Saved Bookmarks
| 1. |
When a mole of magnesium bromide is prepared from 1 mole of magnesium and 1 moleof liquid bromine, 524 kJ of energy is released. The heat of sublimation of Mg metal is 148 "kJ mol"^(-1). The heat of dissociation of bromine gas into atoms is 193 "kJ mol"^(-1). The heat of vaporization of liquid bromine is 31 kJ "mol"^(-1). The ionisation energy of magnesium is 2187 kJ mol^(-1) and the electron affinity of bromine is - 662 "kJ mol"^(-1). Calculate the lattice energy of magnesium bromide. |
|
Answer» Solution :Given : `MG(s) + Br_2(l) to MgBr_(2)(s)"" DeltaH_f^0=-524 "KJ MOL"^(-1)` Sublimation : `Mg(s) to Mg(g) "" DeltaH_1^0=+148 "KJ mol"^(-1)` Ionisation : `Mg(g) to Mg^(2+)(g) +2e^(-) "" DeltaH_2^0 =2187 "KJ mol"^(-1)` Vapourisaton : `Br_2(l) to Br_(2)(g)"" DeltaH_3^0=+31 "KJ mol"^(-1)` Dissociation : `Br_2(g) to 2Br(g) "" DeltaH_4^0=+193 "KJ mol"^(-1)` Electron AFFINITY : `Br(g) +E^(-) to Br^(-) (g) "" DeltaH_5^0=-331 "KJ mol"^(-1)` 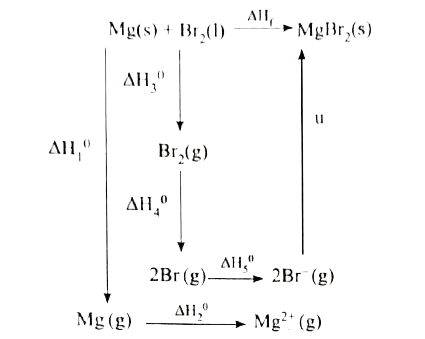 `DeltaH_f=DeltaH_1 + DeltaH_2+DeltaH_3+DeltaH_4+2DeltaH_5+u` -524=148+2187+31+193+(2 x -331) + u -524 =1897 + u u=-524 - 1897 `u=-2421 "kJ mol"^(-1)` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me