Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement-1 : H_2 gas is use in metallurigical process. Statement-2 : H_2 gas is use as fuel. |
|
Answer» If both the STATEMENTS are TRUE and statement-2 is the CORRECT EXPLANATION of statement-1 |
|
| 2. |
STATEMENT-1: ##GRB_ORG_CHM_V02_QB_C03_E01_149_Q02.png" width="80%"> |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 3. |
Statement-1. Free radicals are always planar. Statement-2. They can achieve sp^(2) as well as sp^(3)-hybridisation. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for statement-1. |
|
| 4. |
statement-1 :For the reaction H_(2)(g) +I_(2)(g) hArr2HI(g) if the volume of vessel is reduced to half of its original volume, equliibrium concentration of all gases will be doubled. statement-2:According to Le chatelier's principle, reaction shifts in a direction that tends to minimize the effect of the stress. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 5. |
statement-1 :For PCI_(5)(g)hArrPCI_(3)(g)+CI_(2)(g). If more CI_(2) is added the equilibrium will shift in backward direction,hence, equilibrium constant will decrease. statement-2 : Addition of inert gas to the equilibrium mixture at constant volume,does not alter the equilibrium. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 6. |
Statement -1. For every chemicalreaction at equilibrium ,standard Gibbs energy of reactions is zero. Statement-2. At constant temperature and pressure, chemical reactions are spontaneous in the direction of decreasing Gibbs energy |
|
Answer» Statement -1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT explanation of Statement-1 |
|
| 7. |
statement-1 :For every chemical reaction at equilibrium standard Gibbs energy of reaction is zero. statement-2 : At constant temperature and pressure, chemical reactions are spontaneous in the direction of decreasing Gibbs energy. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 8. |
Statement-1. Fe^(3+) ion is more stable than Fe^(2+) ion Statement-2 Fe^(3+) ion has more number of unpaired electrons than Fe^(2+) ion |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-2 |
|
| 9. |
Statement-1: Equivalent mass of H_(3)PO_(2) is equal to its molecular mass. Statement-2: H_(3)PO_(2) is a monobasic acid. |
|
Answer» STATEMENT-1 is TRUE, statement -2 is true, statement-2 is a correct EXPLANATION for statement-1. |
|
| 10. |
Statement -1. Entropyof all elements is zero at 0K. Statement -2. Standard entropy of allelements is greater than zero. |
|
Answer» STATEMENT -1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION of Statement-1 |
|
| 11. |
Statement-1. Enthalpy change at constantpressure is always greater thanenthalpy change at constant volume for any reaction. Statement -2. Work is done by the system at constant pressure for agiven change in volume but no work is done by the system at constant volume. |
|
Answer» STATEMENT -1 is True, Statement-2 is True, Statement-2 is a correct explanation of Statement-1 |
|
| 12. |
Statement-1. Entropy is not a state function because its value depends upon the conditions of temperature and pressure. Statement -2. A state function dependsonly on the initial and the final states of the system and is independent of the path. |
|
Answer» STATEMENT -1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation of Statement-1 |
|
| 13. |
Statement-1. Electrolysis of NaH in the fused state liberates H_(2) at the anode. Statement -2. NaH contains H^(-) ions. |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is True , Statement-2 is a correct explanation for statement-1 |
|
| 14. |
STATEMENT-1: Disteremers are morror image of each other. STATEMENT-2: Disteromers may be optically active. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 15. |
Statement-1. Dimethyl sulphide is commonly used for the reduction of an ozonide of an alkene to Be carbonyl compounds. Statement-2. It reduces the ozonide giving water soluble dimethyl sulphoxide and excess of it evaporales |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 16. |
Statement -1 : Density of ice is less than water. Statement-2 : Ice has open cage structure. |
|
Answer» If both the statements are True and statement-2 is the correct EXPLANATION of statement-1 |
|
| 17. |
