Saved Bookmarks
| 1. |
Somemercury is poured into a capillary tube of uniform bore and one end closed. When the tube is held horizontally, mercury column has a length of 4 cm and air enclosed in the closed end has a length of 10 cm. If the tube is now held vertically, with open end upwards, the length of the air column will be (take atmospheric pressure at the place of experiment to be 760 mm) |
|
Answer» <P>10.5 cm 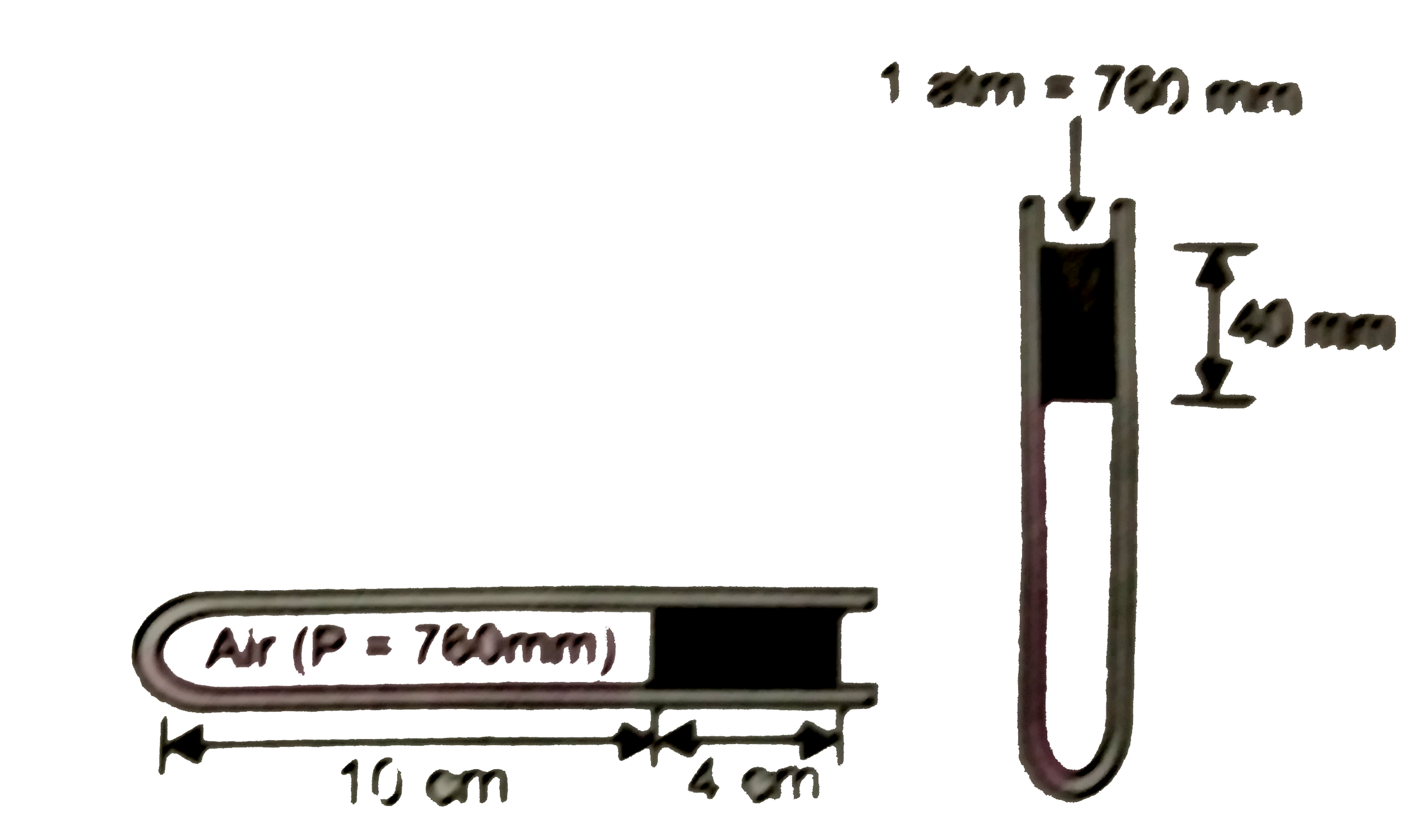 When held horizontally, pressure of enclosed air =760 mm When held vertically, pressure increases by 4 cm, i.e., 40 mm Hence, now pressure=760+40=800 mm Applying `P_(1)V_(1)=P_(2)V_(2)` `P_(1)xx(Axxl_(1))=P_(2)xx(Axxl_(2))` (A=area of cross-section of the capillary TUBE) or `P_(1)xxl_(1)=P_(2)xxl_(2)` `760xx10=800xl_(2)"or"_(2)=9.5" cm"` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me