Saved Bookmarks
| 1. |
Write the Born-Haber cycle for NaCl. |
Answer» Solution :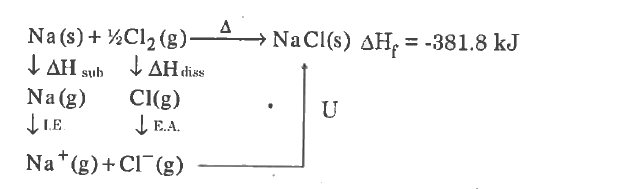 When U= Littice energuy `: Delta H_(s)` = Enthaply of SUBLIMATION,= Enthalpy of ionization, `Delta H_(4)=` enthalpy of dissociatio, `Delta H_(i)=DeltaH_(EA)` enthalpy for ELECTRON affinity. The only one step form formation of NaCl from sodium cholorine is associated with the energy called `Delta H_(f)` = = enthalpy of formation. `:. Delta H_(f)= U+DeltaH_(s)+DeltaH_(i)+DeltaH_(d)DeltaH_(Ea)` `:.` Lattice energy `U=DEltaH_(f)=[DeltaH_(s)+DeltaH_(i)+DeltaH_(d)+DeltaH_(Ea)]` |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me