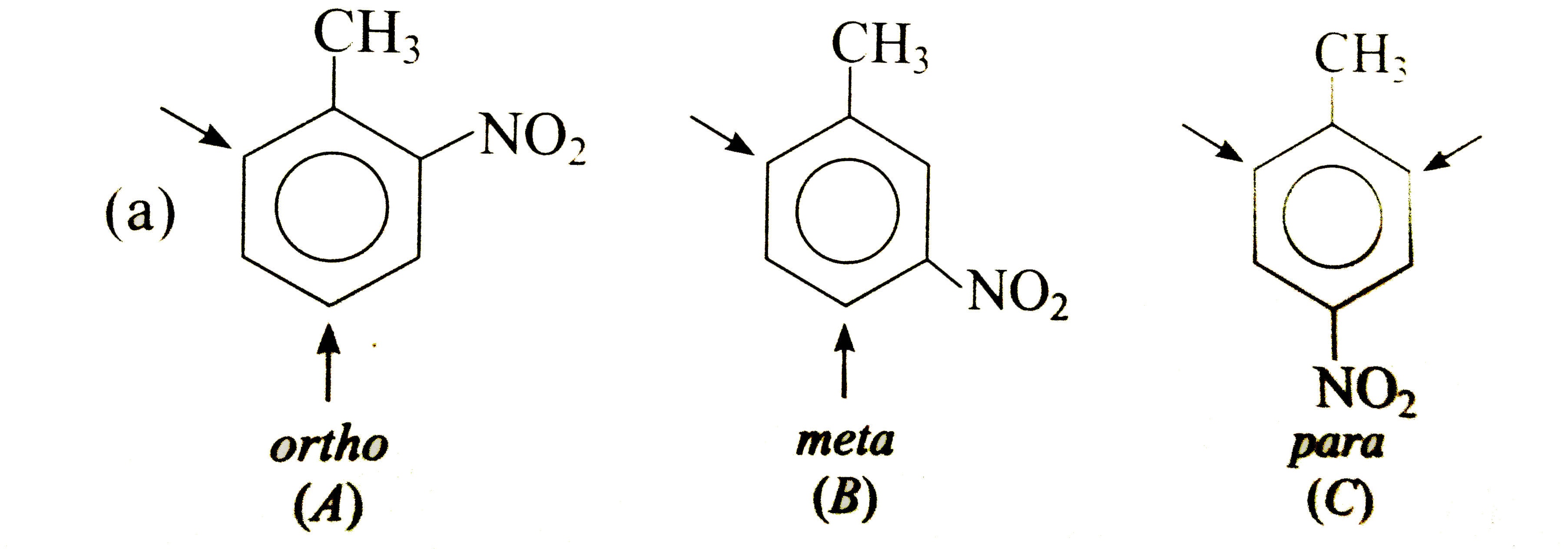 In (A), `CH_(3)` DIRECTS `E^(+)` to ORTHO and para-positions which are also meta to `NO_(2)`, i.e., substituents reinforce each other. In (B), the orientation is in opposition. The o-p-director controls the orientation, i.e., `E^(+)` enters para to `CH_(3)` and ortho to `NO_(2)`. In (C), both groups are reinforcing, i.e., ortho to `CH_(3)` and meta to `NO_(2)`. 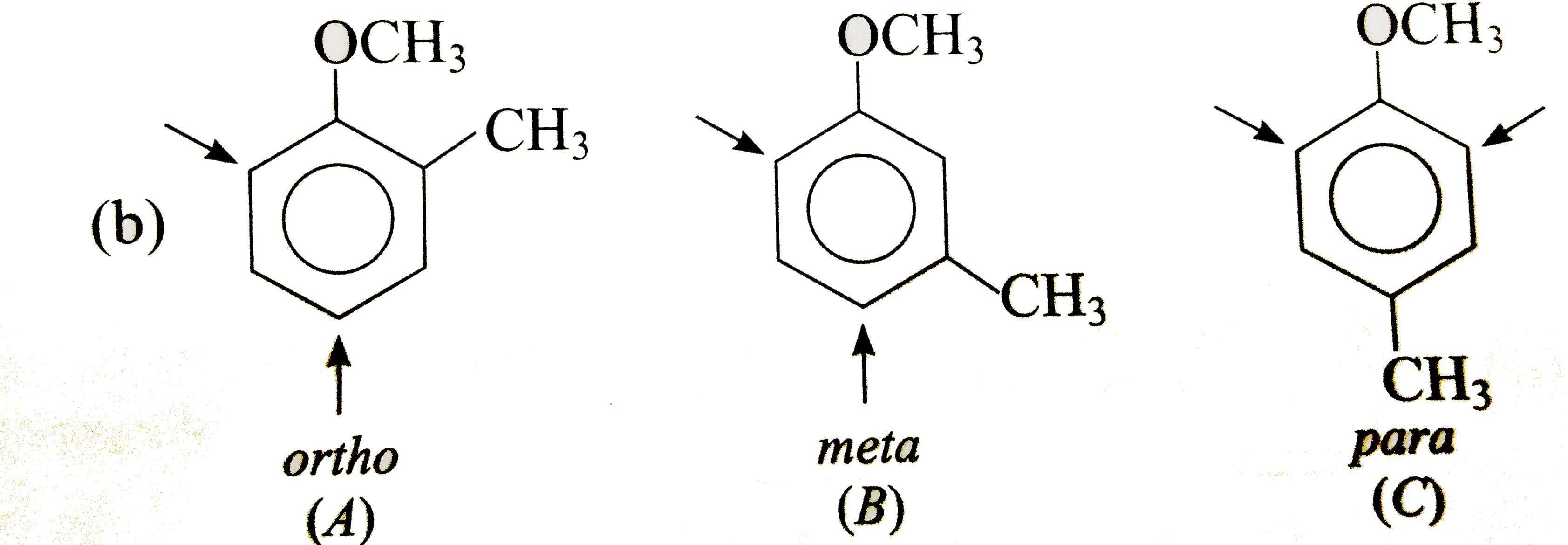 Both groups are o- p-directing. In (B), the substituents reinforce each other, very little substitution occurs between