Saved Bookmarks
| 1. |
The % yield of Ammonia as a function of time in the reaction N_(2)(g) + 3H_(2)(g) hArr2NH_(3)(g), Delta H lt O " at " (P, T)is given below: If this reaction is conducted at T_(2) gt T_(1), then plot the % yield of NH_(3)as a function of time on same graph |
Answer» SOLUTION :Initially on increasing temperature the rate of REACTION increases, however since the reaction is exothermic therefore % yield of `NH_(30`GET decreased overall after a certain interval of TIME. 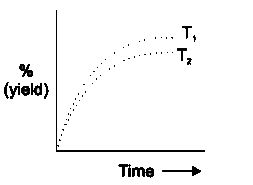
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me