Saved Bookmarks
| 1. |
Using the VSEPR theory , identify the type of hydridisation and draw the structure of OF_(2) . What are oxidation states of O and F ? |
Answer» Solution :Electron dot structures of `OF_(2)` is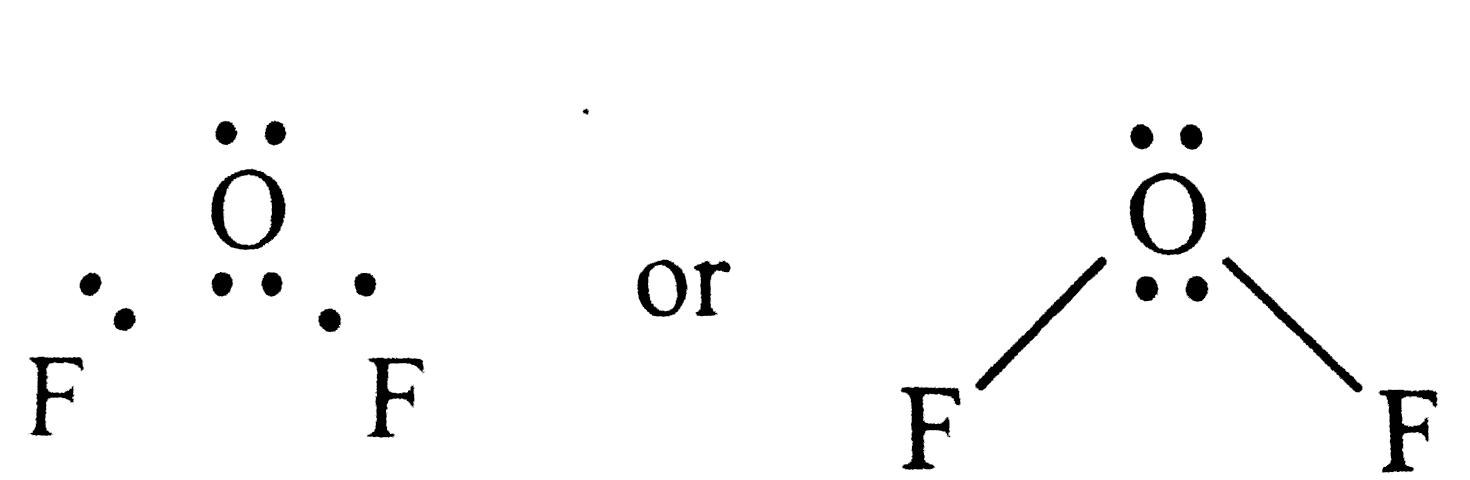 Thus , the central ATOM (O-atom) has 4 pairs of electrons (2bond paris ). HENCE oxygen in `OF_(2) is SP^(2)`hybridised and the molecular is V-shaped . Oxidation state of F = - 1 Oxidation state of O = + 2 . |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me