Saved Bookmarks
| 1. |
The intermediate carbocation formed in the reactions of HI, HBr and HCl with propene is the same and the bond energy of HCl, HBr and HI is 430.5 "kJ mol"^(-1), 363.7 "kJ mol"^(-1) and 296.8 "kJ mol"^(-1) respectively. What will be the order of reactivity of these halogen acids ? |
Answer» Solution :In the reaction of propene with HCl , HBr or HI, first a `H^+` adds to give the same carbocation intermediate (I) which then undergoes nucleophilic attack by the HALIDE ion `(X^-)` to give the addition product. Here, first STEP is slow and hence is the rate-determining step of the reaction while the second step is first and hence does not affect the rate of the reaction. 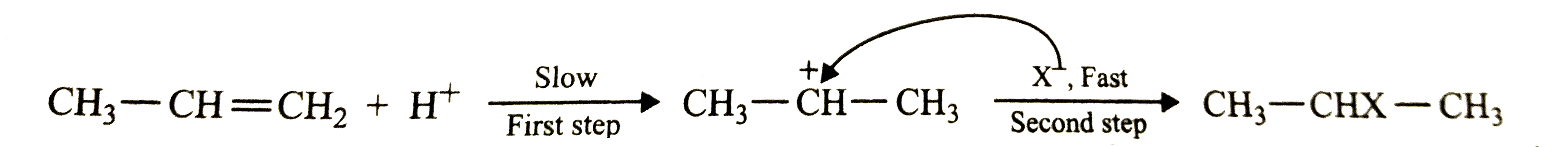 The rate of the first step depends upon how easily is the proton available. The availability of the proton, in turn, depends upon the bond dissociation energy of the H-X MOLECULE. Since the bond dissociation energy decreases in the order : HCl `(430.5 "kJ mol"^(-1)) GT HBr(36. "kJ mol"^(-1)) gt HI (296.8 "kJ mol"^(-1))`, therefore , the REACTIVITY of the halogen acids decreases in the reverse order : HI gt HBr gt HCl |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me