Saved Bookmarks
| 1. |
Write the IUPAC names of the following alkanes: (i) (CH_(3))_(2)CHCH_(2)CH_(3) (ii) (CH_(3))_(2)CH-CH(CH_(3))_(2) (iii) (CH_(3)CH_(2))_(2)CH-CH(CH_(3))_(2) ltbr (iv) CH_(3)CH_(2)underset(CH_(3))underset(|)(CH)CH_(3)underset((CH_(2))_(2)CH_(3))underset(|)(CHCHCH_(3)CH)(CH_(3))_(2) (v) (CH_(3))_(2)CHCH_(2)CHCH_(3)CH(CH_(3))_(2) (vi) (CH_(3))_(3)C CH_(2)C(CH_(3))_(2) (vii) (viii) Isobutane (ix) Neopentane (x) Isooctane (xi) (xii) (xiii) (xiv) (xv) 4-tert-Butyloctane Strategy: Open up the structure if brancehes are present. Select the longest possible continous C chain carrying the maximum number of branches as the parent chain. Number the parent chain usingthe smallest possible number or lowest set of locants for the substituents (s). Assign the name and position number to each and every substituents. Write the name as a single word. Use hyphens to seperate numbers and letters. Do not leave any spaces. |
Answer» Solution :(i) 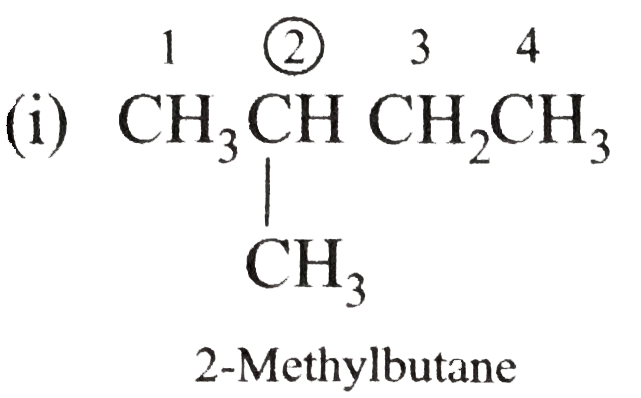 We have numbered the LONGEST chain beginning with the end of the chain nearer the substituents. (II) 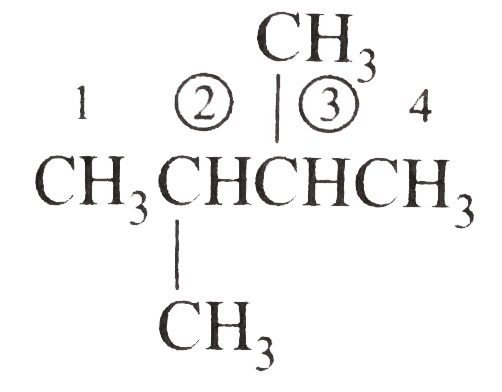 The presence to two identical substituents is INDICATED by the prefix `di`. Every substituent is given a number corresponding to its location on the