Saved Bookmarks
| 1. |
Write down the products of ozonolysis of 1,2-dimethylbenzene (o-xylene) . How does the result support Kekule structure of benzene ? |
Answer» SOLUTION :o-Xylene may be regarded as a resonance hybrid of the following two kekule structures. Ozonolysis of each one of these GIVES two productsas SHOWN below : 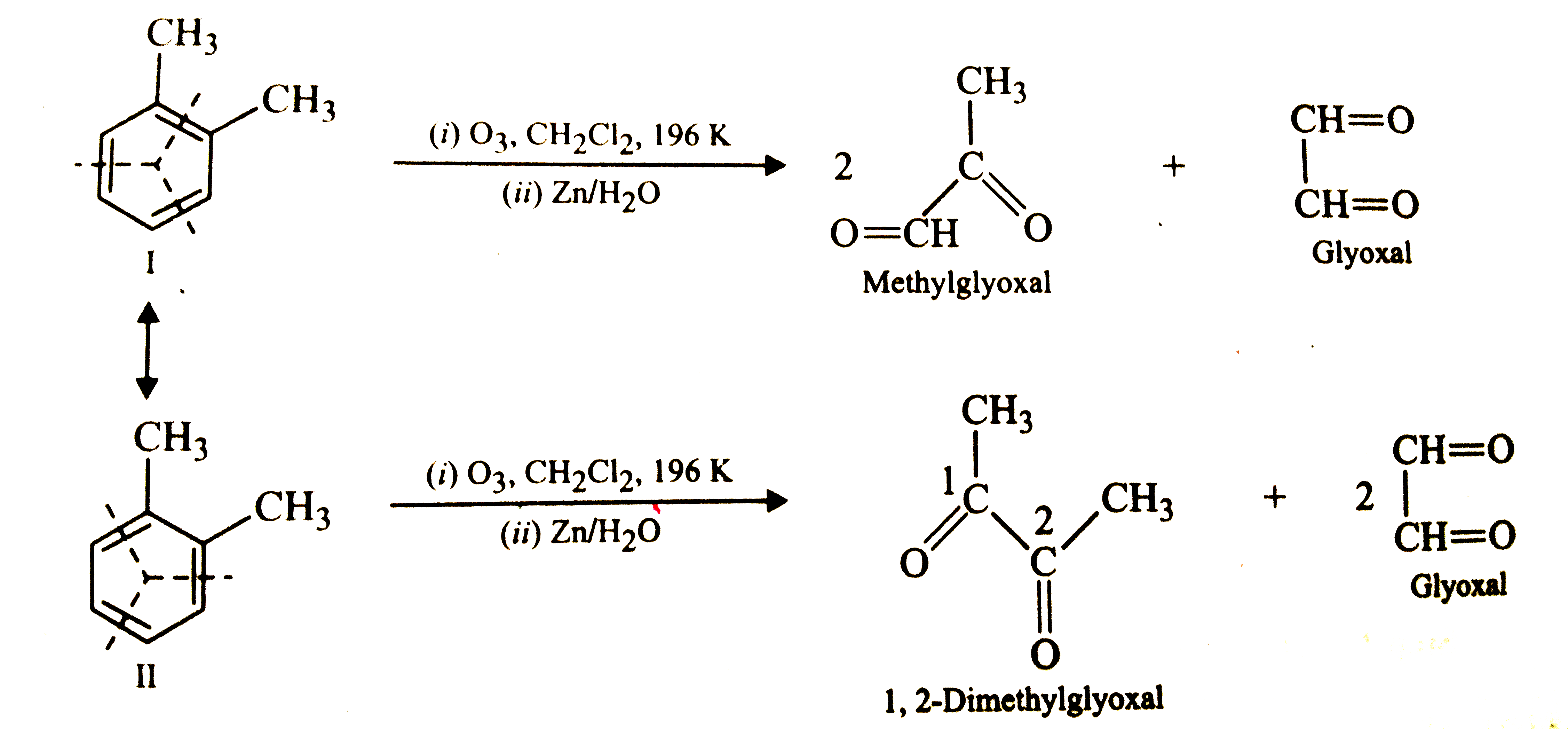 Thus, in all, THREE PRODUCTS are formed . Since, all the three products cannot be obtained from any one of the two Kekule structures, this shows that o-xylene is a resonance hybrid of the two Kekule structures (I and II) |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me