Saved Bookmarks
| 1. |
Two moles of a perfect gas undergo the following processes : (a) a reversible isobaric expansion from ( 1.0atm, 20.0 L)to ( 1.0 atm,40.0 L) (b) a reversible isochoric change of state from ( 1.0 atm,40.0L ) to (0.5 atml, 40.0L) (c ) a reversible isothermal compression from ( 0.5 atm, 40.0 L) to ( 1.0 atm, 20.0L) (i) Sketch with labels each of the processes on the sameP-Vdiagram. (ii) Calculate the total work (w ) and the total heat change ( q) involved in the above processes (iii) What will be the valeu of DeltaU , DeltaH and DeltaS for the overall process? |
Answer» Solution :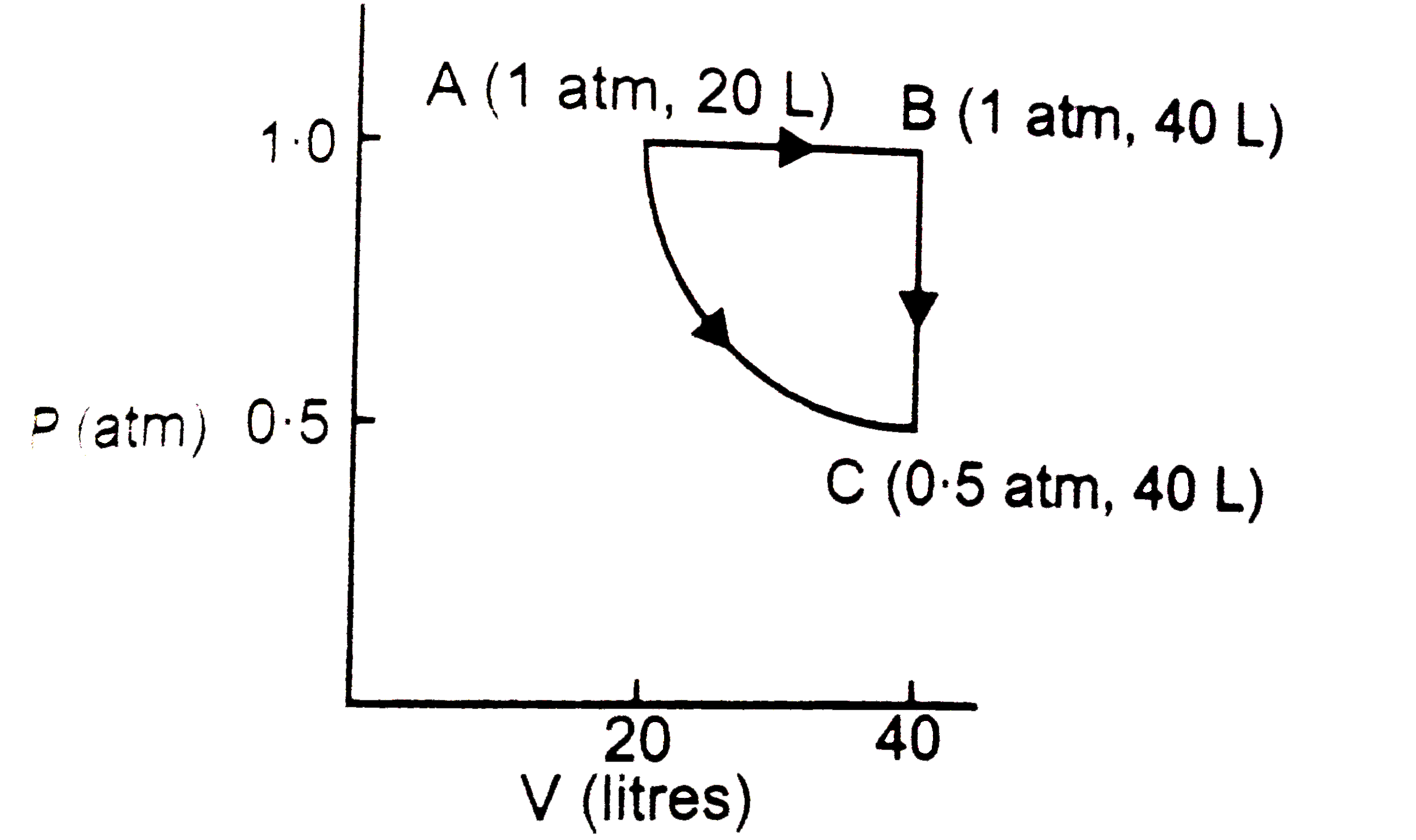 (ii)TOTAL work `(w) = w_(1)+w_(2)+w_(3) =- P DeltaV+ 0 + 2.303 nRT log .(V_(1))/(V_(2))` `= -1 xx 20+2.303 xx 2 xx 0.082 xx 121.95 log 2 (T =(PV)/(nR) = ( 1 xx 20)/( 2 xx 0.082) = 121.95 K) ` `= -20+ 13.86 = - 6.13L atm=-6.13 xx 101.3 J = - 621.0 J ` (In a cyclic process,HEAT abosorbedis completely converted into work) (iii) As U, H and S arestate FUNCTIONS and the system has returned to its original state,`DeltaU =0, DeltaH=0,` and`Delta S=0`. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me