Saved Bookmarks
| 1. |
What happens when a. Borax is heated strongly. b. Boric acid is added to water. c. Aluminium is treated with dil NaOH. d. BF_(3) is reacted with ammonia. |
|
Answer» Solution :a.When borax is strongly heated, a transparent glassy bead which CONSISTS of sodium metaborate and boric anhydride is formed. `underset("Borax")(Na_(2)B_(4)O_(7).10H_(2)O)OVERSET(Delta)toNa_(2)B_(4)O_(7)+10H_(2)O` `Na_(2)B_(4)O_(7)underset("RED Heat")overset(Delta)tounderset(ubrace({:(" Sodium,Boric"),("metaborate,anhydride"):})_("Transport glassy bead"))(2NaBO_(2)" + "B_(2)O_(3))` b. Boric acid behaves as a Lewis acid and hence accepts `overset(ө)OH` ion form `H_(2)O` and realeses `H^o+` in the solution. c. `2Al_((s))+underset(("dil"))(NaOH_((aq)))+6H_(2)Oto2Na^(o+)[Al(OH)_(4)]_((aq))^()+3H_(2(g)) or underset("Sodium meta-aluminate(III)")(Na_(2)AlO_(2).2H_(2)O)` d.`F_(3)B+:NH_(3)to[F_(3)Blarr:NH_(3)]` `BF_(3)` is a Lewis acid and accepts a pair of electron from `N` of `NH_(3)` molecule to form an adduct, `[F_(3)Blarr:NH_(3)]` 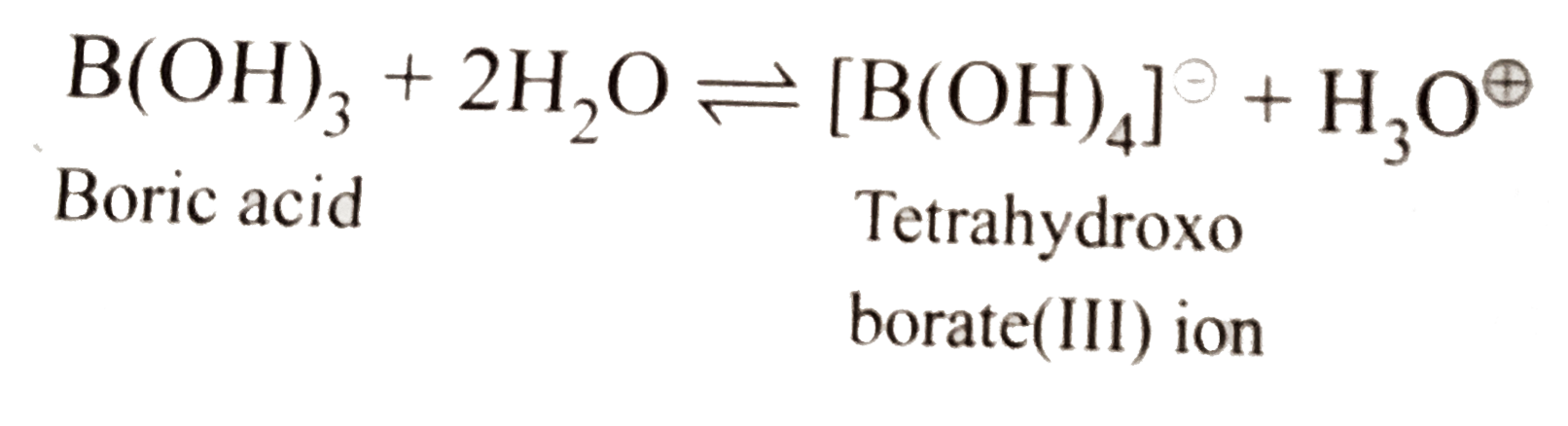
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me