Saved Bookmarks
| 1. |
Write the systematic names of the following aromatic compounds: (i) (ii) (xix) para-Bromo - tert-butylbenzene Strategy: If there is only one group on the ring, no number is needed to designate its position. If there are two groups, we use the traditional designations (1,2-for ortho, 1,3-for meta, and 1,4-for para). If there are three or more goups on the ring then location numbers are assigned to give the lowest set of locants. |
|
Answer» Solution :(i) Ethylbenzene (ii) `n`-PROPHYLBENZENE (iii) (`2`-Methylpropyl) benzene (or Isobutylbenzene) (iv) `p`-Ethyltoluene (V) `1`-Bromo-`3`-nitrobenzene or `m`-Bromonitrobenzene (vi) `m`-Ethylisopropylbenzene (vii) `2`-Methyl-`3`-phenylpentane (VIII) 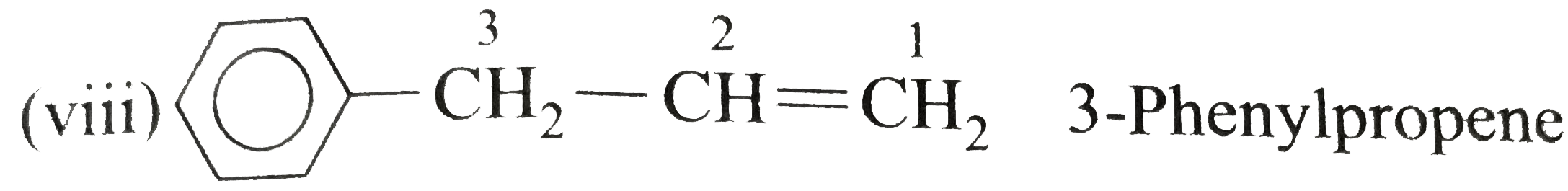 (ix) `2`-Phenylethyne (or phenylacetylene) (xi) `2,4`-Difluorobenzenesulphonic acid (xii) 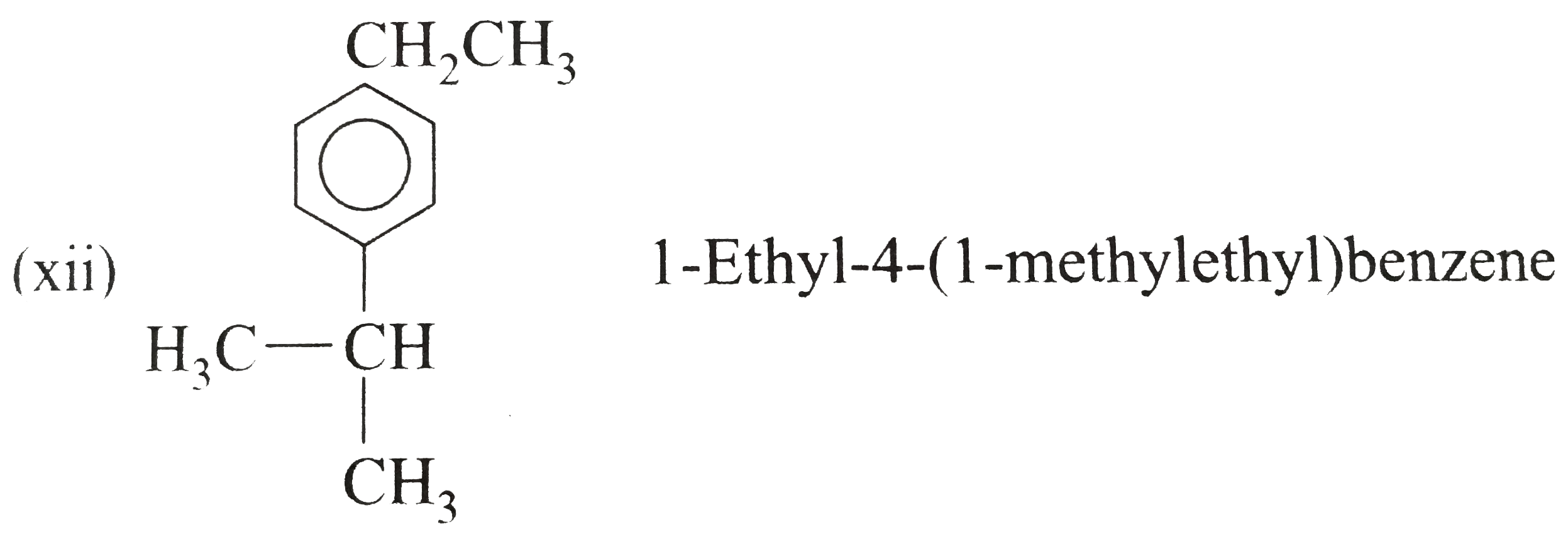 (xiii) 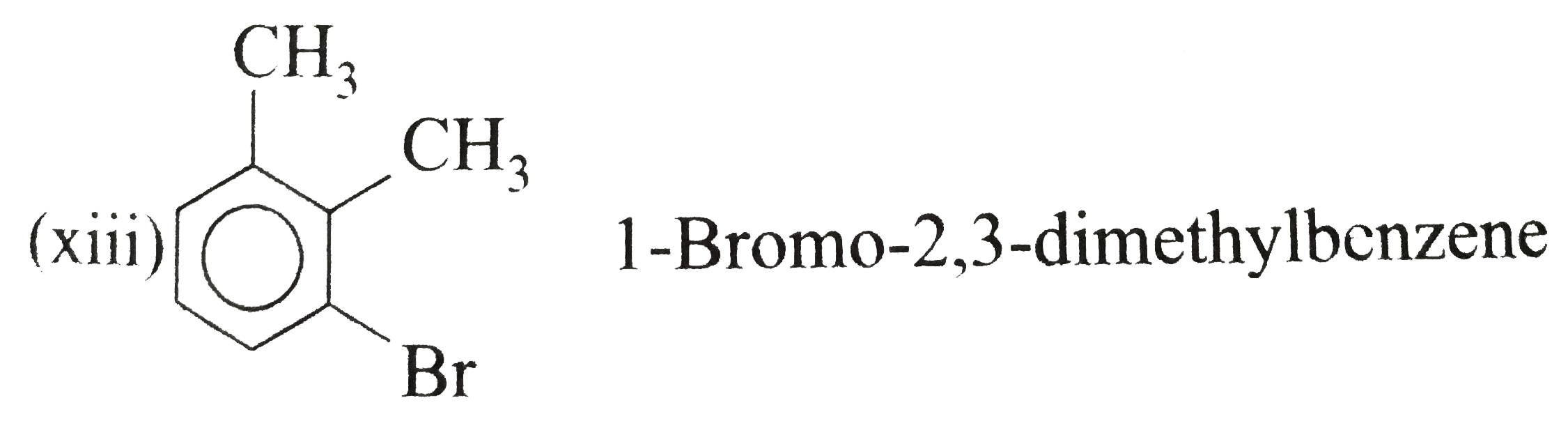 (xiv) `1,2,4`-Trinitrobenzene (xv) `1`-Ethenyl -`3`-ethyl `-5`- etynylbenzene (xvi) `1`-Iodo-`2`-metylbenzene ( or `o`-Iodotoluene) (xvii) `1`-Bromo-`3`-ethenylbenzene (or `m`-bromostyrene) (xviii) `4`-Ethyl -`1,2`-dimethylbenzene (not `1`-Ethyl-`3,4`-dimethyl benzene which has higher SET of locants) (xix) 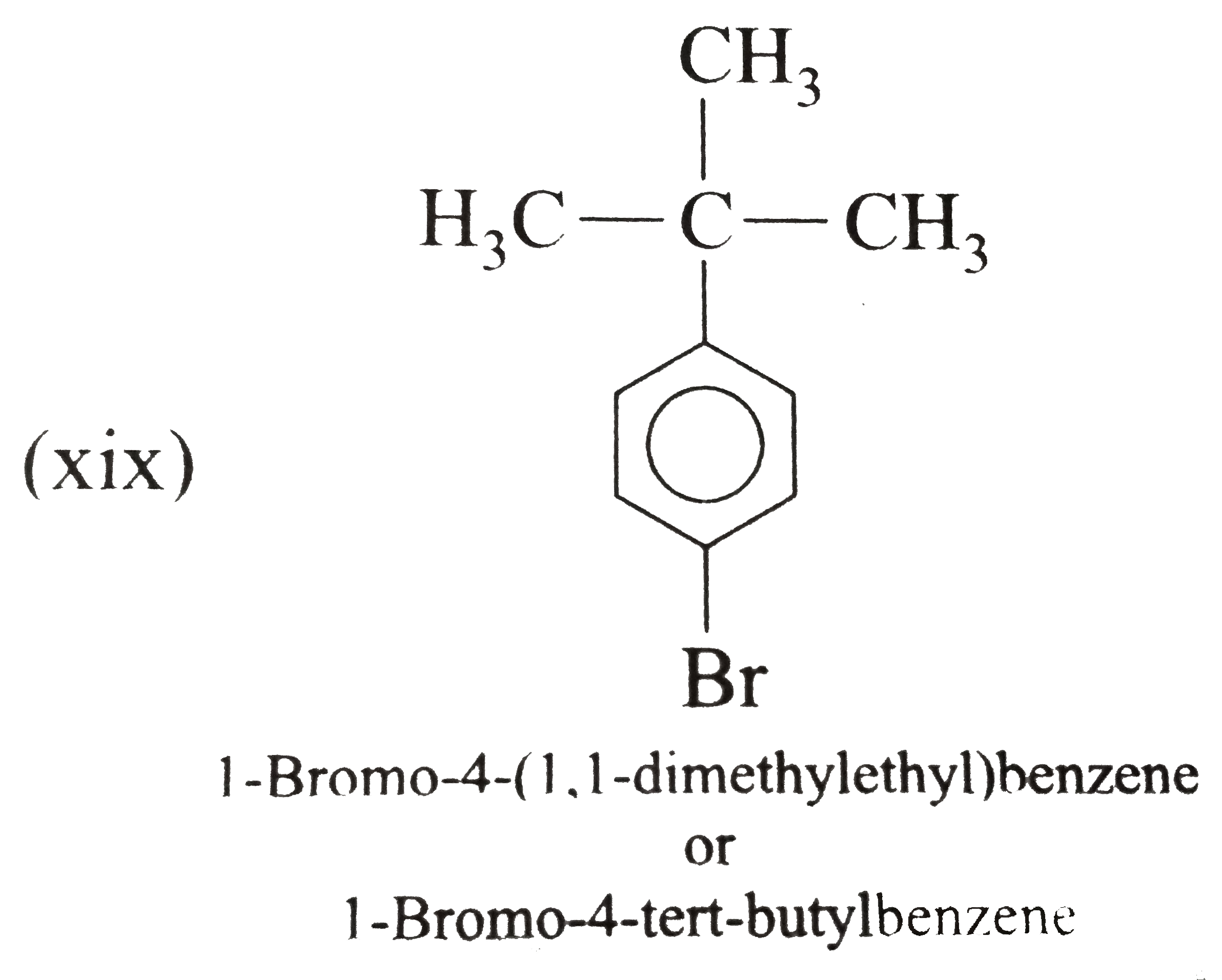
|
|
Discussion
No Comment Found
Related InterviewSolutions
- The weight of one molecule of compound C60H122 is
- Le
- Some important compounds of sodium, notes
- find the position of Zn30 in periodic table
- How to solve ion electron method
- Calculate the amount of water produced by the combustion of 16 g of methane
- Some MCQ between chapter 1and2
- Define reciprocal proportion
- What is the spectrum of hydrogen????
- I am not able to understand ch4 piz help me