Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The carbon atoms in the compound (CN)_(4)C_(2) are |
|
Answer» SP HYBRIDIZED |
|
| 2. |
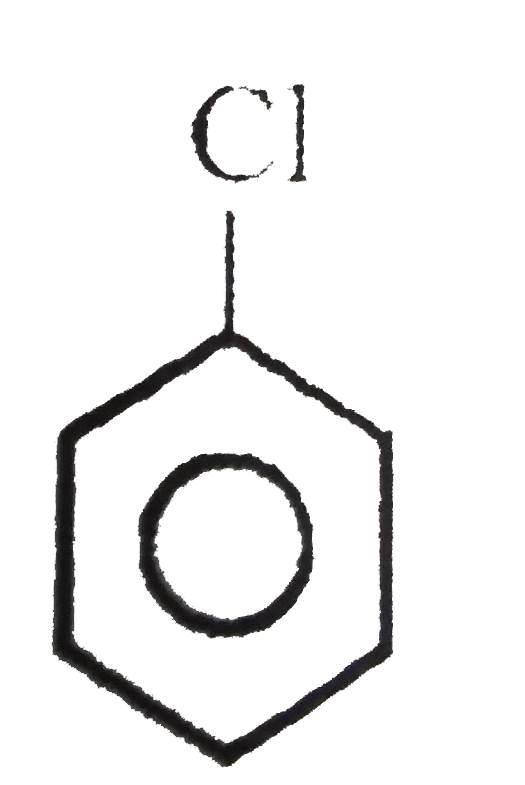
The carbon atom carrying halogen atom is more electron deficient in |
|
Answer»
|
|
| 3. |
The carbocation fomed in S_(N^1) reaction of alkyl halide in the slow step is …………………… . |
|
Answer» `SP^3`hybridised |
|
| 4. |
The carbocation CH_(3)overset(+)(C)HCH_(3) is less stable than |
|
Answer» `CH_(3)CH_(2)overset(+)(C)H_(2)` |
|
| 5. |
The carbo cation formed in SN_(1) reaction of alkyl halide in the slow step is |
|
Answer» `SP^(3)` hybridised |
|
| 6. |
The carbide which gives propyne on hydrolysis |
|
Answer» `Al_(4)C_(3)` |
|
| 7. |
The carbide of which of the following metals on hydrolysis give allylene |
|
Answer» `Be` |
|
| 8. |
The carbanion expels a leaving group LG to yield an alkene as shown above by |
|
Answer» E1CB mechanism |
|
| 9. |
The capacity to bring about coagulation increases with |
|
Answer» IONIC radii |
|
| 10. |
The canonical or resonating structures of a molecule required to describe the structure of a molecule follow which of the following rules ? |
|
Answer» The RELATIVE position of all ATOMS can differ. |
|
| 11. |
The calcium salt of which of the following acid on dry distillation gives 2, 4-dimethylpentan-3-one? |
|
Answer» Propionic acid |
|
| 12. |
The calcium contained in a solution of 1.048g of a substance being analysed was precipitated with 25mL H_(2)C_(2)O_(4). The excess of C_(2)O_(4)^(2-) in one fourth of filrtate was back titrated with 5mLof 0.1025N KMnO_(4). To determine the conc. of H_(2)C_(2)O_(4) soloution, it was diluted foru folds and titrationof 25mLof sameKmnO_(4) solution. Calculate % of Ca in substance. |
|
Answer» |
|
| 13. |
The calcium concentration in plasma is regulated by which harmones |
|
Answer» INSULINE, Glucagon |
|
| 14. |
The Ca^(2+) and F^(-)ions are located in CaF_(2) crystal, respectively at face centred cubic lattice points and in |
|
Answer» TETRAHEDRAL void |
|
| 15. |
The C-O bond order in CO and CO_(2) respectively |
|
Answer» 2,3 |
|
| 16. |
The (C-H) bond length order in hydrocarbons in which the hybridisation of carbon is sp^(3) or sp^(2) or sp |
|
Answer» `SP - s gt sp^(2) - s gt sp^(3) -s` |
|
| 17. |
The C - H bond distance is the longest in |
|
Answer» `C_(2)H_(2)` |
|
| 18. |
The C-H bond distance is largest in |
|
Answer» `C_2H_2` |
|
| 19. |
The C-H bond and C- C bond in ethane areformedby which of the followingtypes of overlap |
|
Answer» `SP^(3)-s` and `sp^(3)-sp^(3)` |
|
| 20. |
The C-Hbond and C-Cbond in ethane are formed by which of the following types of overlap |
|
Answer» `sp^(3)-s` and `sp^(2)-sp^(3)` |
|
| 21. |
The C-C bond lengt in diamond is "………….."while in graphite it is "…………..". |
|
Answer» |
|
| 22. |
The C-C bond length in benzene is equal due to…… |
|
Answer» isomerism |
|
| 23. |
The C-C bond in benzene can be determined by |
|
Answer» X-ray diffraction It is the same between any TWO carbon atoms. There is equal probability of ACH carbon ATOM to get overlapped with p-orbital of another carbon atom |
|
| 24. |
The C-C bond dissociation energy is |
|
Answer» 103 K cals `"mole"^(-1)` |
|
| 25. |
The C-C bond distance is largest in |
|
Answer» `C_2 H_2`<BR>` C_2H_4` |
|
| 26. |
The C-C bond lengths in benzeneare |
|
Answer» `1.54A^@ & 1.34A^@ ` |
|
| 27. |
The C-C and C-H bond length in ethane are …………and ……….respectively |
|
Answer» 145pm and 112pm |
|
| 28. |
The by-product of solvay's ammonia process is..... |
|
Answer» CARBON dioxide |
|
| 29. |
The by product of Solvay process is : |
|
Answer» `NH_(4)CI` |
|
| 30. |
The by-product of Solvay ammonia process is |
|
Answer» CARBON dioxide |
|
| 31. |
The butyl alcohol which gives iodoform test is |
