Saved Bookmarks
| 1. |
Write the energy level diagram of oxygen molecule. |
|
Answer» Solution :Atomic number of oxygen =8. Electronic configuration of oxygen `=1s^(2)2s^(2)2P^(4)`. When two oxygen atoms COMBINES, the molecular orbital energy level diagram is as shown in the figure. From the diagram, the molecular electronic configuration of oxygen is `sigma_(1s^(2)),sigma_(1s^(2))^(***),sigma_(2s^(2))^(***),sigma_(2p_(2)^(2)),{pi_(2p_(x))^(2)=pi_(2p_(y))^(2)}` `{pi_(2p_(x)^(1))^(***)=pi_(2p_(y)^(1))^(***)}` Bond order `=("no. of BMO"e^(-)-"no. of"ABMOe(-))/2` `=(N_(be)-N_(ABE))/2` 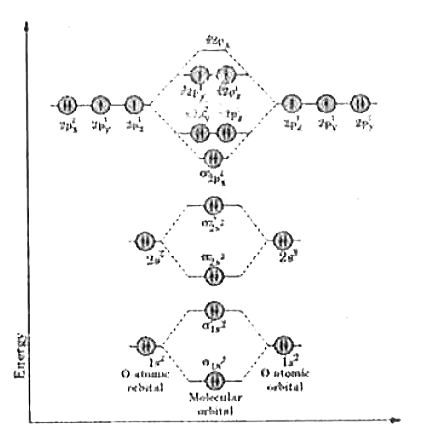 Here BMO electrons `sigma_(1s)^(2),sigma_(2s^(2)),sigma_(2p_(x)^(2)),{pi_(2p_(x)^(2)=pi_(2p_(y)^(2))}` i.e. `N_(be)=10` ABMO electrons `sigma_(1s^(2))^(***),sigma_(2s^(2))^(***),sigma_(2p_(x)^(1))^(***){pi_(2p_(x)^(1))^(***)=pi_(2p_(y)^(1))^(***)}`, i.e. `N_(abe)=6` `BO=(10-6)/2=4/2=2` i.e. between oxygen there exists double bond. Magnetic property: Therefore are two UNPAIRED electrons `{pi_(2p_(x)^(1))^(***)=pi_(2p_(y)^(1))^(***)}` therefore, oxygen molecule is paramagnetic in nature. |
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me