Saved Bookmarks
| 1. |
The values of K_(P_(1)) and K_(P_(2)) for the reactions: X hArr Y'Z and A hArr 2B are in the ratio 9:1 if degree of dissociation and initial concentration of X and A be equal then total pressure at equilibrium P_(1), and P_(2) are in the ratio. |
|
Answer» `36:1`  `(K_(P_(1)))/(K_(P_(2)))=(P_(y)P_(Z))/(P_(x))XX(P_(A))/(P_(B)^(2))` `(K_(P_(1)))/(K_(P_(2)))=(P_(y)P_(Z))/(P_(x))XX(P_(A))/(P_(B)^(2))` 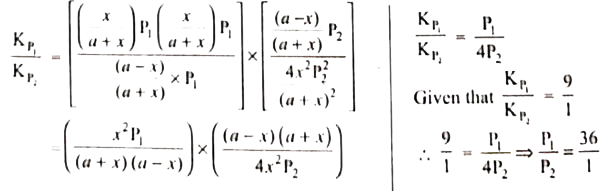
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me