Saved Bookmarks
| 1. |
What will be the work done on an ideal gas enclosed in a cyliner, when it is compressed by a constant external pressure, p_("ext") in a single step as shown in figure ? Explain graphically. |
|
Answer» Solution :Suppose total volume of the gas is `V_(i)` and pressure of the gas inside CYLINDER is p. After compression by constant external pressure, `(p_("ext"))` in a single step, final volume of the gas becomes `V_(f)`. Then volume change, `Delta v = (V_(f) - V_(i))` If W is the work done on the system by movement of the piston, then `W = P_("ext") (-Delta V)` `W = - p_("ext") (V_(f) - V_(i))` This can be calculated from `p - V` GRAPH as shown in the figure. Work done is equal to the shaded are `ABV_(f)V_(i)` 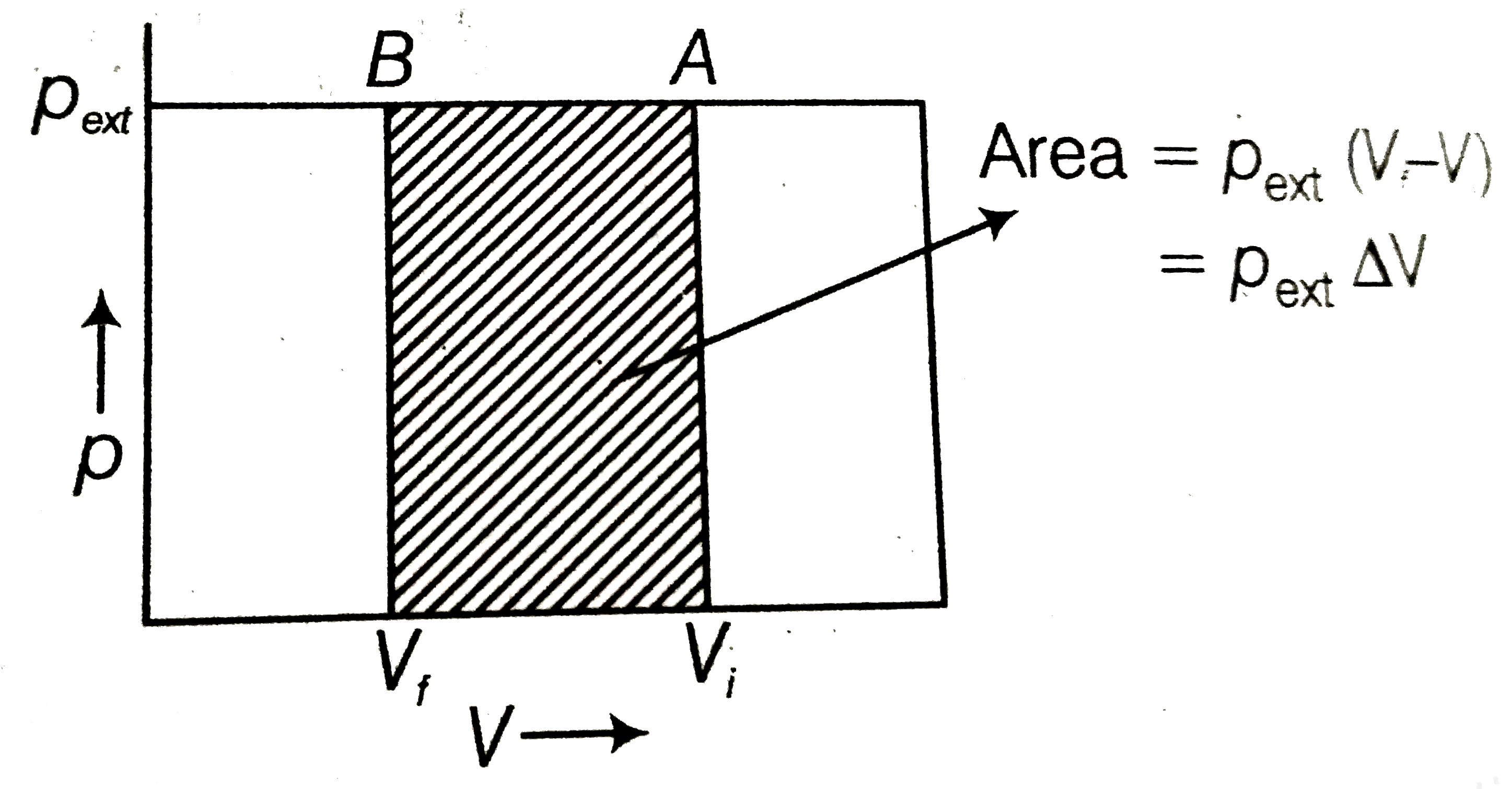 The negative sign in this expression is required to obtain CONVENTIONAL sign for W which will be POSITIVE. Because in case of compression work is done on the system, so `Delta V` will be negative |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me