longest chain. Normally, students make the mistake of not using the prefix `di` (iii) 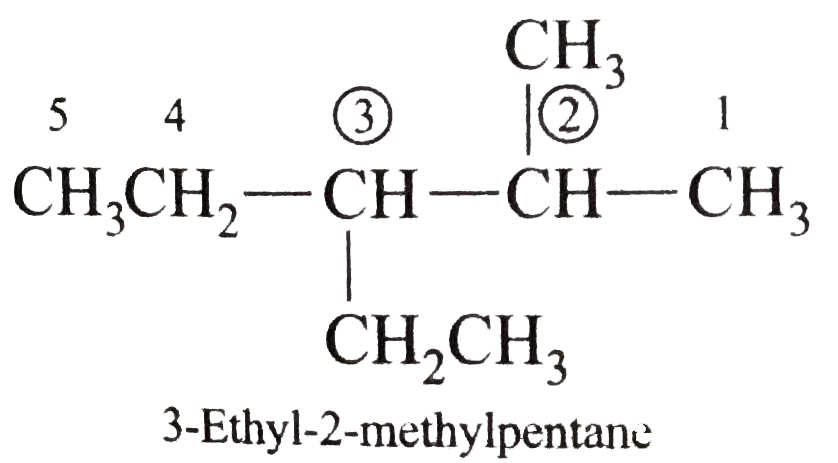 The names of DIFFERENT substituents arel listed alphabetically (iv) 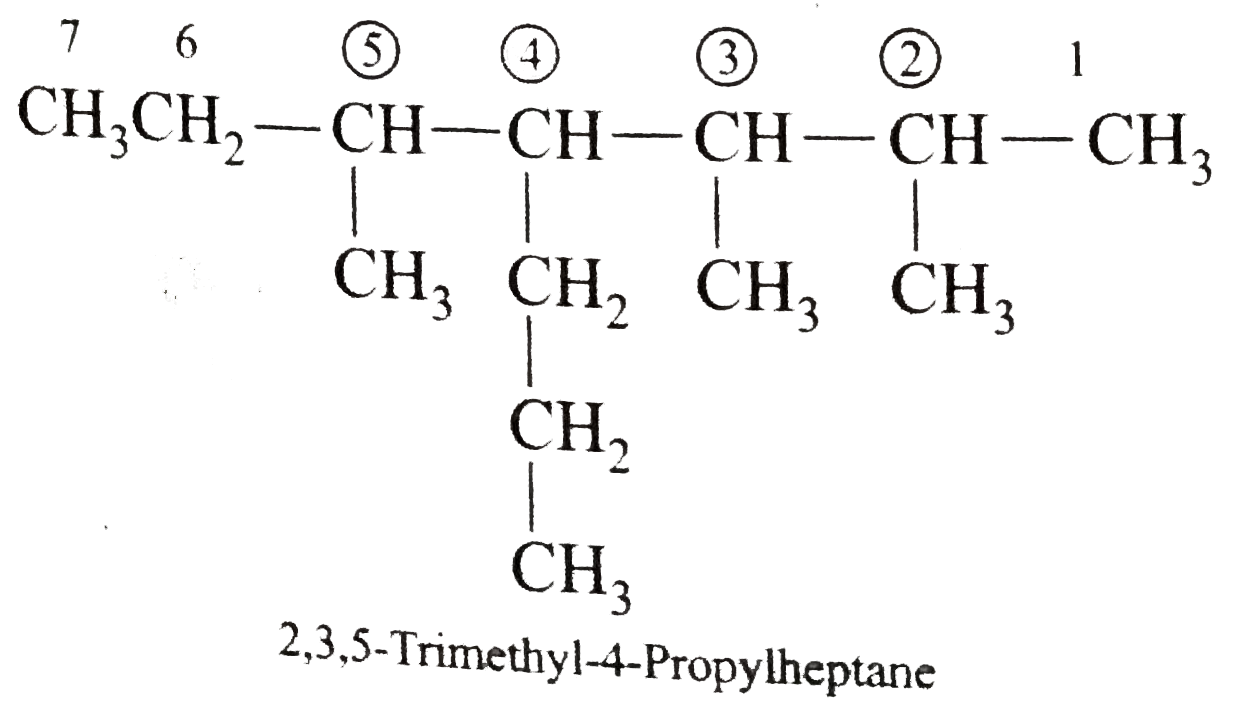 The selected parent chain carries the maximum number of branches. Commas are used to separate