Saved Bookmarks
| 1. |
The alkyl halide, C_4H_9Br (A) reacts with alcoholic KOH and gives an alkene (B) which reacts with bromine to form a dibromide (C ). (C ) is transformed with sodamide to a gas (D) which forms precipitate when passed through an ammonical silver nitrate solution. Give the structural formula of compounds (A), (B) , (C ) and (D) and explain the reactions involved. |
Answer» Solution :According to the question , 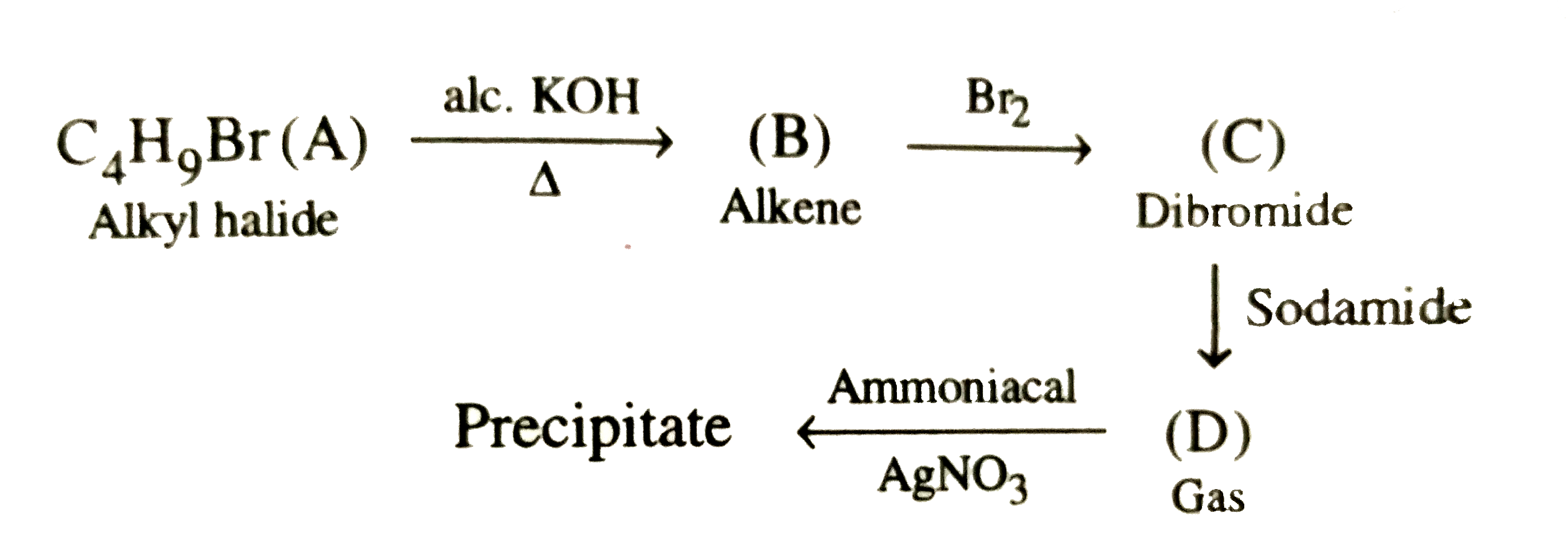 (i)Since the alkyl halide , `C_4H_9Br` (A) on treatment with alcoholic KOH gives an alkene (B) which on subsequent treatment with `Br_2` gives the dibromide (C ) , therefore , the molecular FORMULA of alkene (E) must be `C_4H_8` and that of dibromide (C ) must be `C_4H_8Br_2`. Now since the dibromide , `C_4H_8Br_2` (C ) on treatment with sodamide gives the gas (D), (D) must be an ALKYNE with the molecular formula , `C_4H_6`. Further since the alkyne `C_4H_6` (D) gives precipitate with AMMONIACAL silver nitrate solution, it must be a terminal alkyne. The only terminal alkyne possible for the molecular formula, `C_4H_6` is 1-butyne , i.e., `underset"1-Butyne (D)"(CH_3CH_2-C-=CH)` (ii)Since 1-butyne (D) is obtained by dehydrobromination of dibromide (C ) with sodamide, (C) must be 1,2-dibromobutane i.e., `underset"1,2-Dibromobutane (C)"(CH_3-CH_2-CHBr-CH_2Br)` (iii)Since the dibromide (C ) is obtained by addition of `Br_2` to alkene (B), (B) must 1-butene, i.e., `underset"1-Butene (B)"(CH_3-CH_2-CH=CH_2)` (iv)Since 1-butene is obtained by dehydrobromination of alkyl halide (A) with alcoholic KOH , (A) must be 1-bromobutane or n-butyl bromide , i.e., `underset"1-Bromobutane (A)"(CH_3-CH_2-CH_2-CH_2-Br)` (v) All the reactions cited in the question may now be explained as follows :  Thus , (A) is 1-bromobutane, (B) is 1-butene, (C) is 1,2-dibromobutane and (D) is 1-butyne. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me