Saved Bookmarks
| 1. |
The ONO bond angle is maximum in |
|
Answer» `NO_(3)^(-)` Structures 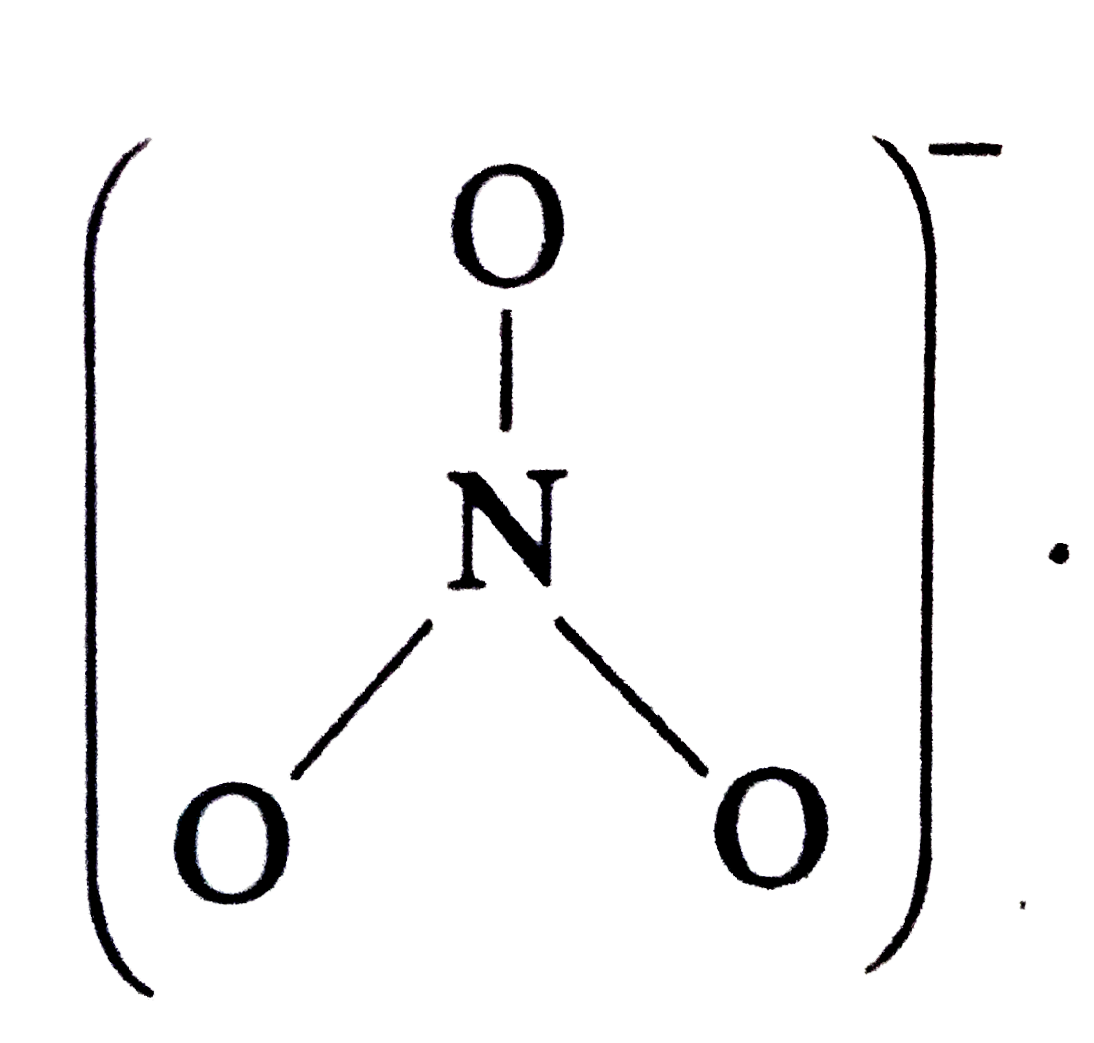 Hence , ONO BOND anlge `= 120^(@)` `NO_(2)^(+)` has no unshared electron . It has only bond PAIRS in two directions . Hence, SHAPE is linear `(underset(. .)overset(. .)O=overset(+)N=underset(. .)overset(. .)O)` with bond angle = `180^(@)` `NO_(2)` has one unshared electron whereas `NO_(2)^(-)` has one unshared electron pair . Hence , in `NO_(2)^(-)` , the repulsion on the bond pairs are more and angle is less 
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me