Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
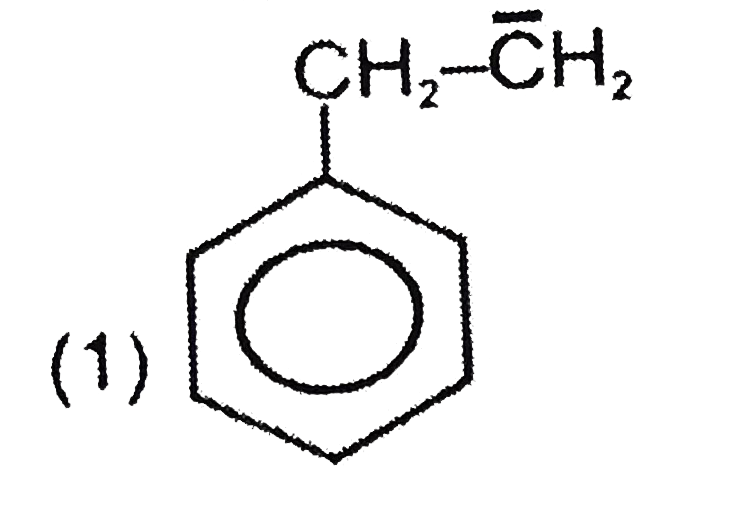
| 3. |