numbers from each other. (v) 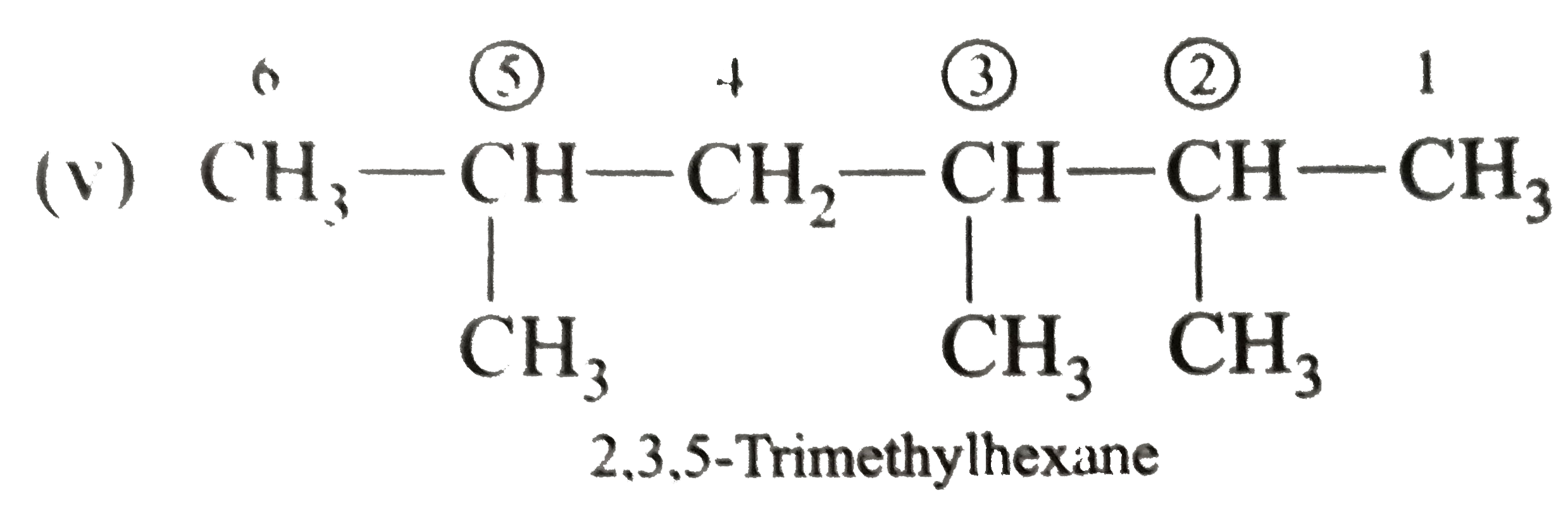 The numerical prefix tri indicates the presence of threee methyl group on the parent chain. (vi) 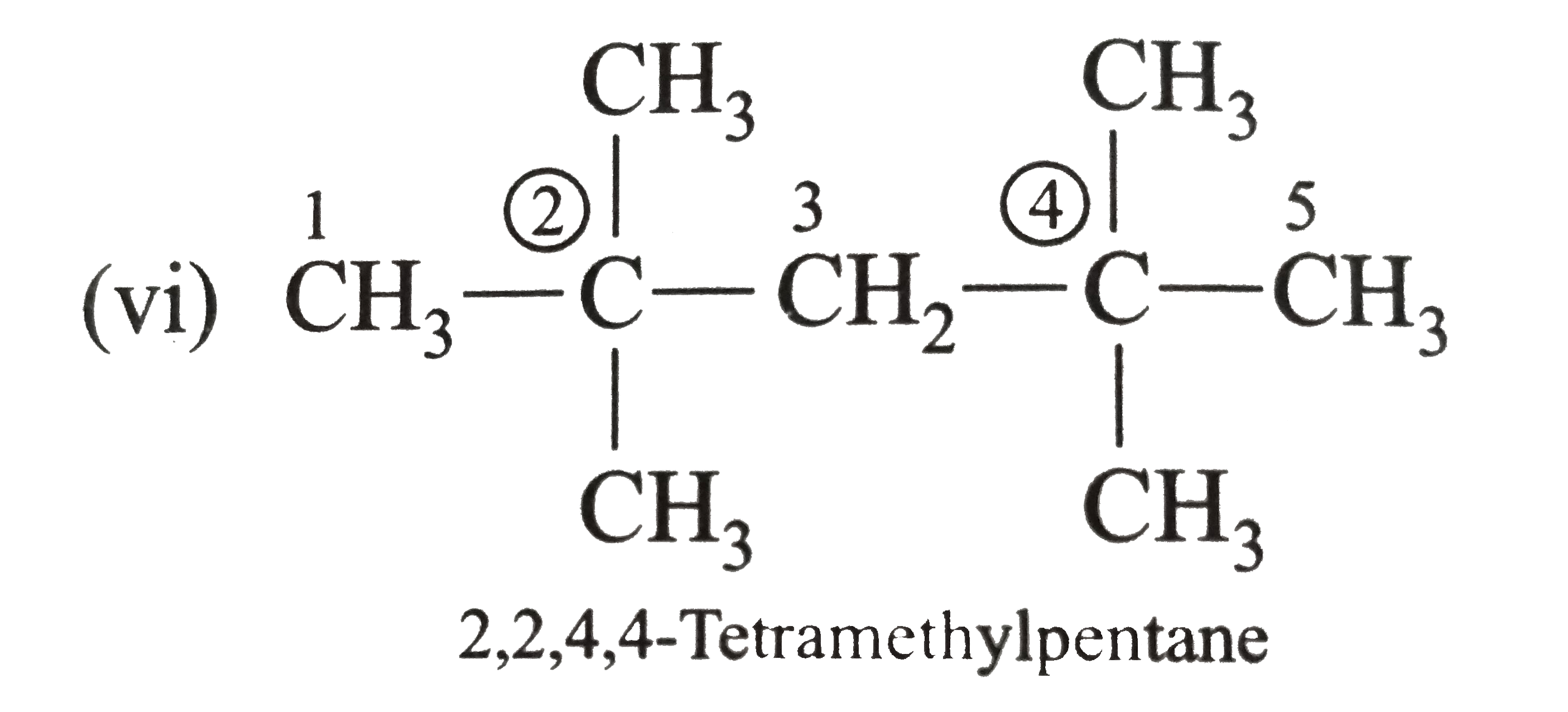 The numerical prefix tetra indicates the presence of four methyl groups on the parents chain. (vii) 