Saved Bookmarks
| 1. |
Which of the following are correct ? |
|
Answer» `CH_3-O-CH_2^(o+)` is more stable than `CH_3-CH_2^(o+)`  `CH_(3)O overset(+)(C)H_(2)` is stabilized by +I effect of the two `CH_(3)` groups while 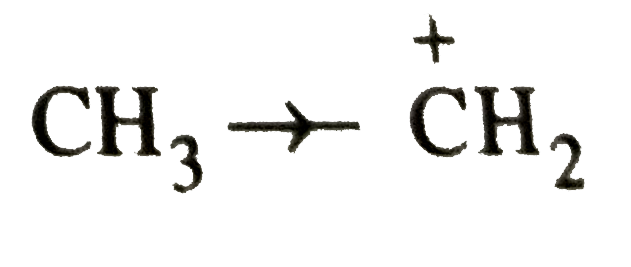 is stabilized by only weak +I- effect of the `CH_3` GROUP. Thus, option (a) is CORRECT.  is stabilized by +I-effect of the ethyl group. SINCE +I-effect of the ethyl group is weaker than +I-effect of the two `CH_3` groups, therefore, `(CH_3)_2CH^+` is more stable than `CH_3CH_2-overset+CH_2` group. Thus , option (b ) is wrong. is stabilized by +I-effect of the ethyl group. SINCE +I-effect of the ethyl group is weaker than +I-effect of the two `CH_3` groups, therefore, `(CH_3)_2CH^+` is more stable than `CH_3CH_2-overset+CH_2` group. Thus , option (b ) is wrong. is stabilized by strong resonance effect while, is stabilized by strong resonance effect while, 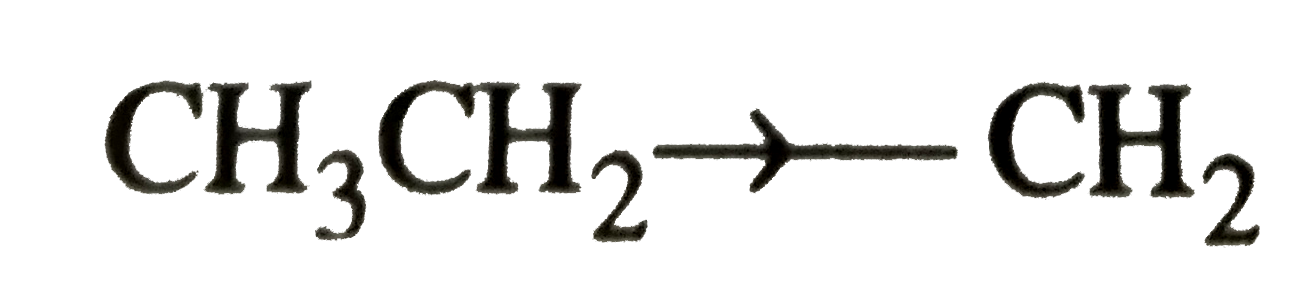 is stabilized by weak +I-effect of the `CH_2CH_3` group. The option (c ) is correct. In `CH_2=CH^+` , +ve charge is present on the more electronegative sp-hybridised carbon while in `CH_3-overset+CH_2`, +ve charge is present on the more electronegative sp-hybridized carbon while in `CH_3-overset+CH_2`. , +ve charge is present on the less electronegative `sp^2`-hybridized carbon, therefore , `CH_2=overset+CH` is less stable than `CH_3-overset+CH_2` . Thus option (d) is wrong. is stabilized by weak +I-effect of the `CH_2CH_3` group. The option (c ) is correct. In `CH_2=CH^+` , +ve charge is present on the more electronegative sp-hybridised carbon while in `CH_3-overset+CH_2`, +ve charge is present on the more electronegative sp-hybridized carbon while in `CH_3-overset+CH_2`. , +ve charge is present on the less electronegative `sp^2`-hybridized carbon, therefore , `CH_2=overset+CH` is less stable than `CH_3-overset+CH_2` . Thus option (d) is wrong.
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me