Saved Bookmarks
| 1. |
The pressure-volume work for an ideal gas canbe calculated by using the expression W=-int_(V_(i))^(V_(f))P_(ex)dV. The work can also be calculated from the pV - plot by using the area under the curve within the specified limits. When an ideal gas is compressed (i) reversibly or (ii)irreversibly from volume V_(i) to V_(f). Choose the correct option. |
|
Answer» `W_("reversible")=W_("IRREVERSIBLE")` 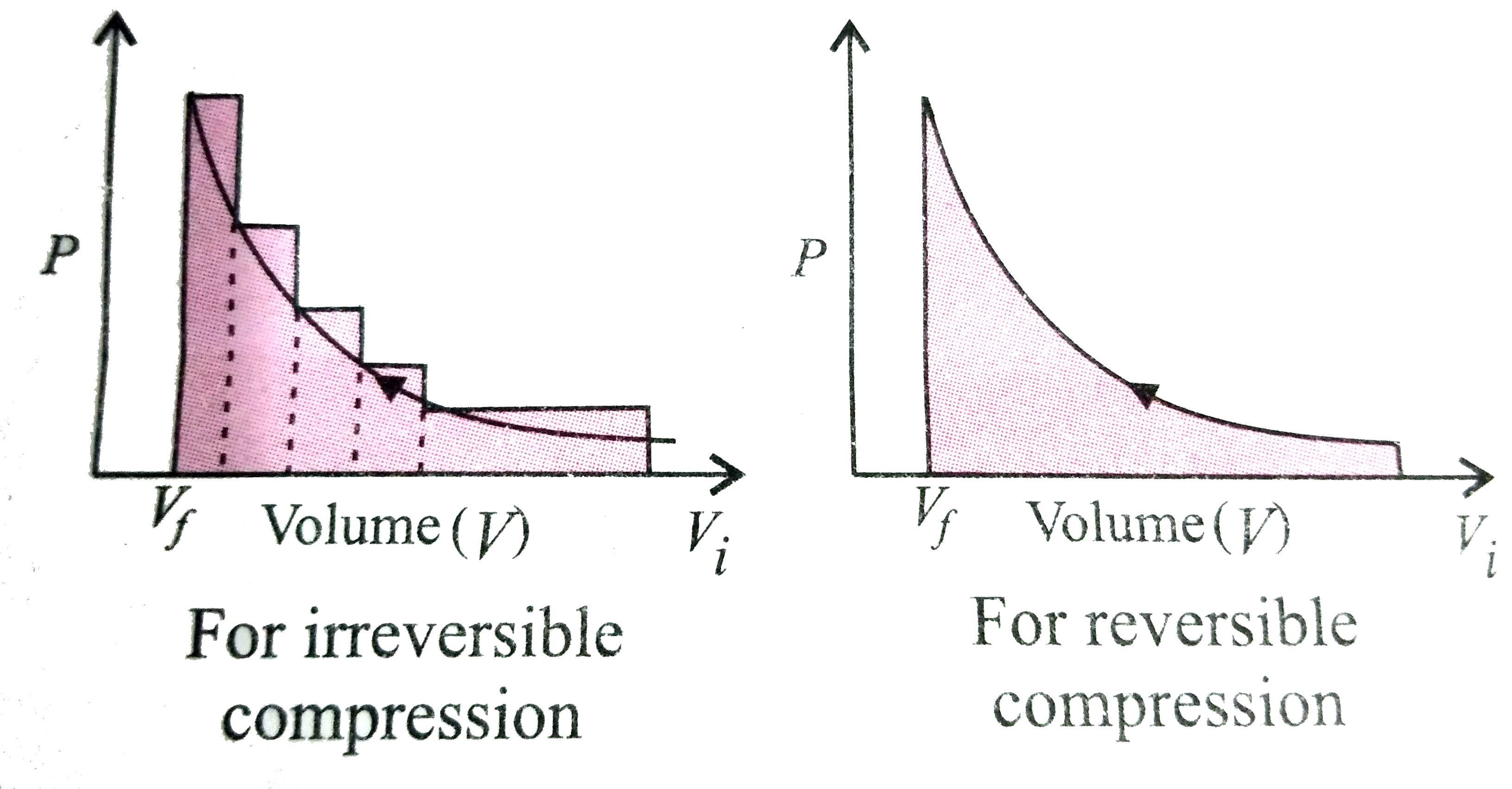 In both the curves, work DONE on the gas is REPRESENTED by the shaded area and the area under the curve is alwaysmore in irreversible compression. Hence, `W_("irreversible") gt W_("reversible")` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me