Saved Bookmarks
| 1. |
Suggest reasons why the B_F bond length in BF_(3)(130 pm) and BF_(4)^(ө)(143 pm) differ. |
|
Answer» Solution :In `BF_(3)`,B is `sp^(2)` hybridised and has only six electrons in the valence shell and behaves as an electron-deficient compound. To reduce its electron deficiency, `B` accept a PAIR of electron from filled `2p` orbital present on it. This is known as `ppi-ppi` back bonding which happens due to compatibility in the size and energy of `2p`orbital on `F` and `2p` orbital on `B` atom in `BF_(3)`. Due to back-bonding in `BF_(3)` `B-F` bond ACQUIRES some double bond character resulting in shortening of the bond. However, in `[BF_(4)](ө)`,B is `sp^(3)` hybridised and hence has eight electrons in its valence shell. Since there is an EMPTY `p`-orbital available in `B` in `[BF_(4)](ө)` ION, back-bonding is not possible and `B-F` is purely single bond. Since double bonds are shorter than single bond, the `B-F` bond length in `BF_(3)` is shorter than in `[BF_(4)](ө)`. 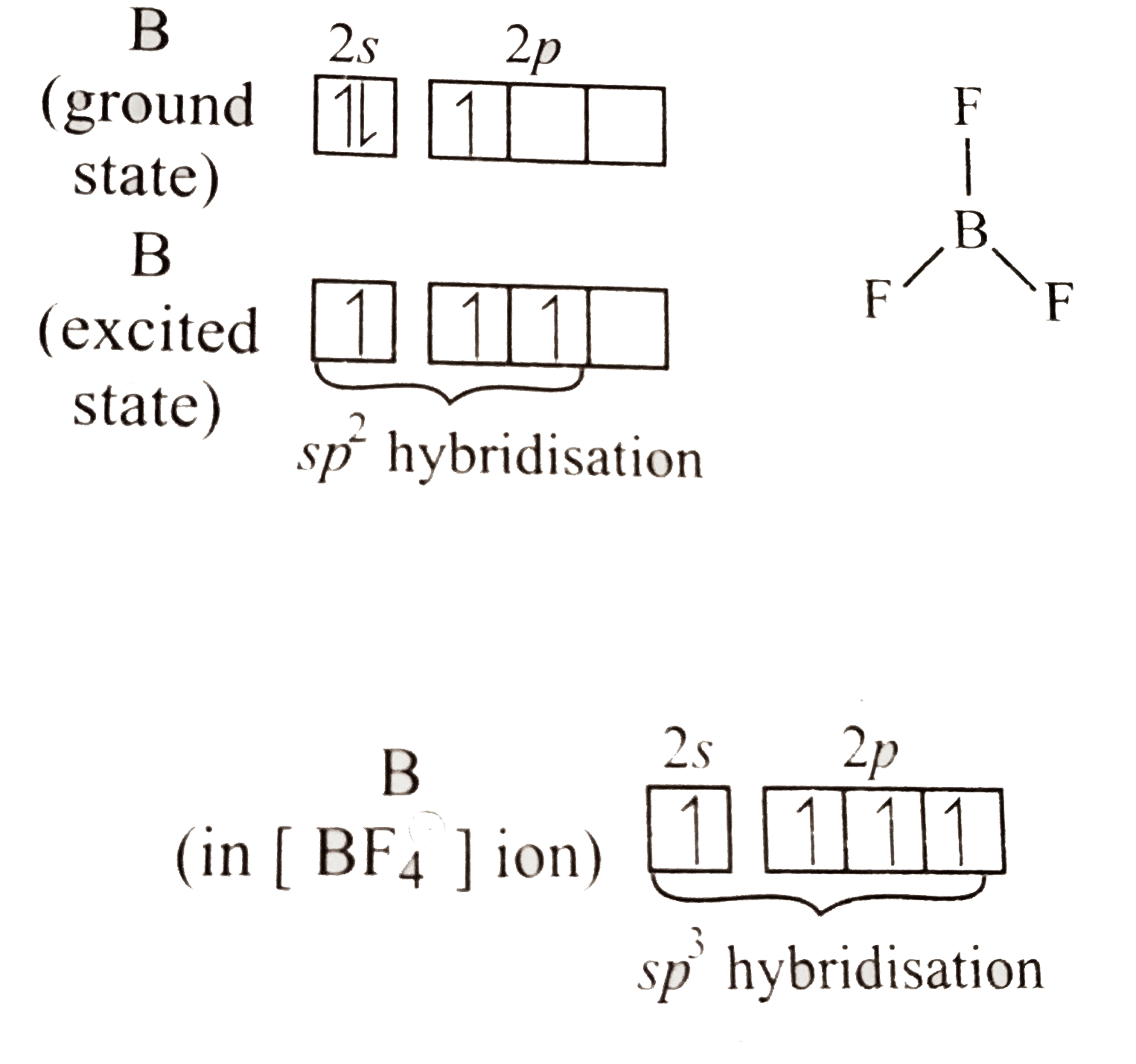 
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me