Saved Bookmarks
| 1. |
The observed value of dipole moment of H_(2)O molecule is found to be 1 .84 D. Calculate the H-O-H bond angle in H_(2)O molecule , given that the bond moment of O-H bond is 1.5 D. |
|
Answer» Solution :As dipole monent of `H_(2)O`is the resultant of the two vactors(O-H BONDS), therefore ,if `ALPHA ` is the angle between the two vactors , then `mu=sqrt(mu_(1)^(2) + mu_(2)^(2) + 2mu_(1) mu_(2) cos alpha )` `mu_(H_(2)O=sqrt(mu_(OH)^(2) + mu_(OH)^(2) + 2mu_(OH)^(2) cos alpha ))` `1.84 = sqrt((1.5)^(2) + (1.5)^(2)+2 (1.5)^(2) cos alpha)` or`3.3856 = 2.25 + 2.25 + 4.50 cos alpha or cos alpha= -0.2476` or `alpha = 104^(@) 20'[cos (180 - THETA )= - cos theta, i.e.,cos (180 - alpha ) = 0.2476 or 180 - alpha = 75^(@)40']` 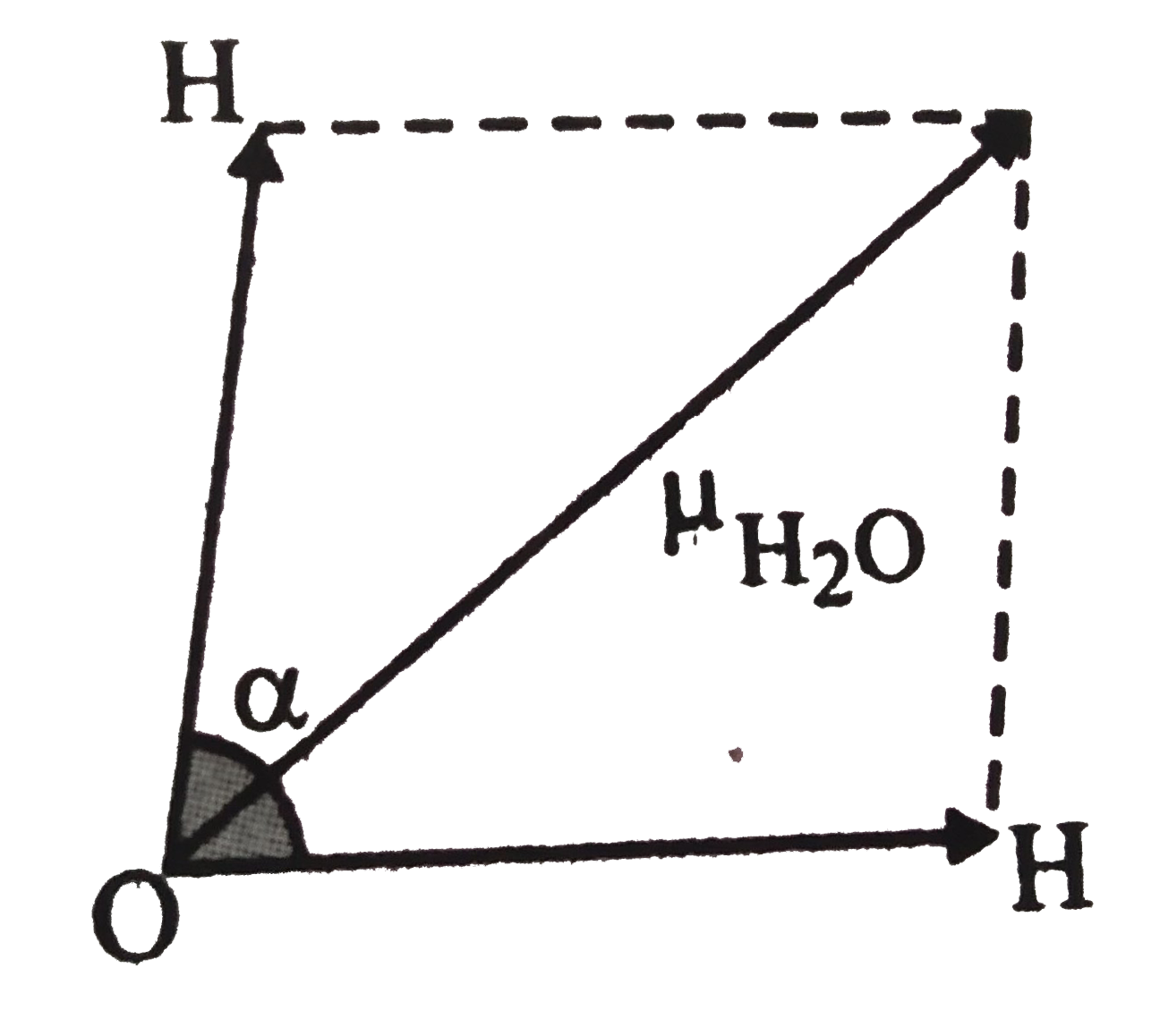
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me