Saved Bookmarks
| 1. |
When BCl_(3) is treated with water, it hydrolyses and forms [B(OH)_(4)]^(-) only whereas AlCl_(3) in acidified aqueous solution forms [Al(H_(2)O)_(6)]^(3+)ion, Explain what is the hybridisation of boron and aluminium in these species? |
|
Answer» SOLUTION :In trivalent state, most of the compounds being covalent are HYDROLYSED in water, e.g., `BCl_(3)` on HYDROLYSIS in water form `[B(OH)_(4)]^(-)` species, the hybridisation state of B is `sp^(3)` `{:(BCl+3H_(2)OrarrB(OH)_(3)+3HCl),(B(OH)_(3)+H_(2)Orarr[B(OH)_(4)]^(-)+H^(+)):}` `AlCl_(3)` in acidified aqueous solution form OCTAHEDRAL `[Al(H_(2)O)_(6)]^(3+)` ion. In this complex, the 3d orbital of Al are INVOLVED and the hybridisation state of Al is `sp^(3)d^(2)` 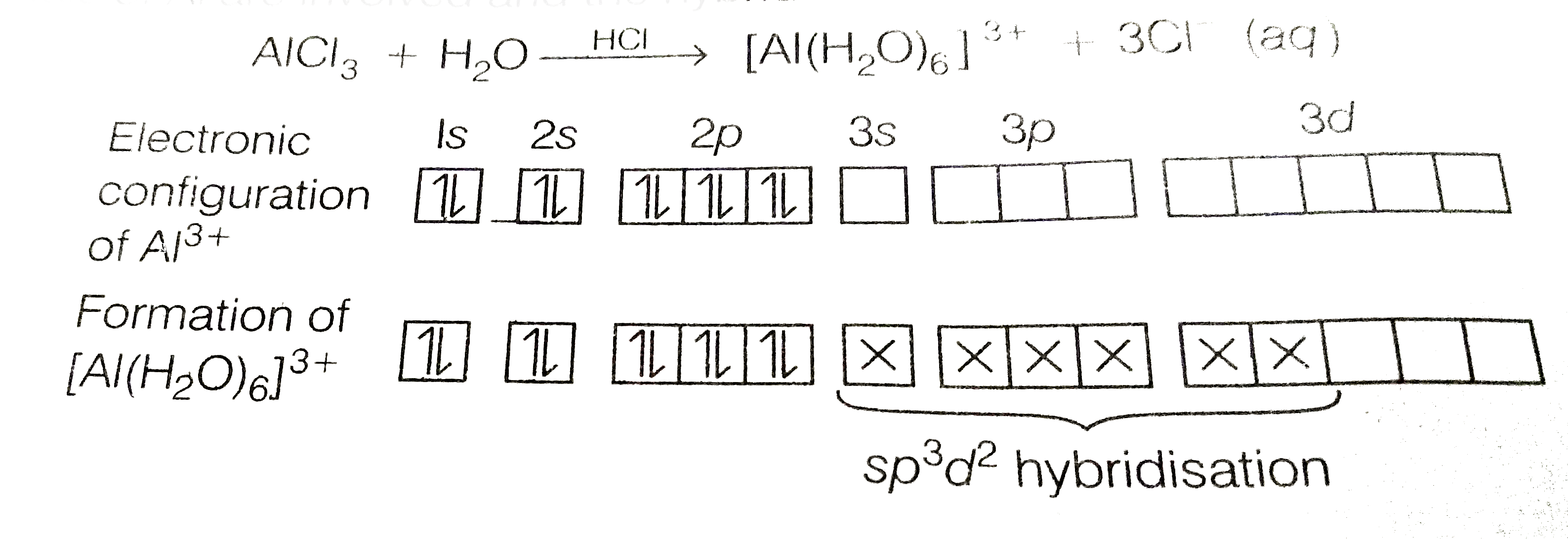
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me