Saved Bookmarks
| 1. |
Write hydrocarbon radicalsthat can be formed as intermediates during monochlorination of 2-methylpropane ? Which of them is more stable ? Give reasons . |
|
Answer» Solution :2-methylpropane has two sets of equivalent hydrogens marked as 'a' and 'B' and hence GIVES two radicals, I and II. `undersetI(oversetalphaCH_3-underset*oversetoversetoversetalpha(CH_3)|C-oversetalphaCH_3) larr underset"2-Methylpropane"(oversetalphaCH_3-undersetboversetoversetoverseta(CH_3)|C-oversetaCH_3) to underset"II"(CH_3-oversetoverset(CH_3)|(.^alphaCH)-oversetdotCH_2)` Radical (I) is more STABLE than radical (II) because of the following two reasons : (i)Radicals (I) is `3^@` while radical (II) is `1^@` (ii)Radical (I) has nine `ALPHA`-hydrogen and hence is STABILIZED by nine hyperconjugation structues while radical (II) has only one `alpha`-hydrogens and is stabilized by one hyperconjugation structures as shown below : 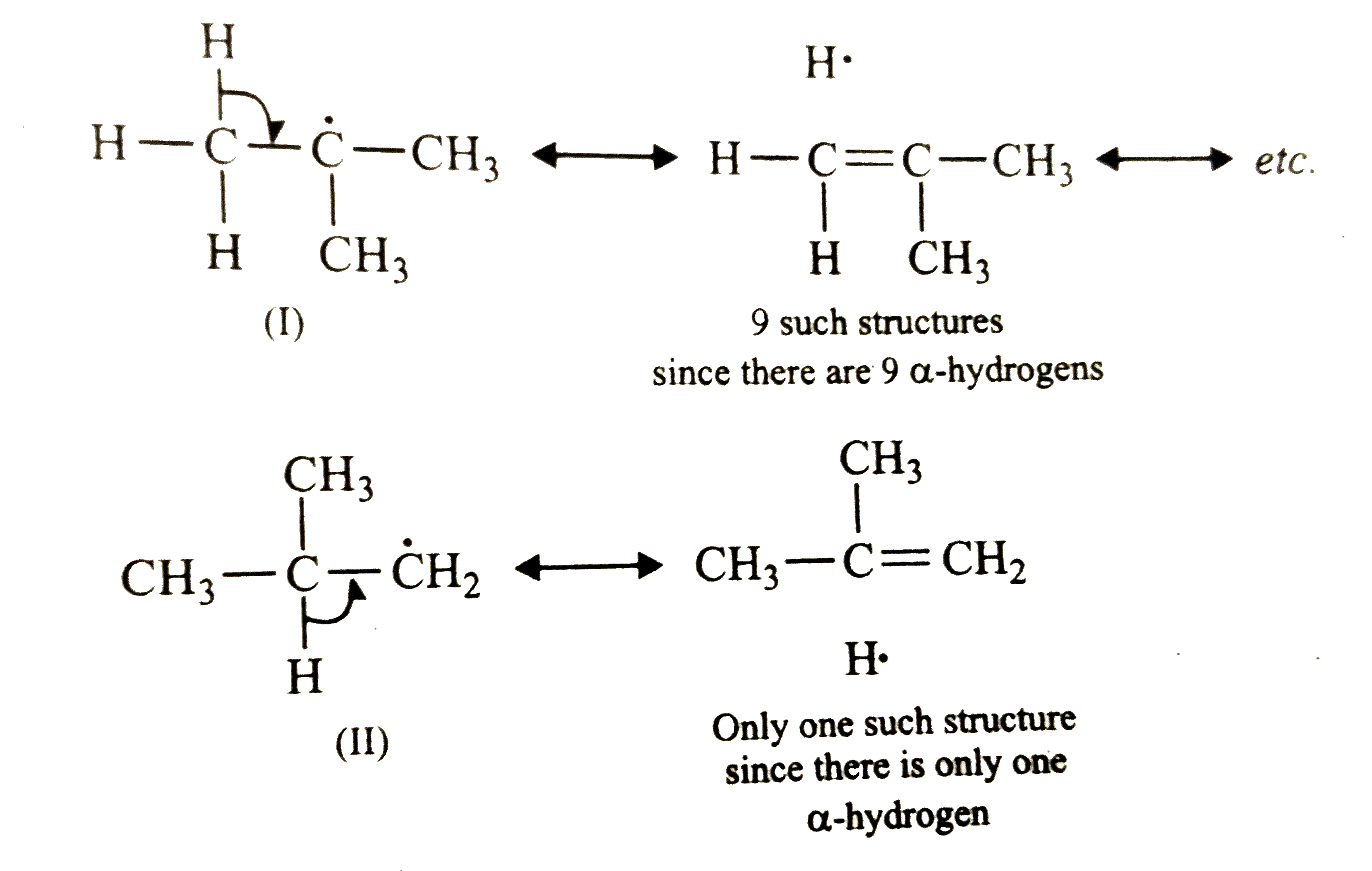
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me