Saved Bookmarks
| 1. |
Which of the following statements is/are correct about the reaction. H_(2)SO_(4)+Cu_(3)P+Cr_(2)O_(7)^(2-)toCuSO_(4)+H_(3)PO_(4)+2Cr^(3+) |
|
Answer» The number of moles of `Cr_(2)O_(7)^(2-)` required to oxidise 6 " mol of "`Cu_(3)P` to `CuSO_(4)` and `H_(2)PO_(4)` is 11 mol. `Cr^(1+)to3Cu^(2+)3e^(-)` ("oxidation")` `underline(underset(x=-3)(P^(3-))toundersetunderset(x=5)(x+x-8=0)(H_(3)PO_(4)))` `underline(4H_(2)O+Cu_(3)Pto3Cu^(2+)+H_(2)PO_(4)+11e^(-)+5H^(o+))` REDUCTION: `Cr_(2)O_(7)^(2-)+6e^(2-)+14H^(o+)to2Cr^(3+)+7H_(2)O`..(ii) Multiply equation (i) and 6 and equation (ii) by 11, net redox equation is: 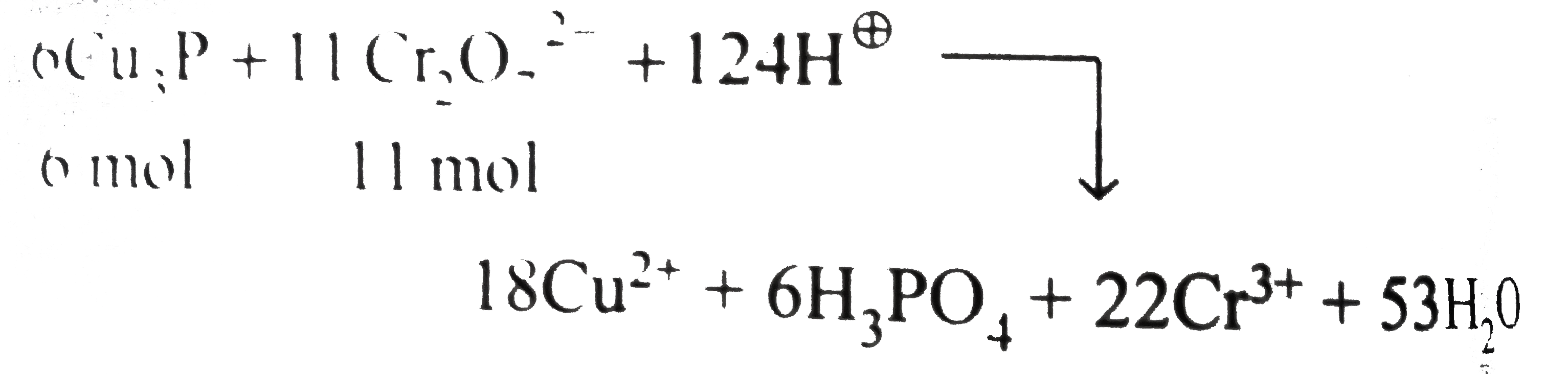 (ii). Number of moles of `H_(2)SO_(4)` used `=62H_(2)SO_(4)` `(=124H^(o+)=62SO_(4)^(2-))` Number of moles of `CuSO_(4)` formed`=18CuSO_(4)(=18SO_(4)^(2-))` ltBrgt Number of moles of `Cr_(2)(SO_(4))_(3)` formed`=11Cr_(2)(SO_(4))_(3)` ltBrgt `(=33SO_(4)^(2-))` Total number of `SO_(4)^(2-)` ION in reactant `=62` Total number of `SO_(4)^(2-)` ion in product`=18+33=51` Rest 11 " mol of "`SO_(4)^(2-)` ion in the reactant react with `(11)/(2mol)` of `K^(o+)` ion to give 11 " mol of "`K_(2)SO_(4)`. Net redox reaction is: `6Cu_(3)P+11K_(2)Cr_(2)O_(7)+62H_(2)SO_(4)to18CuSO_(4)` `+6H_(3)PO_(4)+43H_(2)O+11Cr_(2)(SO_(4))_(3)+11K_(2)SO_(4)` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me