Saved Bookmarks
| 1. |
Show that the circumference of the Bohr orbit for the hydrogen atom is an integral multiple of the de Broglie wave length associated with the electron revolving around the nucleus. |
|
Answer» SOLUTION :In ORDER for the electron WAVE to exist in phase,the circumference of the orbital should be an intergral multiple of the wavelength of the electron wave.otherwise ,the electron wave is out of phase. mvr=ng/2pi `2pir = N lambda` where mvr=angular momentum 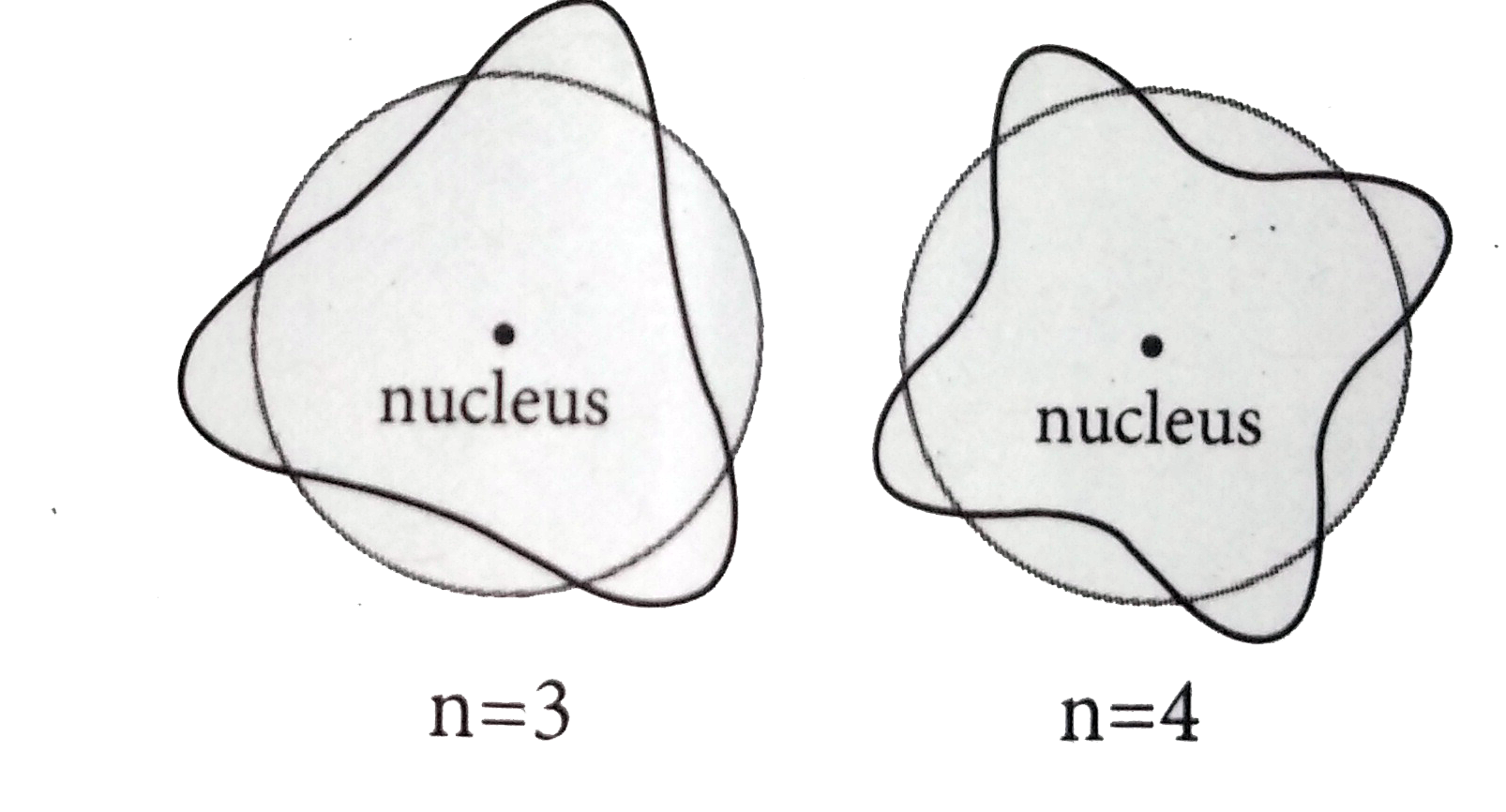 where `2pir`=circumference of the orbit. where `2pir`=circumference of the orbit.n= 3, n=4 |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me