Statement-1. Demineralized water does not contain any ions. Statement-2. Permutit process for water softening gives demineralized water. |
|
Answer» Statement-1 is true, Statement-2 is True , Statement-2 is a correct explanation for statement-3 |
|
| 18. |
STATEMENT-1: Cyclohexen shows gemetrical siomerism. STATEMENT-2: Its two sp^(2) H-atoms are cis to each other. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. 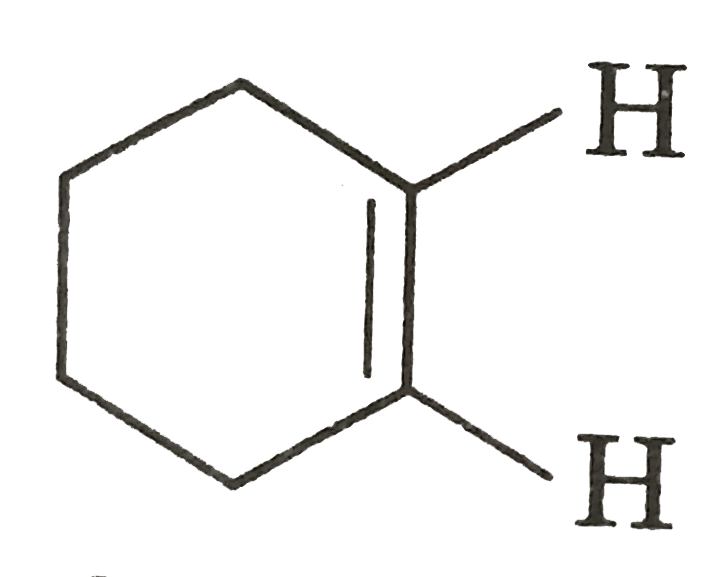
|
|
| 19. |
Statement- 1 . CuCl is more covalent than NaCl. Statement- 2 . Na^(+) ion is more polarising than Cu^(+) ion . |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for statement-1. |
|
| 20. |
Statement 1 Copper liberates hydrogen from a dilute solution of hydrochloric acid Statement 2 E^(@) of Cu is higher than of H_(2) |
|
Answer» Statement 1 is true statement 2 is true , statement 2 is a corrrect explanation for statement 1 |
|
| 21. |
Statement-1 : Covalent crystals have the highest melting point Statement-2 : Covalent bonds are stronger than ionic bonds. |
|
Answer» Statement 1 is True , Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for statement-1 |
|
| 22. |
Statement-1: correct IUPAC name of this compound is 1-chloro-6-fluoro-3,4-epoxyhexane. Statement-2: CI and F both are groups without suffix, CI comes before F in alphabetical order so numbering is done from this end. |
|
Answer» |
|
| 23. |
Statement -1. copper liberates hydrogen from a dilute solution of hydrochloric acid Statement-2 E^(@) of Cu is higher than that of H_(2) |
|
Answer» STATEMENT -1 is true, statement -2 is ALSO true, statement -2 is the correct explanation of statement-1 |
|
| 24. |
statement-1 :Consider a reaction A(g)hArrB(g) the equilibrium moles of A and B are respectively alphaand beta in 1 litre container.If 5 moles of 'A'and 3 moles of 'B' are added then reaction must move in forward direction. statement-2: If amount of reactant added to a system at equilibrium is more than amount of product added at same time than reation can move in any direction. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 25. |
Statement-1. Compressibility factor of non-ideal gases is always less than1. Statement-2. Non-ideal gases exert less pressure than expected for ideal gas. |
|
Answer» Statement-1 is correct, Statement-2 is correct , Statement-2 is the correct explanation for Statement-1. |
|
| 26. |
Statement-1. CO_(2) is a gas but SiO_(2) is a solidat room temperature. Statement-2. CO_(2) contains C = O bonds but C=O bonds butSiO_(2) has a three-directional network structure. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 27. |
Statement-1. CO and NO both combine with haemoglobin. Statement-2. Both have equal affinity for haemoglobin. |
|
Answer» Statement-1 is TRUE, Statement-2 is True : Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 28. |
Statement-1 : CH_4 is a covalent hydrides. Statement-2 : CH_4 is dectron precise type hydrides. |
|
Answer» If both the STATEMENTS are TRUE and statement-2 is the correct explanation of statement-1 |
|
| 29. |
Statement-1 . CH_(3)CHO and CH_(2)=CHOH are resonance structures. Statement-2. Tautomers differ both in the position of atoms as well as electrons. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT explanation for statement-1. |
|
| 30. |
Statement-1: Calcium carbide on hydrolysis gives methane. Statement-2: Calcium carbide contains C_(2)^(2-) anion. |
|
Answer» Statement-1 is TRUE, Statement-2 is True. Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 31. |
Statement-1. Bromobenzene upon reaction with Br_2 /Fe gives 1. 4-dibromobenzene as the major product Statement-2. In bromobenzene, the inductive effect of the bromo group is more dominant than the mesomeric effect in directing the incoming electrophile. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a correct explanation for Statement-1 |
|
| 32. |
Statement-1. Boron alwaysforms covalent bonds. Statement-1. The small size of B^(3+) favours formation of covalent bond. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 33. |
STATEMENT -1:Boiling point of ketones is higher thencorresponding Aldehyde.STATEMENT -2 :Dipolemoment of Aldehyde is higher then Ketone. |
|
Answer» |
|
| 34. |
Statement-1. BF_(3) molecule is planar whileNF_(3) is pyramidal. Statement-2. N atom is smaller than B . |