the groups due to steric HINDRANCE. In (A)and(C), the two groups ar in opposition. The more strongly activating `-OCH_(3)` controls the orientation. 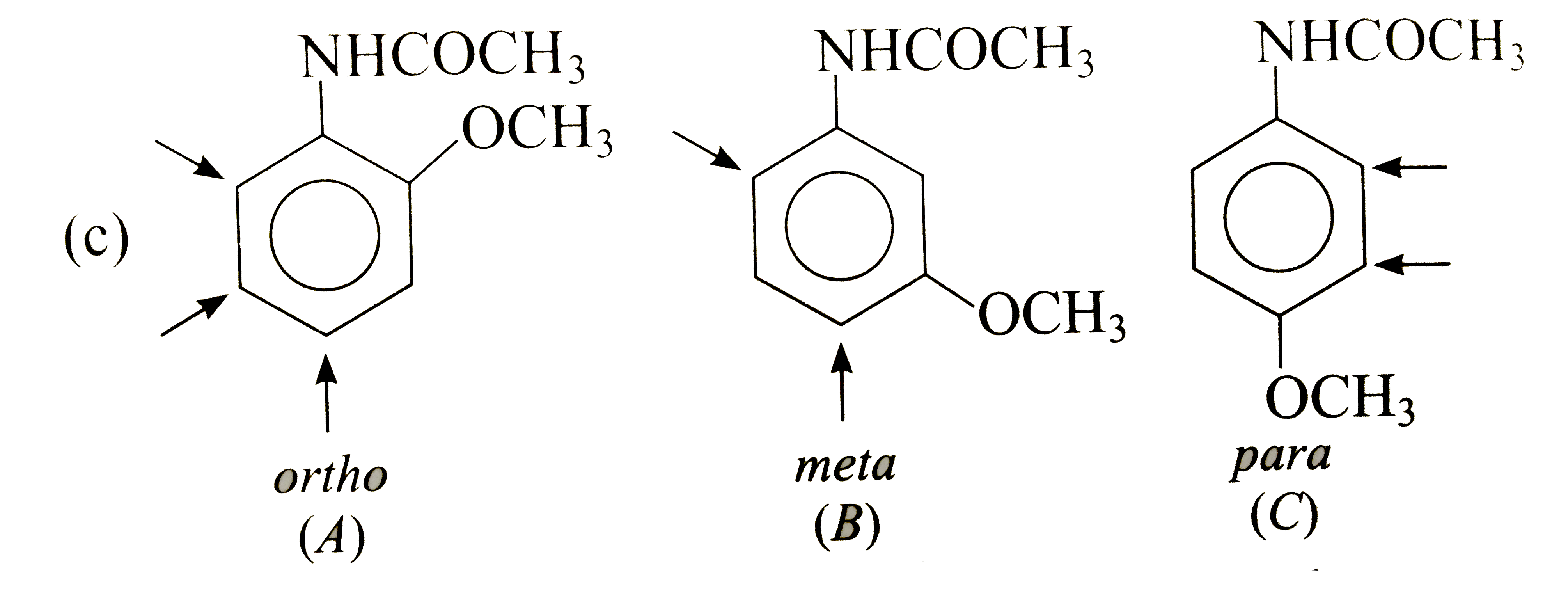 Both the substituents are o- p-directing and equally MODERATE STRONG activators. In (B), both the groups are reingorcing and a mixture of two isomers is obtained. In (A) and (C), two groups are in opposition. In (A), Substantial amounts of all isomers are obtained while in (C), substitution occurs ortho to both the groups. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me