The most unstable carbocation is: |
|
Answer» `Ch_(3)underset(o+)CH_(2)` |
|
| 4. |
The most unstable carbocation, amongst the following, is |
|
Answer» `(CH_3)_(3)C^(+)` |
|
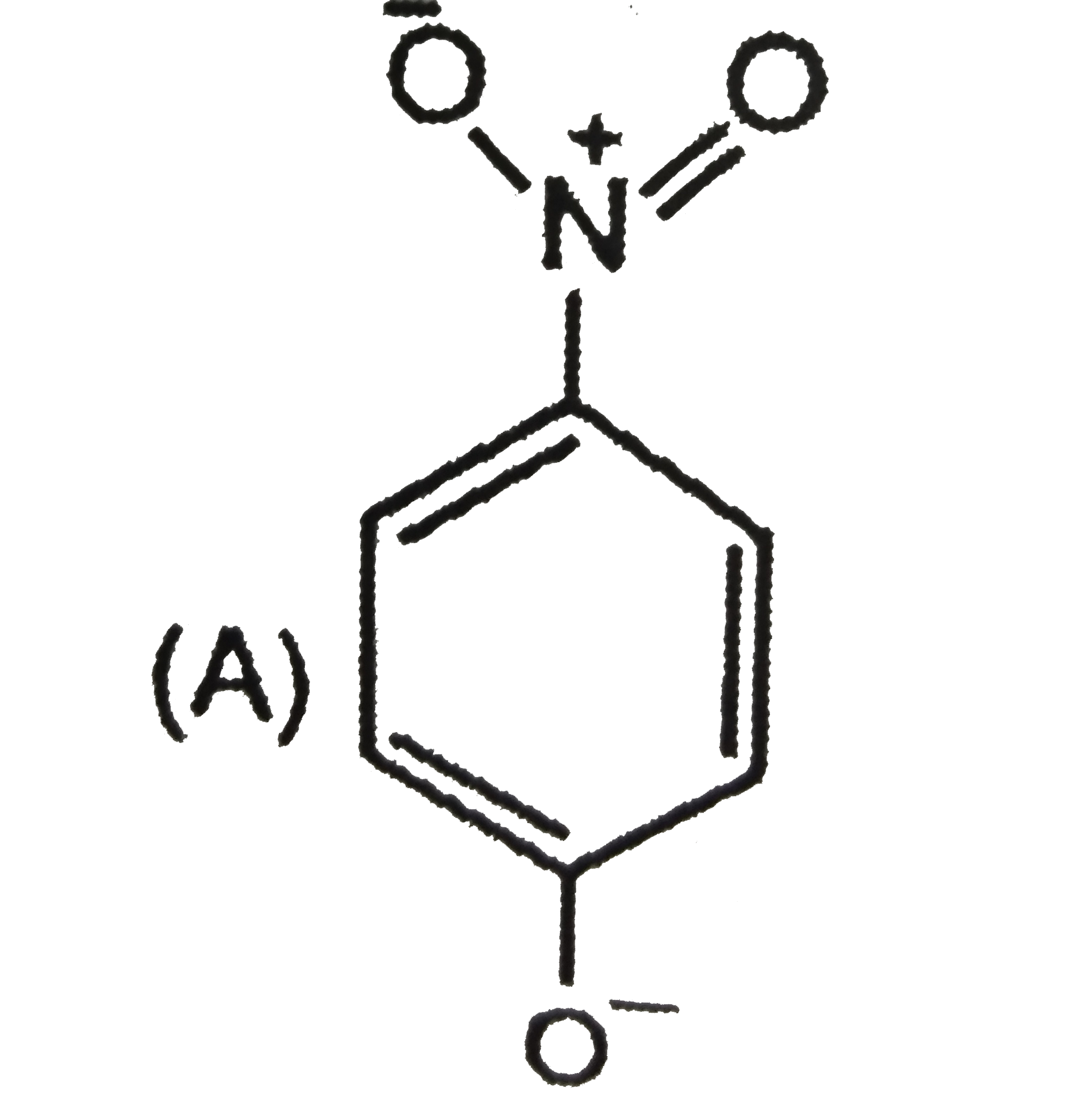
