Saved Bookmarks
| 1. |
What is the formulae of inorganic benzene? Write the structure of Diborane and explain the nature of bonding in it. |
|
Answer» SOLUTION :Formulae of inorganic benzene is `B_(3)N_(3)H_(6)` Structure of Diborane 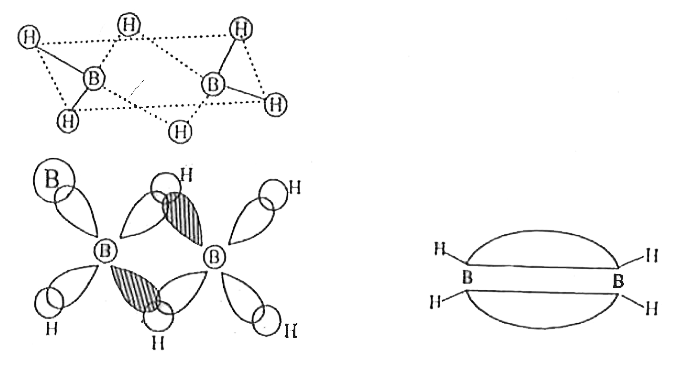 Bonding in diborane: Each B atom uses SP3 hybrides for boding. Out of the four sp3 hybrids one each B atom, one is without electrons shown in shaded region. The terminal B-H bonds are normal 2-centre-2electron bonds but the two bridge bonds adre 3-centre -2 -ELECTRON bridge bonds are also referreed to as banance bonds. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me