Saved Bookmarks
| 1. |
What does [H_(9)0_(4)] stands for ? Draw its structure . (b) Can sodium bicarbonate make water hard ? (c) Hard water is softened before make water hard ? (d) What is sequestration ? How is hard water made soft by sequestration ? |
|
Answer» Solution :(a) `H_(9)O_(4)^(+)` stands for hydrated proton . In `[H_(9)O_(4)]^(+) `, `H^(+)` is tetrahedrally surrounded by four water molecules as shown. (b) `NaHCO_(3)` cannot MAKE water hard because soaps themselves are sodium salts of fatty acids which are soluble in water. (c) Hard water on boiling gives precipitates of `MgCO_(3), CaCO_(3) and CaSO_(4)` which form scales in boilers . The formation of these boiler scales causes rapid deterioration of the boiler due to over heating . Further , these boiler scales are non-conducting . Therefore, more fuel is consumed . Therefore, in order to avoid formation of these scales, hard water is softened before using in boilers. (d) Sequestration means to render ineffective . The hardness of water is due to the presence of `Ca^(2+)` and `Mg^(2+)` ions. These ions are rendered ineffective by TREATING them with sodium hexametaphosphate , commercially called calgon. When calgo is added to hard water, the `Ca^(2+) and Mg^(2+)` ions PRESENT in it combine with sodium hexametaphosphate to form soluble complexes of calcium and magnesium salts . These complex calcium and magnesium ions do not form any precipitate with soap and hence READILY produce lather with soap solution. In this way, hard water is softened by sequestration. 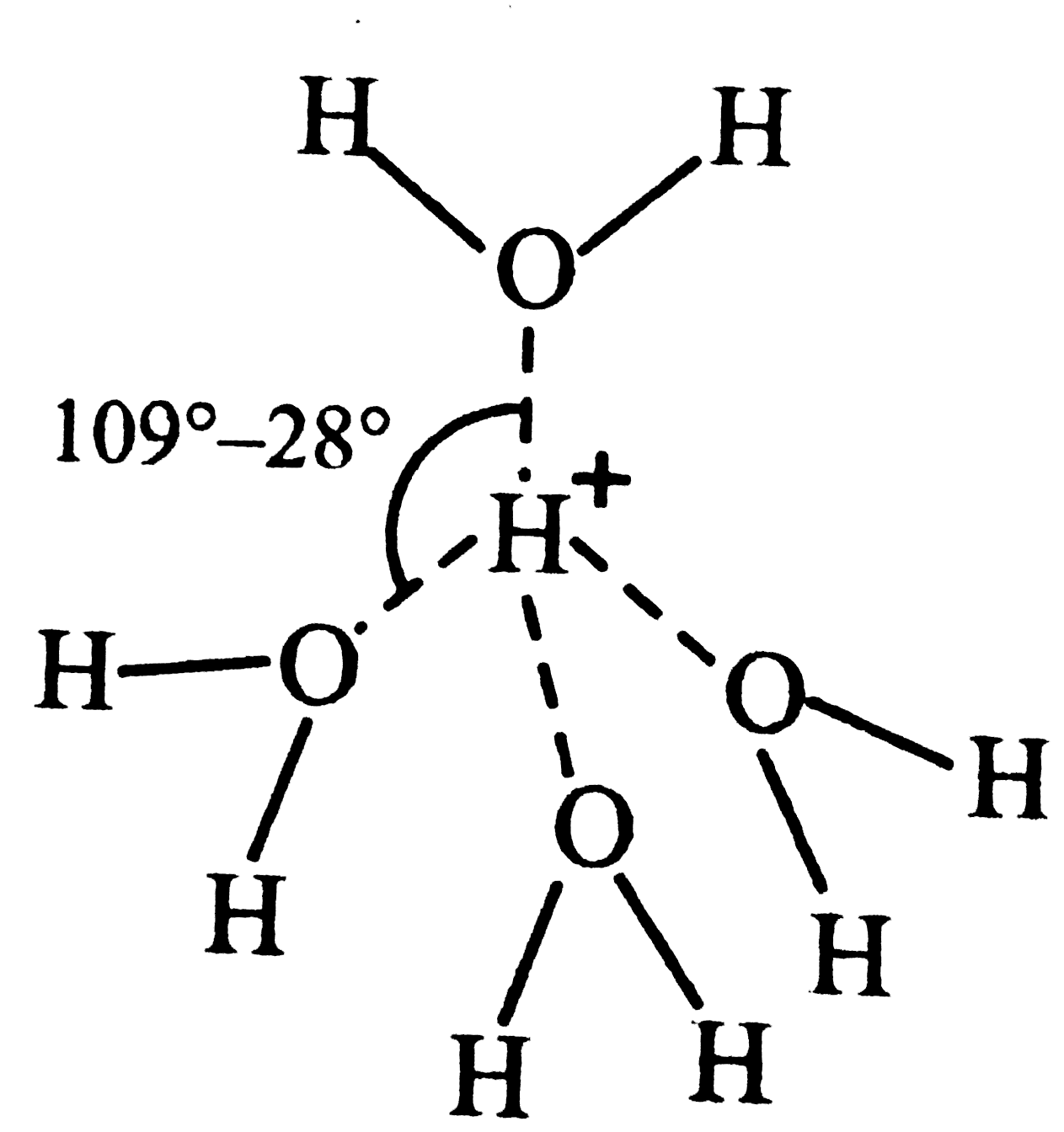
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me