Saved Bookmarks
| 1. |
What is the hybrid state ofBeCl_(2) ? What will be the change in the hybrid state ofBeCl_(2)in the solidstate ? |
|
Answer» <P> Solution :In the vopour stateat hight temperature , `BeCl_(2)`existsas linear molecule , `Cl - Be - Cl`. The hybridizationof the central atom sp . In the solid state, it has a polymericwith chlorine bridges as follows : 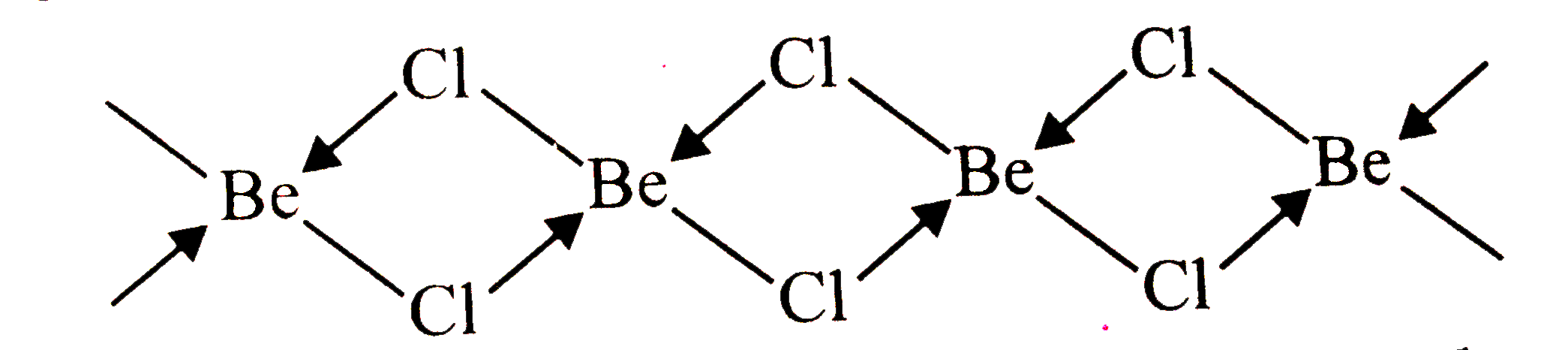 Two Clatoms are linked to Be atom by two coordinate bonds and two by covalent bonds. Forthese bonds to be formed , Be in the excited state with the configuration` 1s^(2)2s^(1) 2 p_(X)^(1) 2p_(y) ^(0) 2p_(Z)^(0)`undergoes ` sp^(3)`hydridisation . Two half-filled HYBRID orbitals will form normal covalent bonds with two Cl atom. The other two Cl atoms are COORDINATED to Be atom by donating ELECTRON pairs into the empty hybrid orbitals |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me