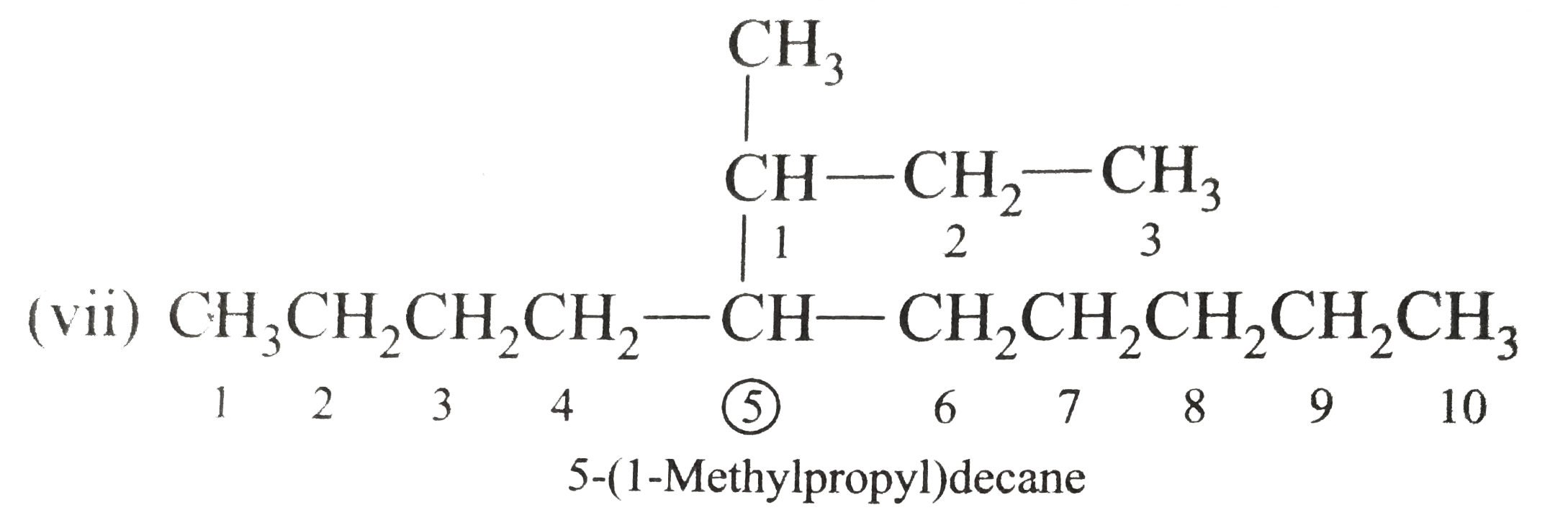 The name of complex substitutent is ALWAYS enclosed within parentheses. 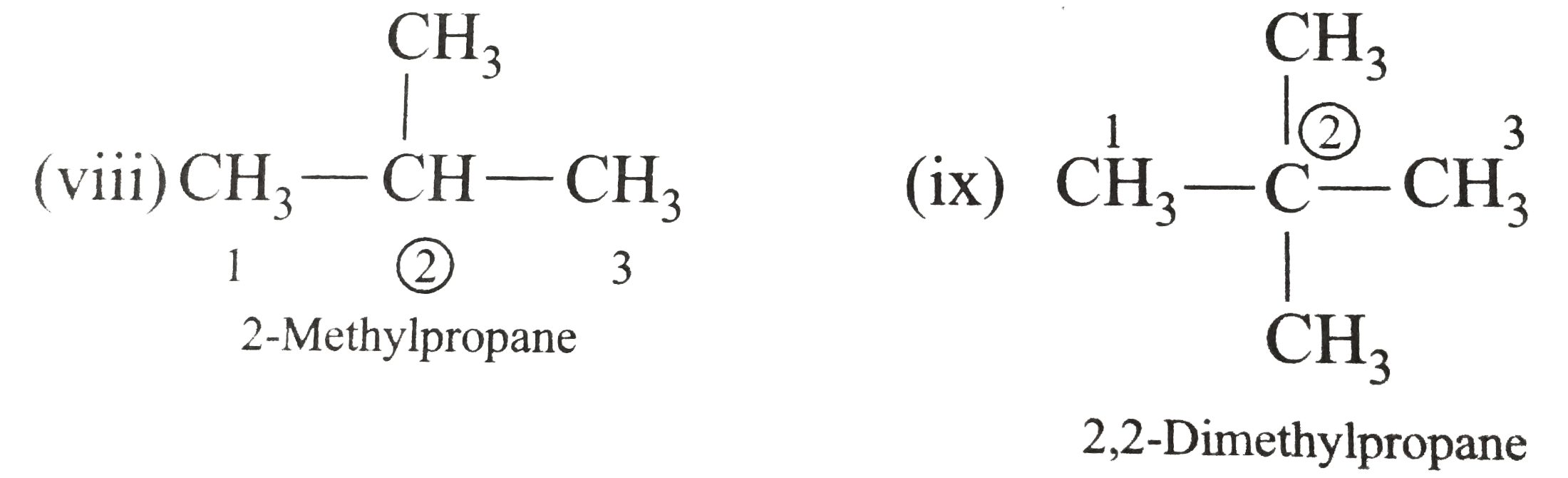 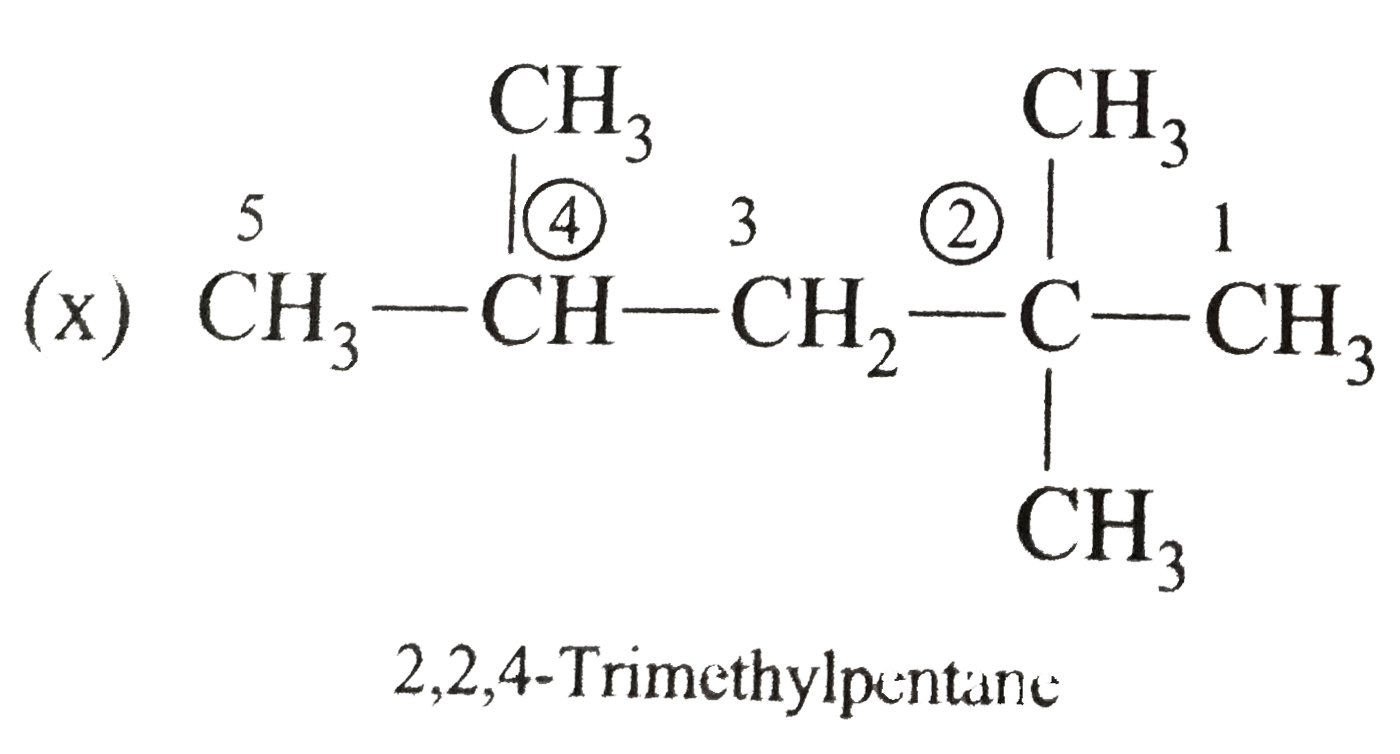 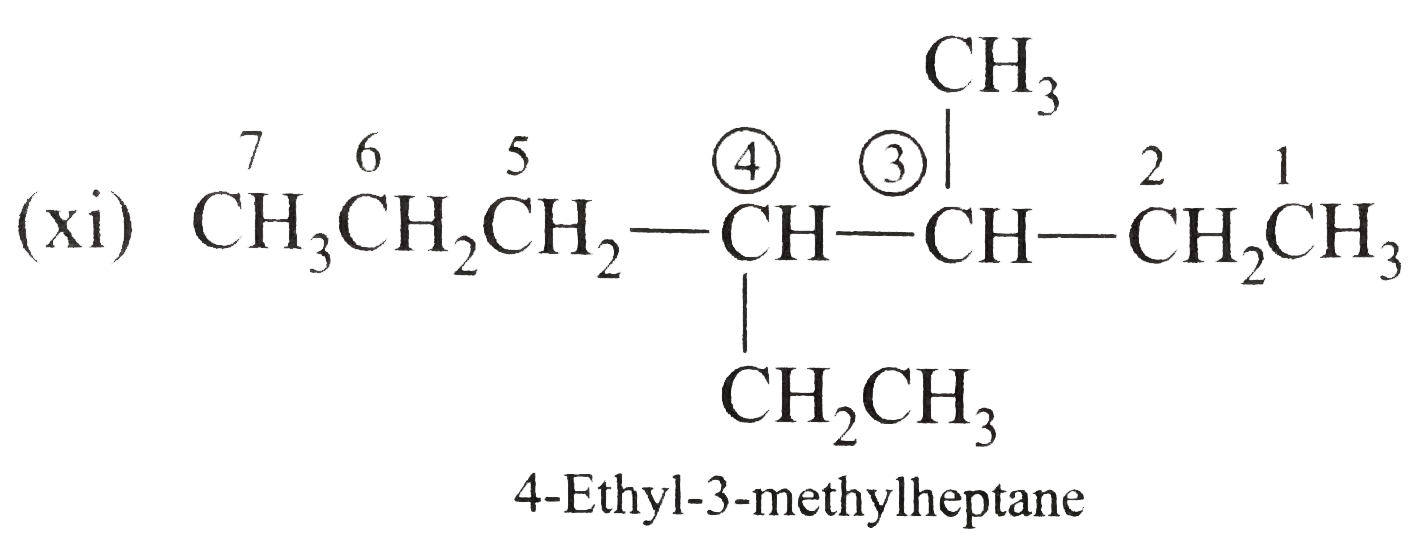 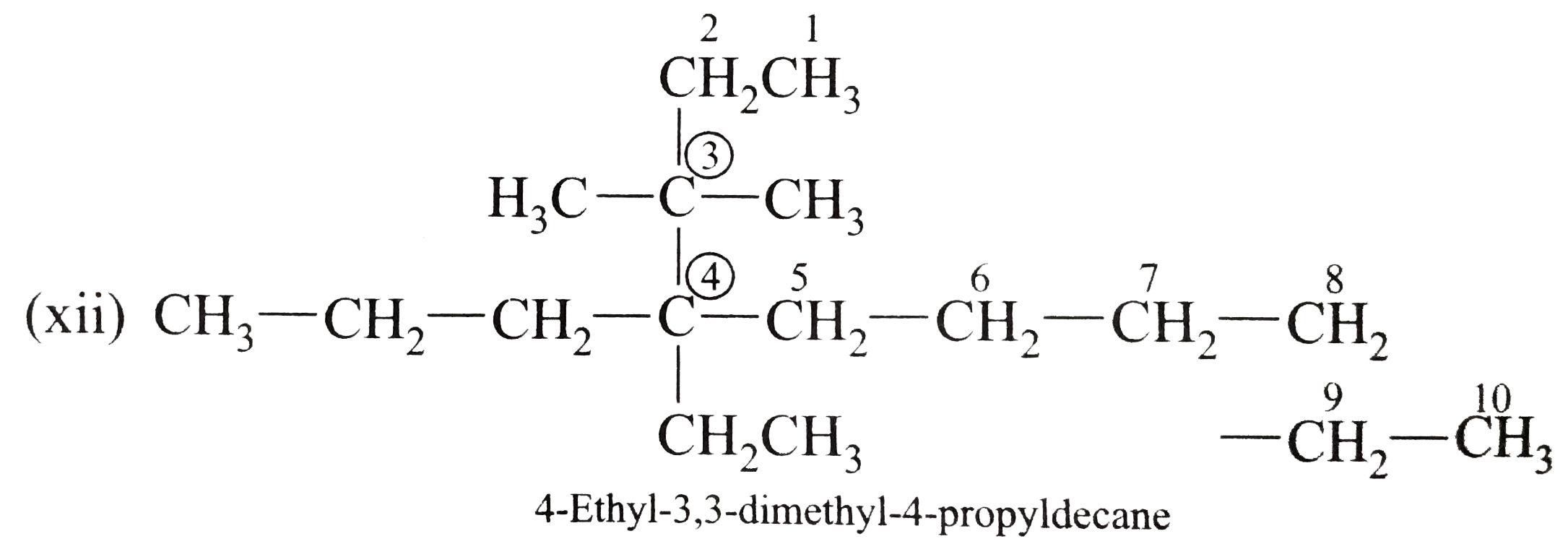 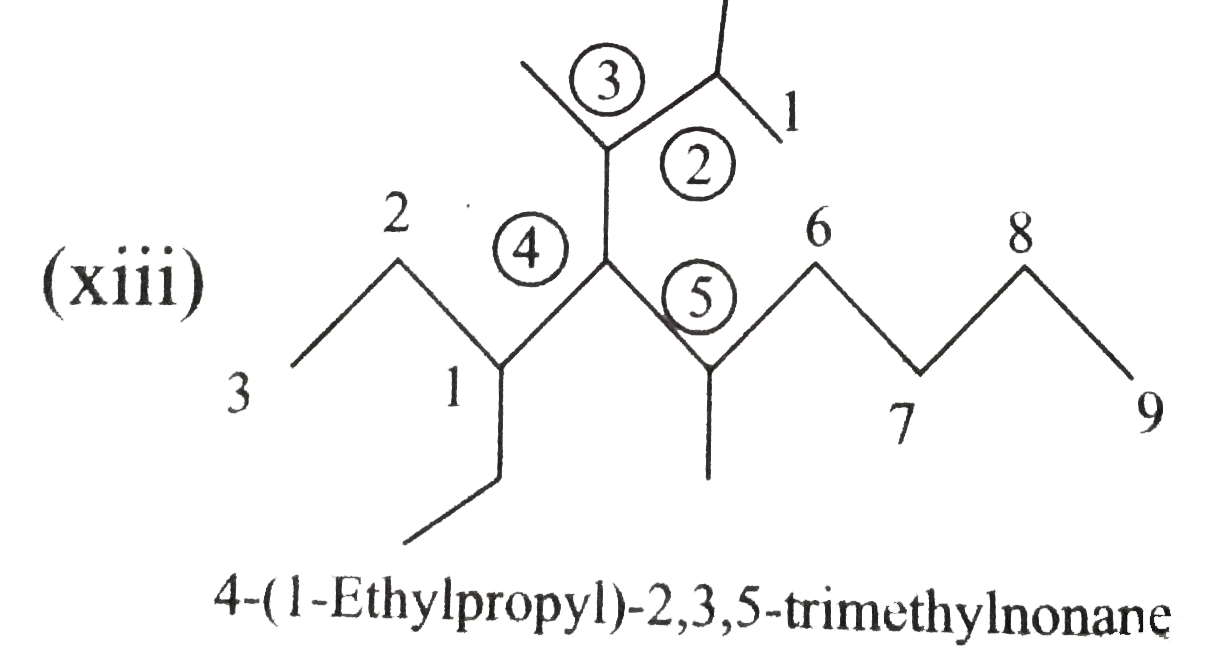 