|
Answer» Statement-1 is TURE , Statement-2 is Ture , Statement-2 is a correct explanation for Statement-1. atom is `sp^(2)` while iin `NF_(3)` , hybridisation o N atom is `sp^(3)` (of course , N atom is smaller than B because along a period the size decreasses.) |
|
| 35. |
Statement-1: Boiling point of methane is lower than that of butane. Statement - II: The boiling point of continuous chain alkanes increases with increase in length of carbon chain. |
|
Answer» STATEMENT -I and II are correct and statement-IL is correct EXPLANATION of statement-1. |
|
| 36. |
Statement-1. BF_(3) is a weaker Lewis acid than BCl_(3). Statement-2. The ppi-ppi back bonding is stronger in BF_(3) than in BCl_(3). |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 37. |
Statement- 1 . Be(OH)_(2) is soluble both in HCl and NaOH . Statement- 2 . Be(OH)_(2) is amphoteric in nature . |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 38. |
Statement-1. Beilstein test can be used to detect fluorine in the organic compound. Statement-2. CuF is not volatile and hence does not impart any colour to the flame. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct explanation for statement-1. |
|
| 39. |
Statement- 1. Be and Mg give characteristic flame colourations . Statement- 2 . As compared to other alkaline earth metals , ionization enthalpy of Be and Mg is high . |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct explanation for statement-1. |
|
| 40. |
Statement- 1 . Be forms [BeF_(4)]^(2-) but Al forms [AlF_(6)]^(3-). Statement- 2 . Be does not have d-orbitals in the valence shell but Al has . |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 41. |
Statement-1. At zero degree Kelvin, the volume occupied by a gas is negligible. Statement-2. All molecullar motion ceases at 0 K. |
|
Answer» Statement-1 is CORRECT, Statement-2 is correct , Statement-2 is the correct explanation for Statement-1. |
|
| 42. |
Statement- 1. Among the alkali metals , caesium salts exhibit the maximum electrical conductance in aqueous solutions . Statement- 2 . The radius of hydrated caesium ion is the highest among alkali metals . |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for statement-1. |
|
| 43. |
statement-1 :Ammonia at a pressure of 10 atm and CO_(2) at a pressure of 20atm are introduced into an evacuated chamber. If K_(P) for the reaction. NH_(2)COONH_(4)hArr2NH_(3)(g)+CO(g) is 2020atm^(3) the total pressure after a long time is less than 30atm. statement-2 : Equilibrium can be attained from both directions. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 44. |
STATEMENT -1: Amixture of on treatment with dil . NaOH gives STATEMENT -2 :The ketone is very hindered and conjugated and so less reaction than aldehyde. |
|
Answer» STATEMENT -1: Amixture of |
|
| 45. |
Statement-1. Aluminium acts as a strong affinity for oxygen. Statement-2. Aluminium has a strong affinity for oxygen. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 46. |
Statement-1. Although NaH_(2)PO_(2) contains two H-atoms, it is not an acid salt. Statement-2. It contains two ionisable hydrogens. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 47. |
STATEMENT -1:Alphatic Ketone are Less reactive then aliphatic aldelydeSTATEMENT -2 :Rate of Electrophillic addition and substitution in aliphatic aldehyde is faster then aliphatic ketones |
Answer»  more`e^(-)` DEFFICIENCY is CREATED |
|
| 48. |
Statement 1 all decompostion reaction are redox reactions Statement 2 H_(2)O on decompostion gives H_(2) and O_(2) |
|
Answer» Statement 1 is true statement 2 is true , statement 2 is a corrrect explanation for statement 1 |
|
| 49. |
Statement- 1 . Alkali metals dissolved in liquid ammonia to give blue solutions . Statement- 2 . Alkali metals in liquid ammonia give solvated species of the type [M(NH_(3))_(n)]^(+) (M = alkali metals) |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION for statement-1. |
|
| 50. |
Statement-1: Addition of HCl (aq). " to " CH_(3)COOH (aq). decrease the ionisation of CH_(3)COOH (aq). Statement-2 : Due to common ion effect H^(+), ionisation of CH_(3)COOH decreases. |
|
Answer» If both the STATEMENTS are TRUE and statement -2 is the correct EXPLANATION of statement-I |
|
 on
on