Saved Bookmarks
| 1. |
"Stability of carbocation depends upon the electron releasing inductive effect of groups adjacent to positively charged carbon atom, involvement of neighbouring groups in hyperconjugation and resonance". Draw the possible resonance structures for CH_(3)-underset(..)overset(..)(O)-overset(+)(C)H_(2) and predict which of the structures is more stable. Give reason for your answer. |
Answer» Solution :The given CARBOCATION has two RESONANCE structures, i.e., I and II. 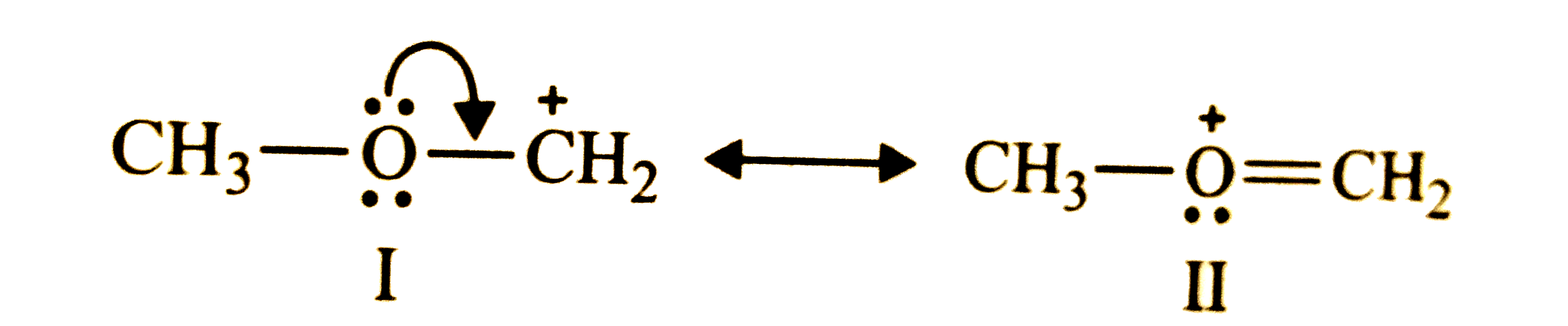 STRUCTURE (II) is more stable since both the carbon ATOMS and the oxygen atom have an octet of electrons. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me