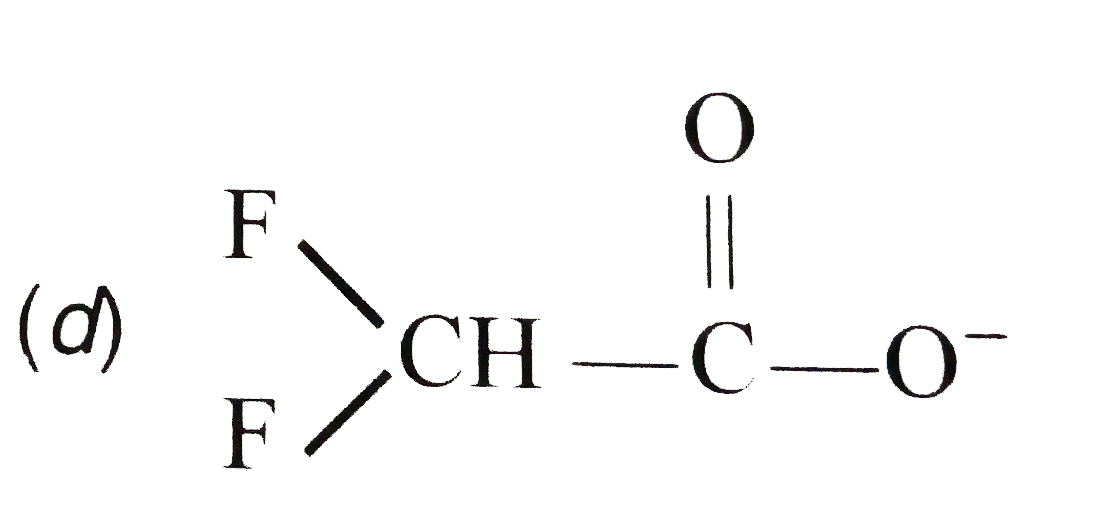Saved Bookmarks
| 1. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. Which of the following carboxylate ions is the most stable ? |
|
Answer» `CH_(3)-overset(O)overset(||)(C)-O^(-)` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me