Saved Bookmarks
| 1. |
The relative reactivity of 1^@,2^@,3^@ hydrogen's towards chlorination is 1:3.8:5. Calculate the percentages of all monochlorinated products obtained from 2-methylbutane. |
|
Answer» Solution :2-Metylbutane has 4 different sets of equivalent HYDROGEN atoms marked as 'a', 'b' , 'c' and 'd' `oversetaCH_3-oversetoverset(.^aCH_3)|(.^BCH)-oversetcCH_2-oversetdCH_3` 2-Methylbutane and HENCE gives four different types of monochlorinated products . These are : 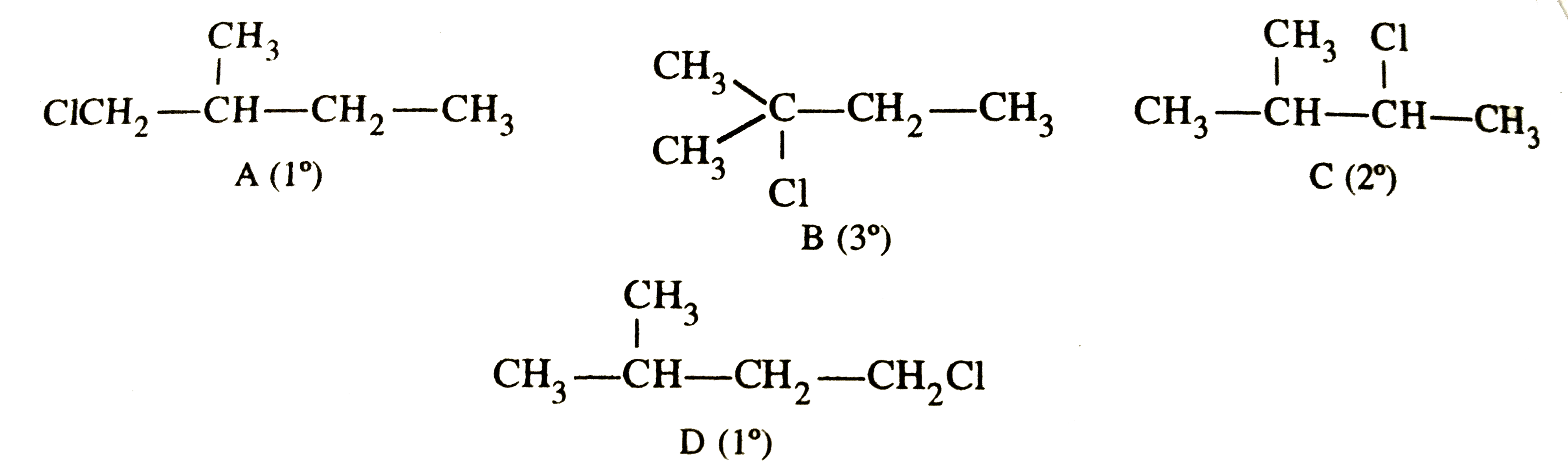 Relative number of equivalent hydrogens , a:b:c :d =6:1:2:3 Relative amounts of A, B, C and D = No. of hydrogen x relative REACTIVITY `{:("Relative amounts :",A(1^@),B(3^@), C (2^@),D (1^@)),(,6xx1=6,1xx5=5,2xx3.8=7.6,3xx1=3):}` Total amount of monohaloginated COMPOUNDS = 6+5+7.6+3=21.6 % A =`(6xx100)/21.6`=27.72 , %B=`5/21.6xx100`=23.15, %C=`7.6/21.6xx100=35.22` , %D=`3/21.6xx100=13.85` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me