Saved Bookmarks
| 1. |
Which element in 3d, 4d, and 5d transition series has the highest paramagnetism in (a) elemental form, (b) +1 O.S. ( c) +2 O.S., (d) +3 O.S. [O.S. ="oxidation state"] |
Answer» Solution :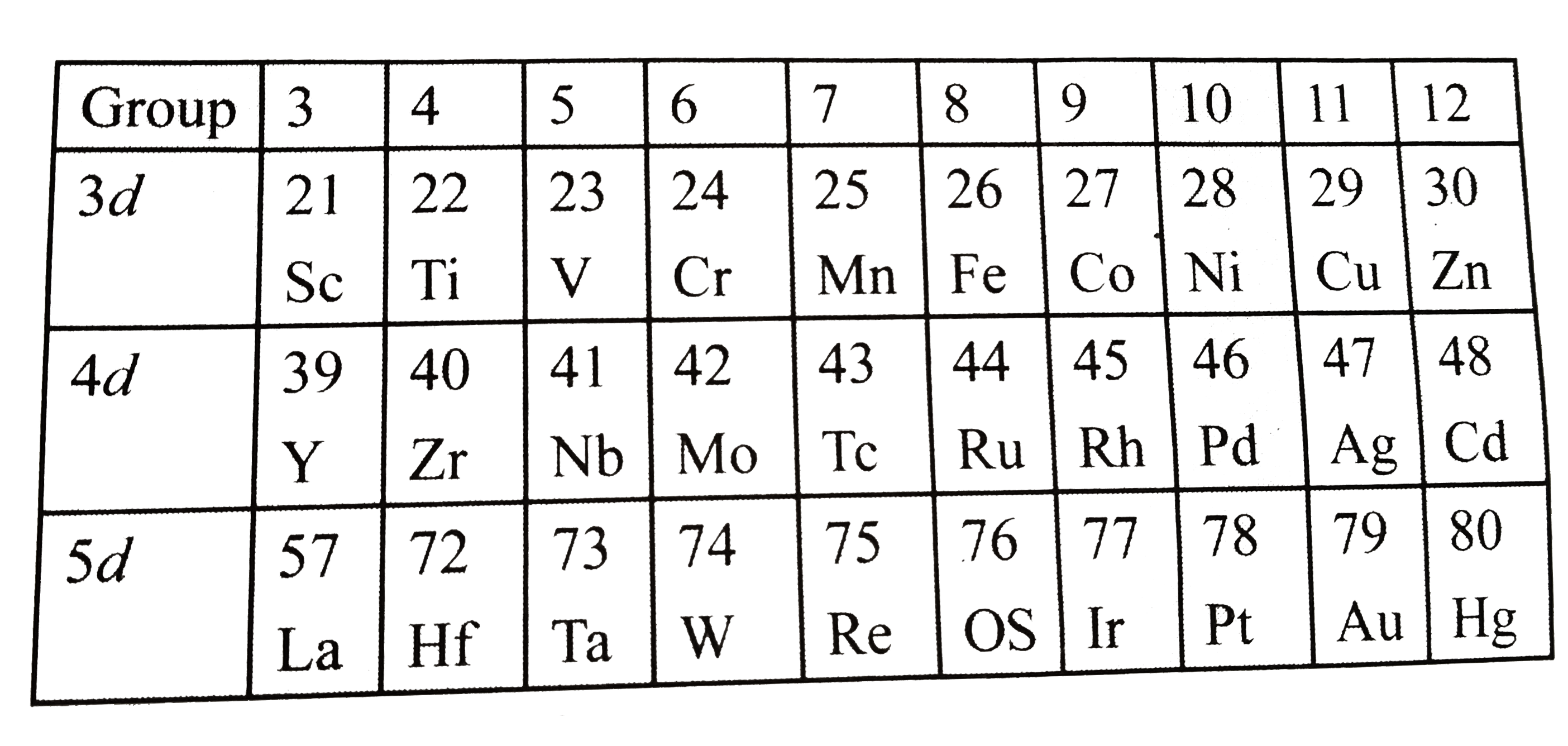 (a) In elemental form: `3d` series: `Cr (Z = 24) RARR 3d^(5) 4S^(1),n = 6, mu = sqrt(6+(6+2))` `= sqrt(48) BM` `4d` series: `Mo(Z=42) rArr 4d^(5) 5s^(1),n=6, mu = sqrt(48) BM` `5d` series: `W(Z=74) rArr 5d^(4) 4s^(2),n=4, mu = sqrt(24) BM` But `Re` has the highest number of unpaired `e^(-)`, as shown: `Re(Z=75) rArr 5d^(5) 6s^(2),n = 5, sqrt(35) BM` (b) `In + 1` oxidation state: `3d` series: `Mn(Z=25) rArr3d^(5) 4s^(2), Mn^(1+) = 3d^(5) 4s^(1), n=6, mu = sqrt(48) BM` `4d` series: `Te(Z = 43) rArr 4d^(5) 5s^(2), Te^(1+) = 4d^(5) 5s^(1), n=6, mu = sqrt(48) BM` `5d` series: `Re(Z=75) rArr 5d^(5) 6s^(2),Re^(1+)=5d^(5) 6s^(-),n=6, mu=sqrt(48) BM` (c ) `In+2` oxidation state: `3d` series: `Mn(Z=25) rArr 3d^(5) 4s^(2), Mn^(2+) = 3d^(5), 4s^(0),n=5, mu = sqrt(35) BM` `4d` series: `Te(Z=43) rArr 4d^(5) 6s^(2), Te^(2+)=4d^(5) 5s^(0),n=5, mu = sqrt(35) BM` `5d` series: `Re(z=75) rArr 5d^(5) 6s^(2), Re^(2+)=5d^(5) 6s^(0),n=5, mu = sqrt(35) BM` (d) `In+3` oxidation state: `3d` series: `Fe(Z=26) rArr 3d^(6) 4s^(2), Fe^(3+)=3d^(5) 4s^(0),n=5, mu = sqrt(35) BM` `4d` series: `Ru(Z=44) rArr 4d^(7) 5s^(1)` (exxeption electronic configuration). `5d` series: `Os (Z=76) rArr 5d^(6) 7s^(2), Os^(3+) = 5d^(5) 7s^(0), n=5, mu = sqrt(35) BM` Note: (i) From the abov calculation it is clear that in the elemental form (zero `OS`), group `6` has the highest `mu`, except `W`, in `5d` series, but it is `Re` of group `7`. (ii) `In+1 OPS`, group `7` has the highest `mu`. (iii) `In+2 OS`, again group `7` has the highest `mu`. (iv) `In+3 OS`, group `8` has the highest `mu`. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me