|
Answer» n-Butyl alcohol |
|
| 32. |
The buring of hydrogen is called as |
|
Answer» Hydrogenation |
|
| 33. |
The bubble of a gas released at the bottom of alake increases to eight times its original volume when it reaches the surface. Assuming that atmospheric pressure is equivalent to the pressure exerted by a column of water 10 m height, the depth of the lake is |
|
Answer» 80 m |
|
| 34. |
The brown ring test for the NO_(3)^(-) ions is due to the formation of : |
|
Answer» `FeSO_(4).NO_(2)` |
|
| 35. |
The brown ring test for nitrates depends upon |
|
Answer» the reduction of NITRATE to nitric oxide |
|
| 36. |
The bromineatom possesses35electrons.itcontains6 electrons in 2porbitals6 elecrons in 3porbitaland 5electronin 4porbitalswhich of theseelectronexperience the lowesteffectivenuclear charge |
|
Answer» SOLUTION :4p orbitalhas LESS effectivenuclearcharge Asn=4is morethann=2of 2pandn=3of 3p . |
|
| 37. |
The bromoderivative of C_(3)H_(7)CH_(2)COOH is formed on treating it with the bromine in presence of red phosphorus. The process is known as |
|
Answer» Dehydrogenation |
|
| 38. |
The bromine atom possesses 35 electrons. It contains 6 electrons in 2p orbital, 6 electrons in 3p orbital and 5 electrons in 4p orbital. Which of these electrons experience the lowest effective nuclear charge ? |
| Answer» SOLUTION :OBVIOUSLY the `5.e^(-)` in 4p-orbital EXPERIENCE the LOWEST effective NUCLEAR charge. | |
| 39. |
The bromine atom possesses 35 electrons. It contains 6 electron in 2p orbitals, 6 electrons in 3p orbitals and 5 electrons in 4p orbitals. Which of these electron experiences the lowest effective nuclear charge? |
|
Answer» SOLUTION :4p electrons, being farthest from the nucleus, experience the lowest effective NUCLEAR charge. For calculation of exact effective nuclear charge, These may be applied to bromine ATOM as follows: `._(35)Br = (1s^(2)) (2s^(2) 2p^(6)) (3s^(2) 3p^(6)) (3d^(10)) (4S^(2) 4p^(5))` For `2p^(6), " " S = (0.35 xx 7) + (0.85 xx 2) = 2.45 + 1.70 = 4.15` `Z_("eff")` for 2p electron `= 35 - 4.15 = 30.85` For `3p^(6), S = (0.35 xx 7) + (0.85 xx 8) + (1.0 xx 2) = 2.45 + 6.80 + 2.0 = 11.25` `Z_("eff")` for 3p electron `= 35 - 11.25 = 23.75` For `4p^(5), " " S = (0.35 xx 6) + (0.85 xx 18) + (1.0 xx 10) = 2.10 + 15.30 + 10.0 = 27.40` `Z_("eff")` for 4p electron `= 35 - 27.40 = 7.60` Thus, 4p has the lowest effective nuclear charge |
|
| 40. |
The bromide impurity in a 2.00 g sample of a metal nitrate is precipitated as silver bromide. If 6.40 mL of 0.200"M AgNO"_(3) solution is required, what is the mass percentage of bromide in the sample? |
|
Answer» `1.28` |
|
| 41. |
The branching of alkanes that produces summetrical structures |
|
Answer» RAISES the BOILING POINT, raises the MELTING point |
|
| 42. |
The branching in an alkane increases its tendency of knocking. Comment over the statement and name the commonly used antiknocking agent. |
|
Answer» |
|
| 43. |
The branch of science associated with determining the changes in energy of a system by measuring the heat exchanges with surrounding is called …………. |
|
Answer» MECHANICS |
|
| 44. |
The Boyle's law can be expressed graphically as |
|
Answer» <P> |
|
| 45. |
The bonds present in [Cu(NH_3)_4] SO_4 between copper and ammonia are |
|
Answer» ionic |
|
| 46. |
The bonds present in HCI moleculc are |
|
Answer» Non-polar COVALENT |
|
| 47. |
The bonds present in [Cu(NH_(3))_(4)]SO_(4) between copper and ammonia are |
|
Answer» IONIC |
|
| 48. |
The bonds not present in diborane is |
|
Answer» B-H  In `B_(2)H_(6), B-B` BONDS are not PRESENT. |
|
| 49. |
The bonds not present in diborane are |
|
Answer» B-H |
|
| 50. |
The bond strength of C-X for the C-CI,C-Br,C-I,C-F decreases in the order is ……………….. . |
|
Answer» SOLUTION :C-F < C-Cl < C-Br |
|