Saved Bookmarks
| 1. |
The molecules having dipole moment are _______ |
|
Answer» 2,2-Dimethylpropane 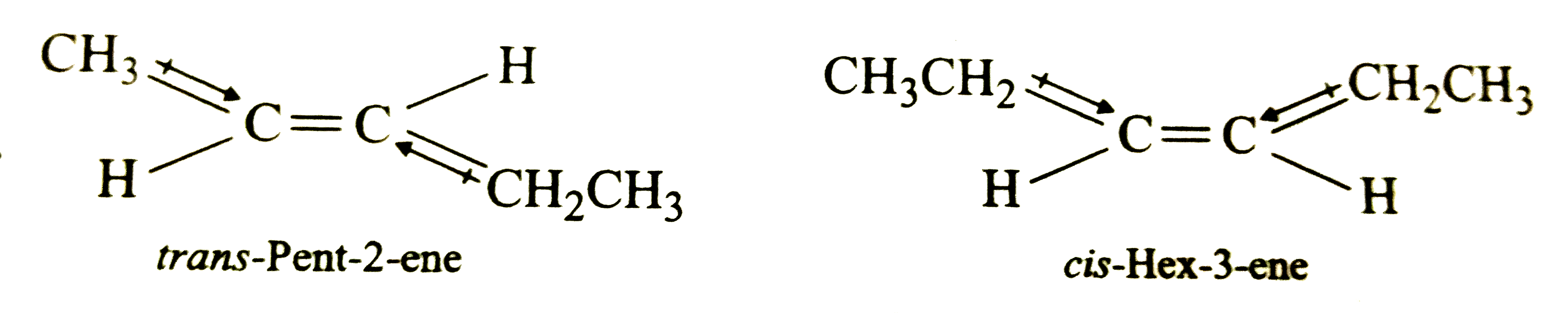 Since the +I-effect of `CH_3CH_2` group is higher than that of `CH_3` group, therefore , the dipole moments of `C-CH_3` and `C-CH_2CH_3` BONDS are unequal . Although these two dipoles oppose each other, YET they do not exactly cancel out each other and HENCE trans-2-pentene has small but finite dipole moment.In cis-hex-3-ene , although the dipole moment of the two `C-CH_2CH_3` bonds are equal , they are inclined to each other at angle of `60^@` and hence have a finite resultant. Further, 2,2-dimethylpropane and 2,2,3,3-tetramethylbutane have zero dipole moments because of the following two reasons : (i)C-C and C-H bonds in alkanes are almost non-polar and have no FIXED orientations in SPACE , therefore, their resultant dipole moment, if any , are almost negligible . (ii)Both these are symmetricalmolecules and hence their dipole moments is zero. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me