Saved Bookmarks
| 1. |
What happens when: a. Whater is added to CaC_(2) and the resulting gas is passed through dil H_(2)SO_(4) containing HgSO_(4). B. Hydrated MgCl_(2) is heated in presence of NH_(4)Cl. C. FeCl_(3) solution is treated with Mg. D. NH_(4)Cl is heated with Mg.e. CO_(2) is passed through limewater. f. SO_(2) is passed through limewater. |
Answer» Solution :`CaC_(2)+H_(2)OrarrCa(OH)_(2)+underset("Acetylene gas")(C_(2)H_(2)uarr)` 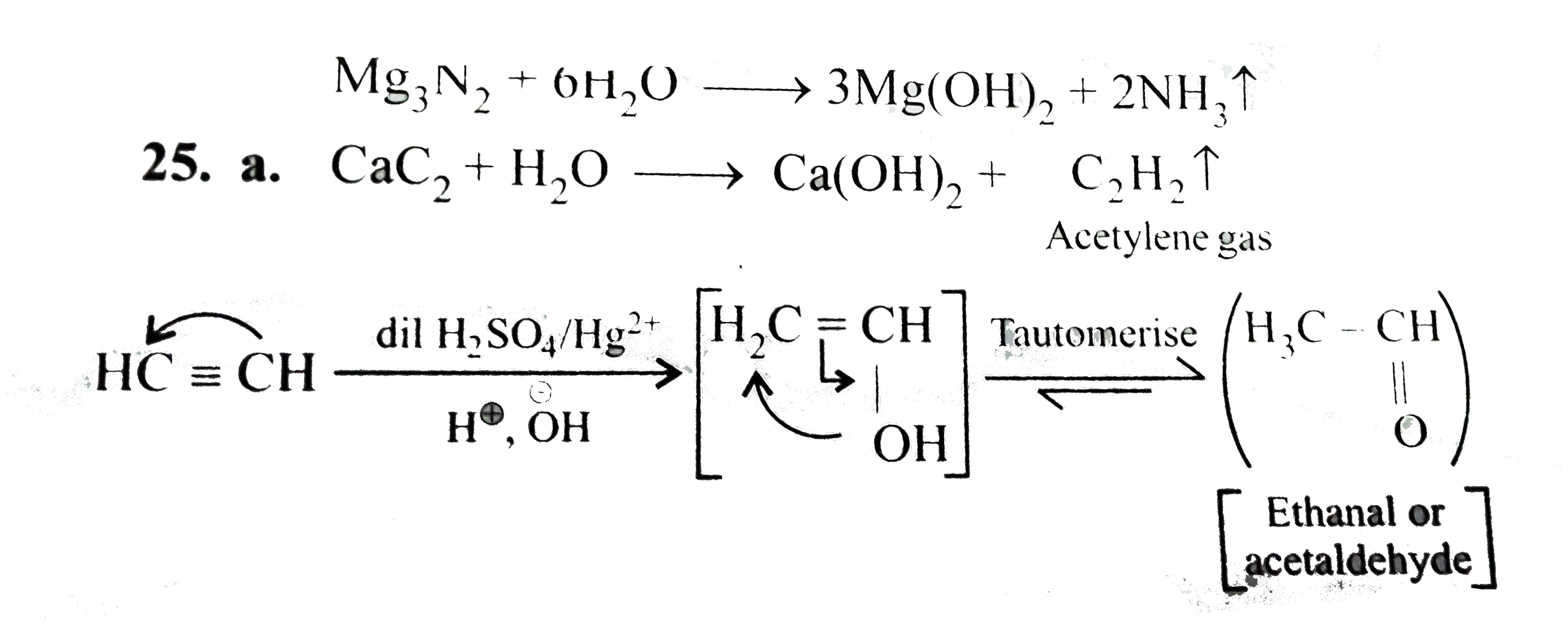 `CaC_(2)` reacts with `H_(2)O` to produce `CA(OH)_(2)` and acetylene, `C_(2)H_(2)` gas, when `C_(2)H_(2)` gas is further passed through `H_(2)SO_(4)` containing `HgSO_(4)`, acetaldehyde `(CH_(3)CHO)` is produced. (b). `MgCl_(2).6H_(2)O+NH_(4)ClrarrMgCl_(2).underset("Double salt")(NH_(4)Cl.6H_(2)O` `MgCl_(2).NH_(4)Cl.6H_(2)Ooverset(Delta)(rarr)MgCl_(2).NH_(4)Cl+6H_(2)O` `MgCl_(2).NH_(4)Cloverset(Delta)(rarr)MgCl_(2)+NH_(4)Cl` Hence, anhydrous `MgCl_(2)` is produced. (c). `FeCl_(3)+3H_(2)OrarrFe(OH)_(3)+3HCl` `Mg+2HClrarrMgCl_(2)+H_(2)` (d). `Mg+2NH_(4)ClrarrMgCl_(2)+2NH_(3)+H_(2)` (e). When `CO_(2)` is passed through limewater initially the solution becomes milky due to formation of `CaCO_(3)`. When excess of `CO_(2)` is passed, the solution becomes clear again due to the formation of `Ca(HCO_(3))_(2)` `underset("Limewater")(Ca(OH)_(2))+CO_(2)rarrunderset(("Milkiness"))(CaCO_(3))+H_(2)O` `CaCO_(3)+underset(("excess"))(CO_(2))+H_(2)Orarrunderset(("Soluble, hence the milkiness disappears and the solution becomes clear"))(Ca(HCO_(3))_(2))` (F). `Ca(OH)_(2)+SO_(2)rarrCaSO_(3)+H_(2)O` Solution becomes milky due to formation of `CaSO_(3)` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me