Saved Bookmarks
| 1. |
The system shown in the figure is in equilibrium, where A and B are isomeric liquids and form an ideal solution at TK. Standard vapour pressures of A and B are P_(A)^(0) and P_(B)^(0), respectively, at TK. We collect the vapour of A and B in two containers of volume V, first container is maintained at 2 T K and second container is maintained at 3T//2. At the temperature greater than T K, both A and B exist in only gaseous form. We assume than collected gases behave ideally at 2 T K and there may take place an isomerisation reaction in which A gets converted into B by first-order kinetics reaction given as: Aoverset(k)rarrB, where k is a rate constant. In container (II) at the given temperature 3T//2, A and B are ideal in nature and non reacting in nature. A small pin hole is made into container. We can determine the initial rate of effusion of both gases in vacuum by the expression r=K.(P)/(sqrt(M_(0))) where P= pressure differences between system and surrounding K= positive constant M_(0)= molecular weight of the gas If partial vapour pressure of A is twice that of partial vapour pressure of B and total vapour pressure 2 atm at T K, where T=50 K and V=8.21 L, then the number of moles of A and B in vapour phase is: |
|
Answer» `(8)/(3),(4)/(3)` 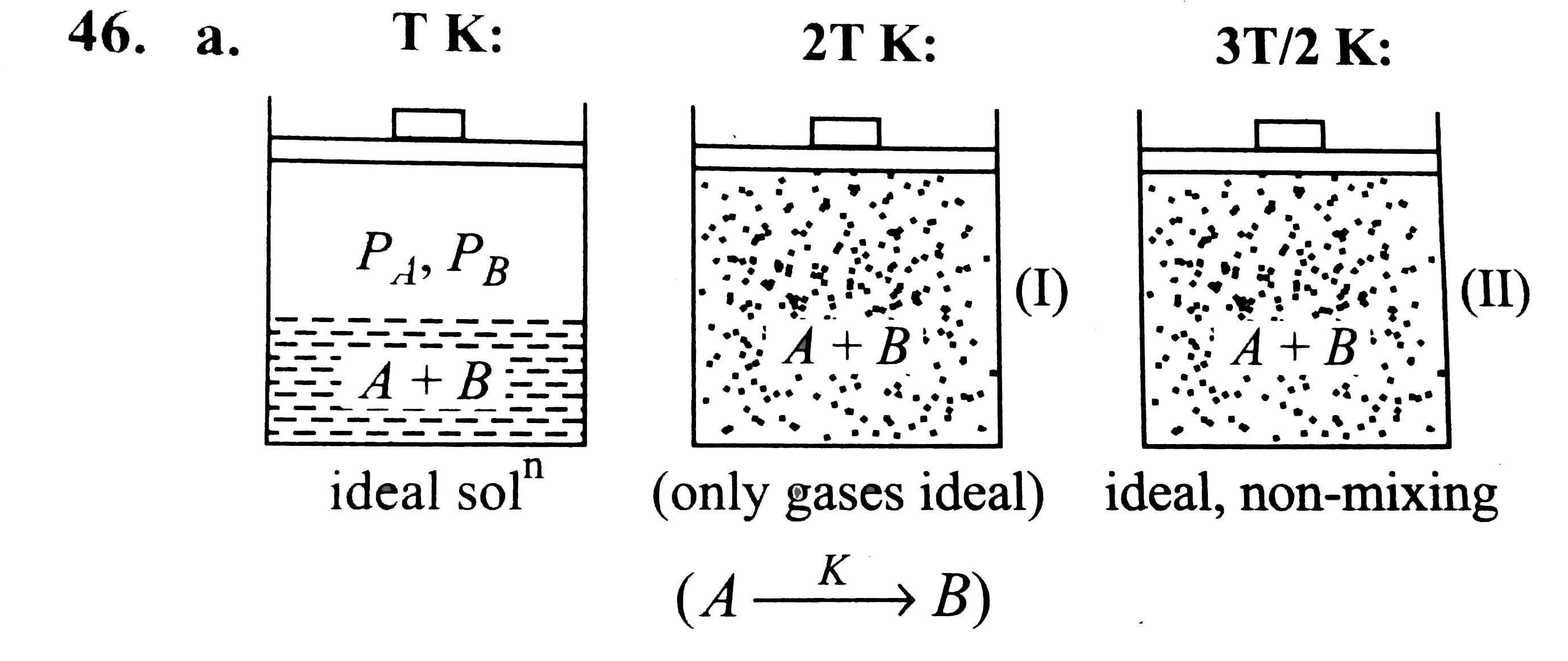 `(n_(A))/(n_(B))=(P_(A))/(P_(B))=2`, Total `V.P.=2 ATM`, `PV=nRTimplies 2xx8.21=(n_(A)+n_(B))xx0.0821xx50` `implies n_(A)+n_(B)=4` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me