Saved Bookmarks
| 1. |
Which of the following statements are correct ? (I) In ICl_(2)-CIF_(3) and TeCl_(4), the number of ions pairs (s) of electrons on central atoms are 3,2 and 1 respectively. (II) Amongst CO,CO_(2),CO_(3)^(2-),CH_(2)OH the correct order from the weakest to the strongest carbon-oxygen bond -=CH_(3)OH lt CO_(3)^(2-) lt CO_(2) lt CO. (III) The I^(-),b^(-)idisation of boron in BF_(2) is the same which nitrogen has in CINO molecule. |
|
Answer» Only `(I) & (III)` 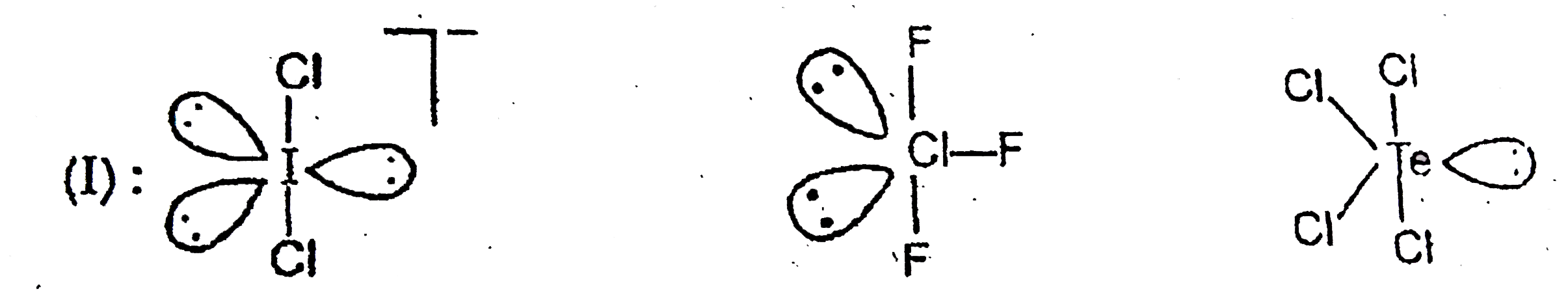 `(II)" ":C-O=1.43Å,C=O" "1.23Å,C=O" "1.09Å` `CO,Coverset(LARR)(-=)=O` partial triple bond character, borid LENGTH is intermediat of `C=O` and `C-=O` bonds `CO_(2),O=C=O,` double bond length 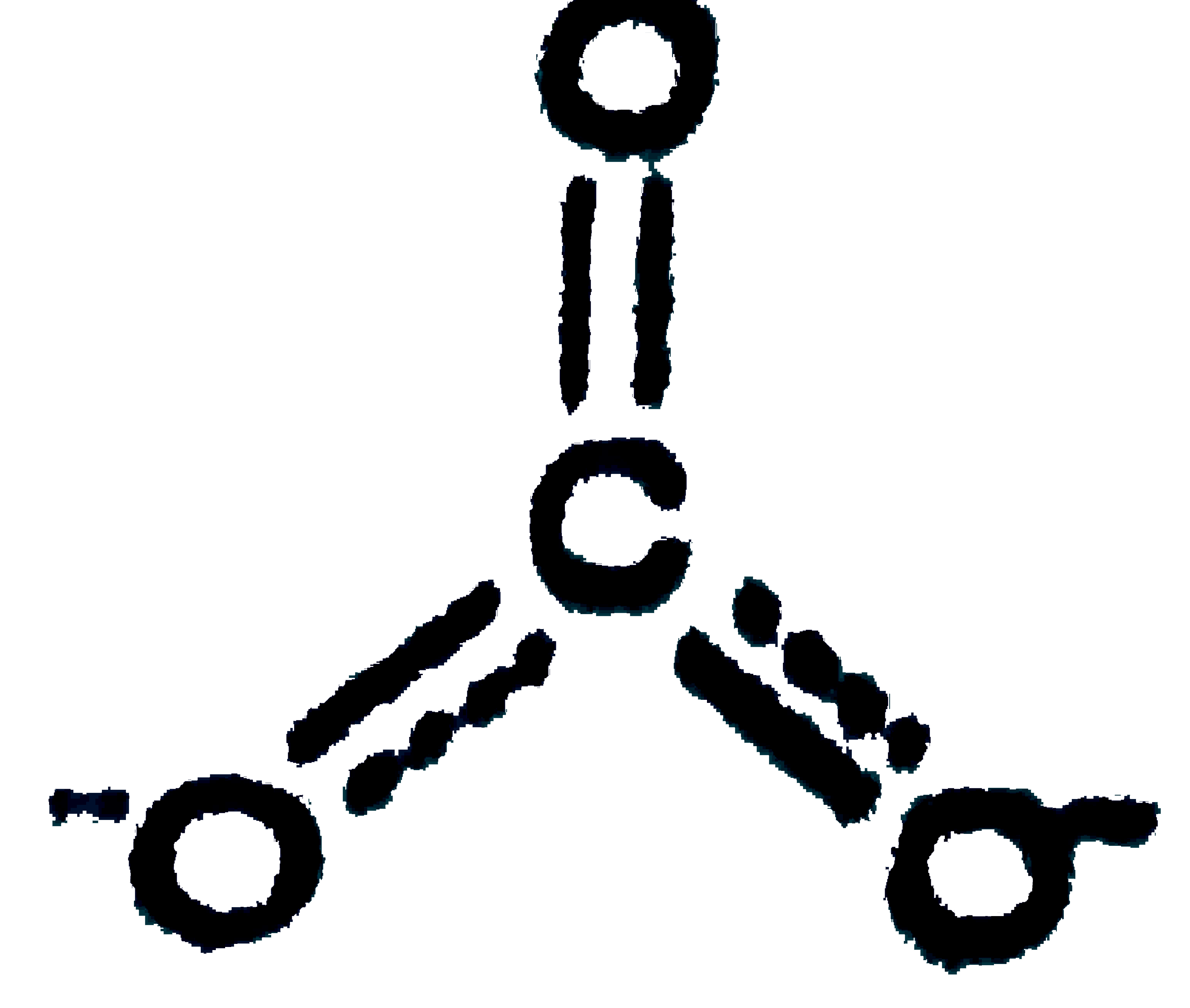 partial double bond character, intermediate of `C=O` and `C-O` partial double bond character, intermediate of `C=O` and `C-O` 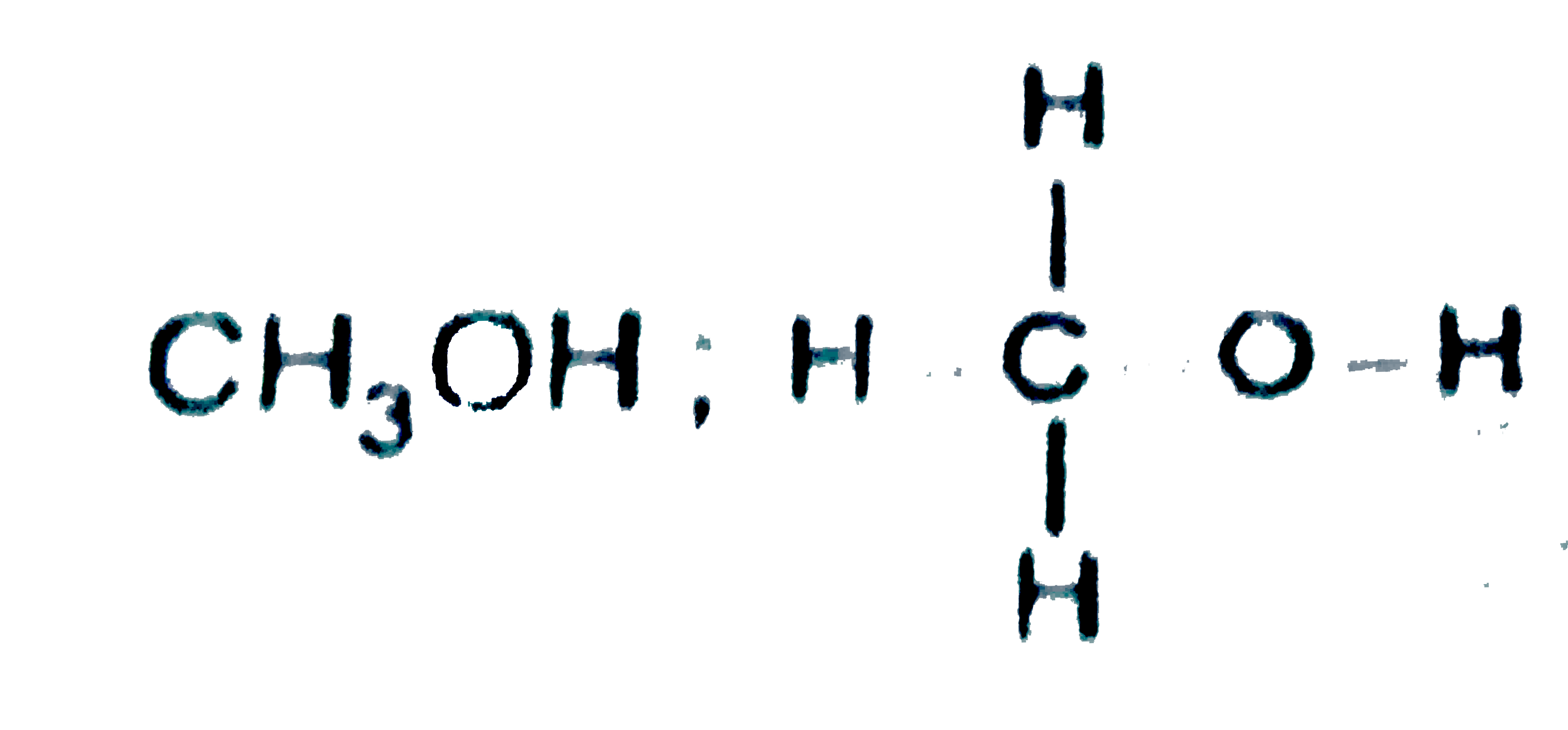 single bond length. single bond length. 
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me