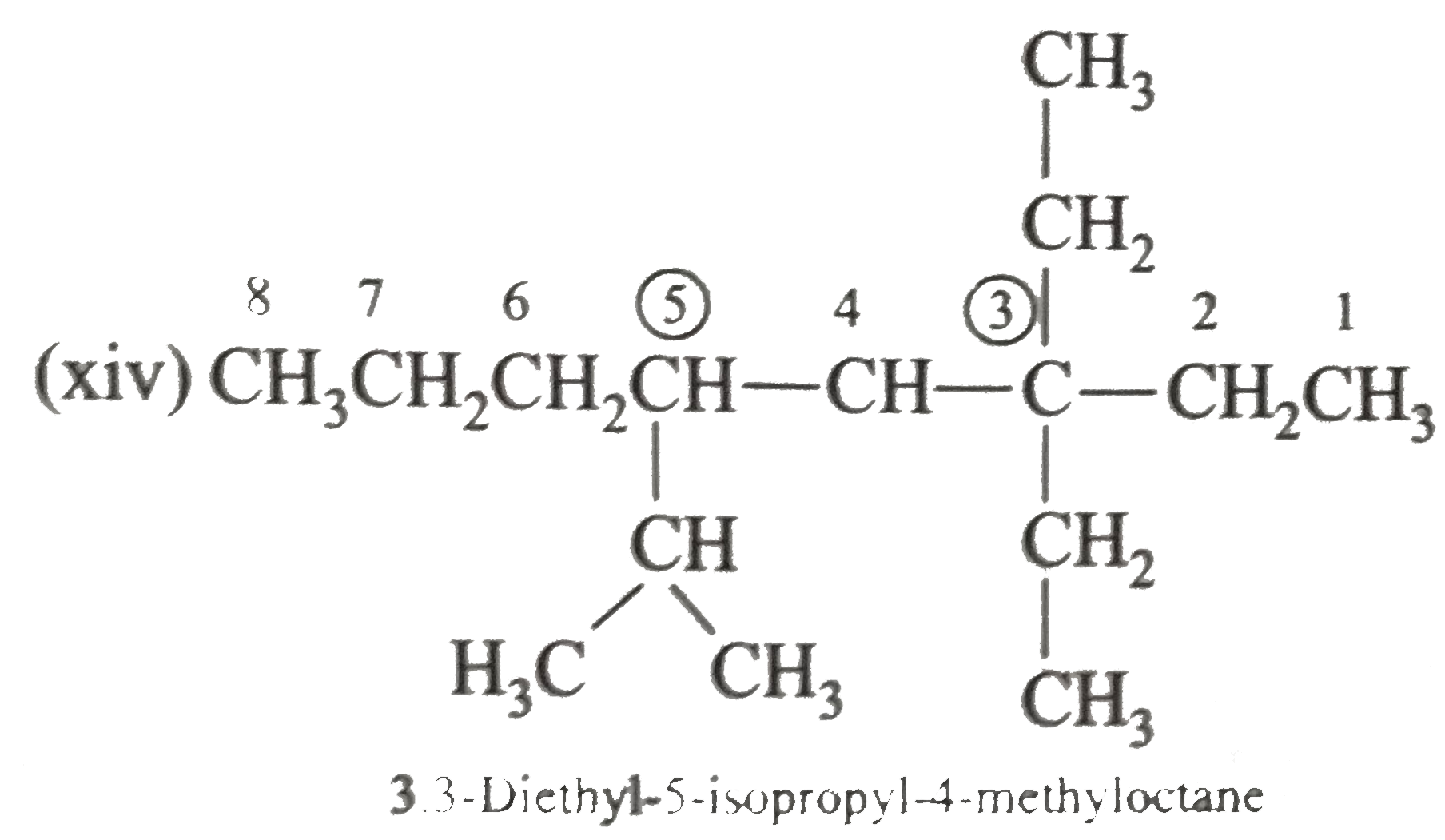 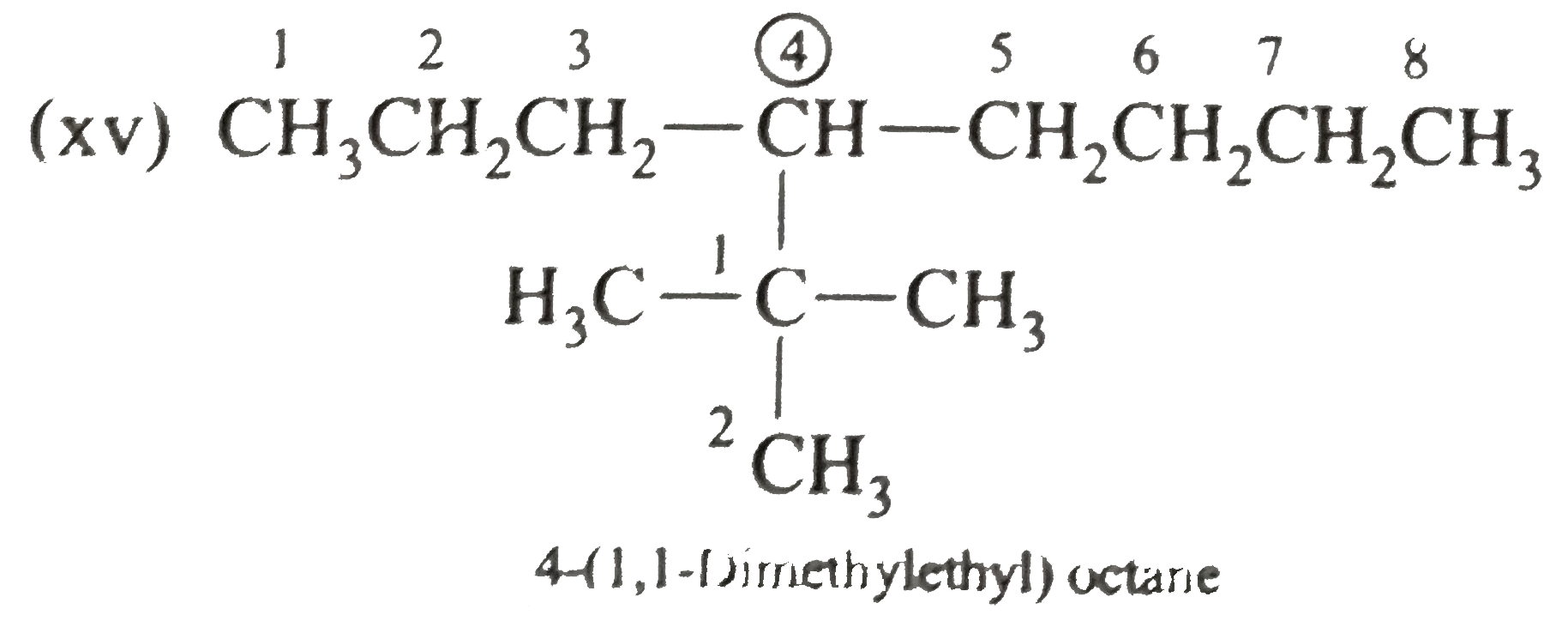 In deciding the alphabetical order for isopropyl, isobutyl, sec-butyl, and tert-butyl, we should disregard structure-defining prefixes that are written initalics and are spearated from the name by a hypen. 
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me