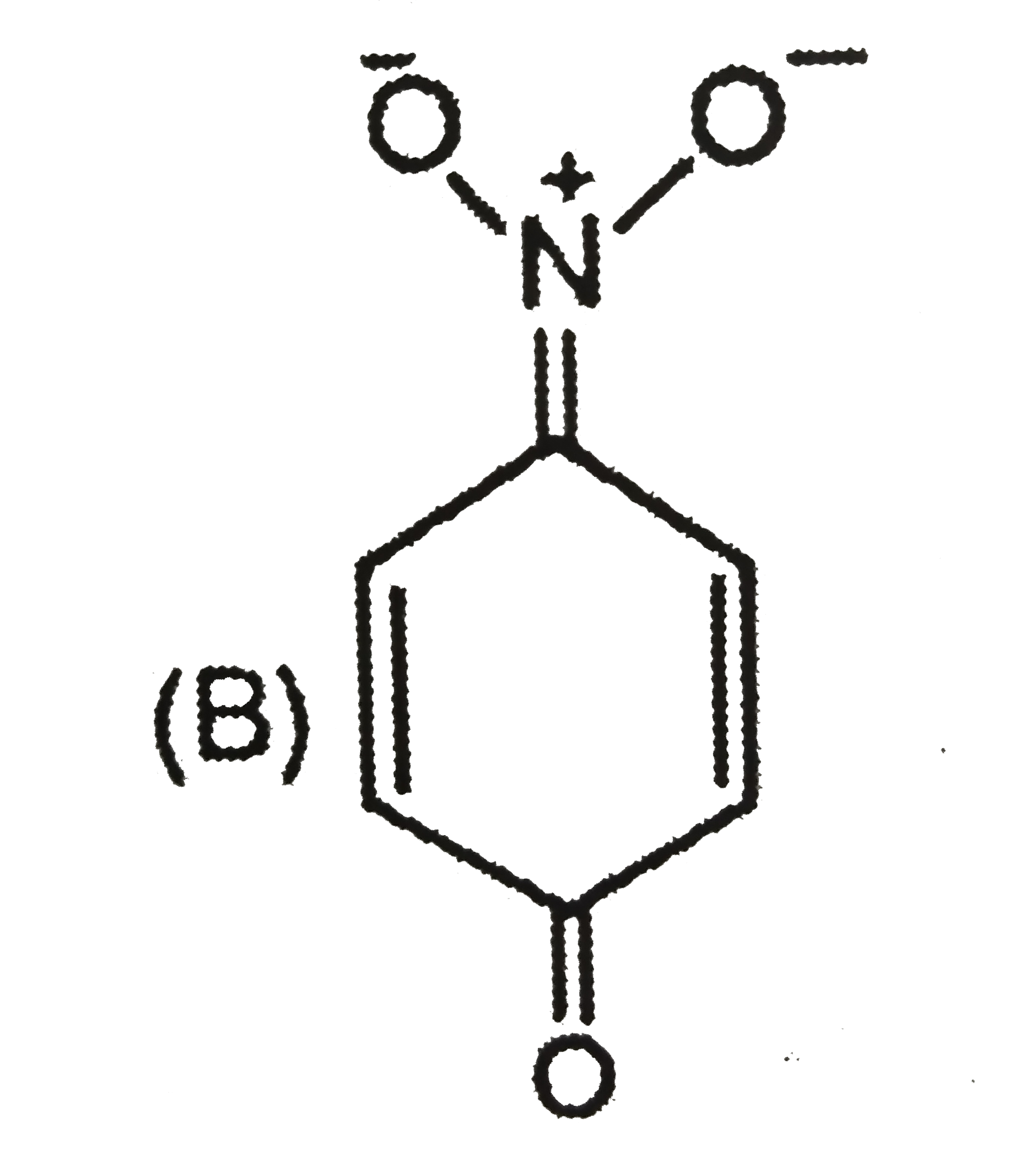
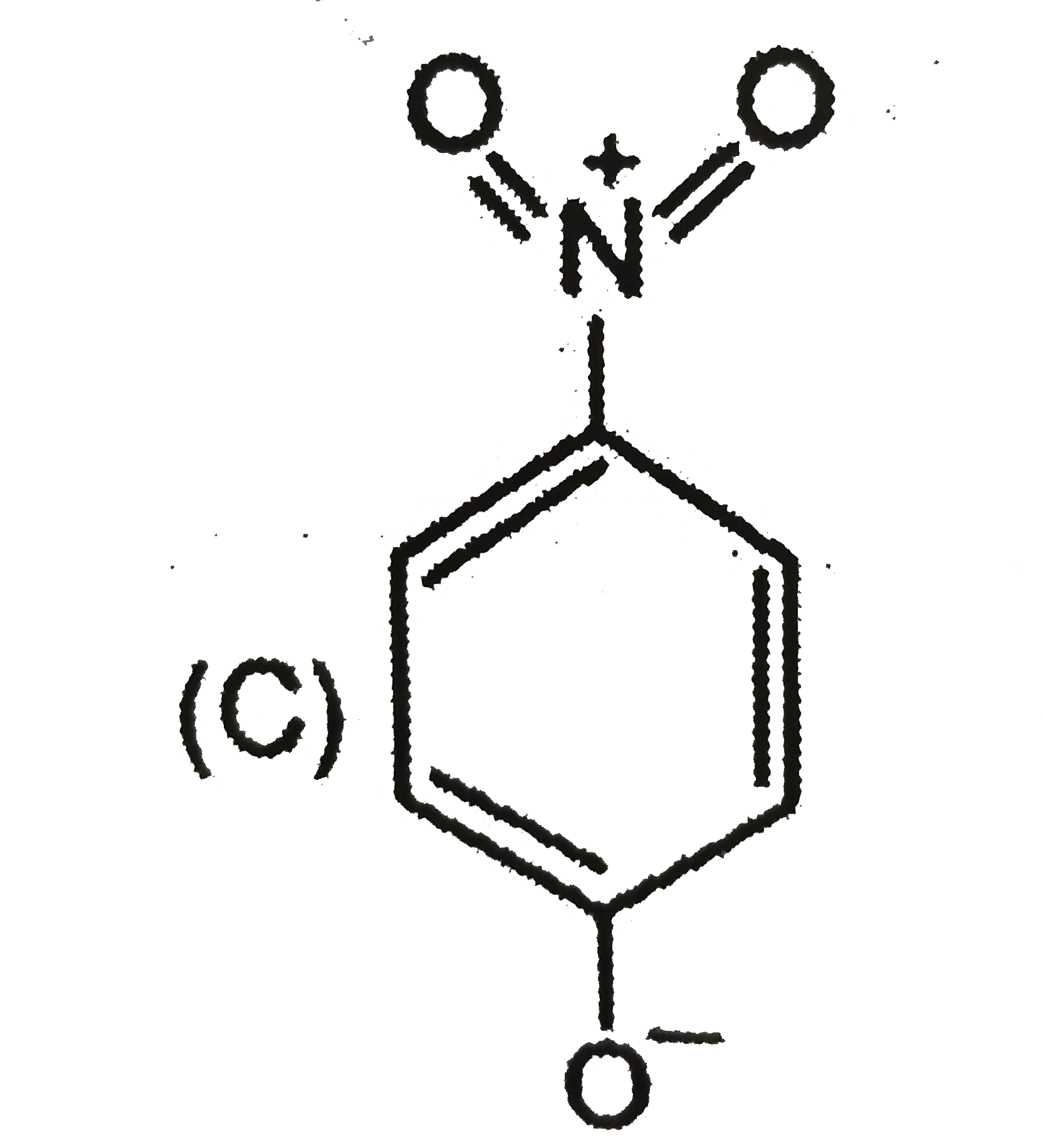
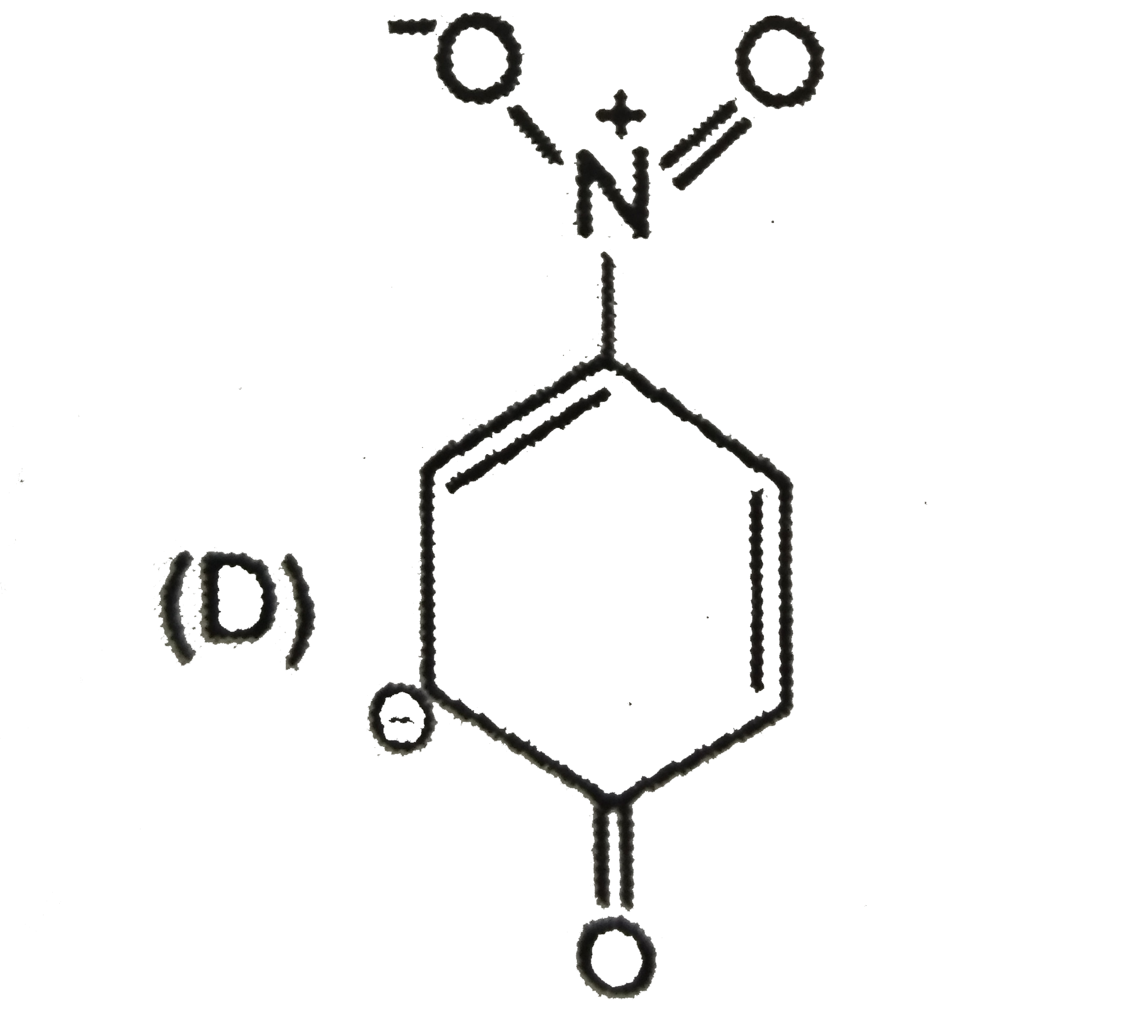
| 5. |
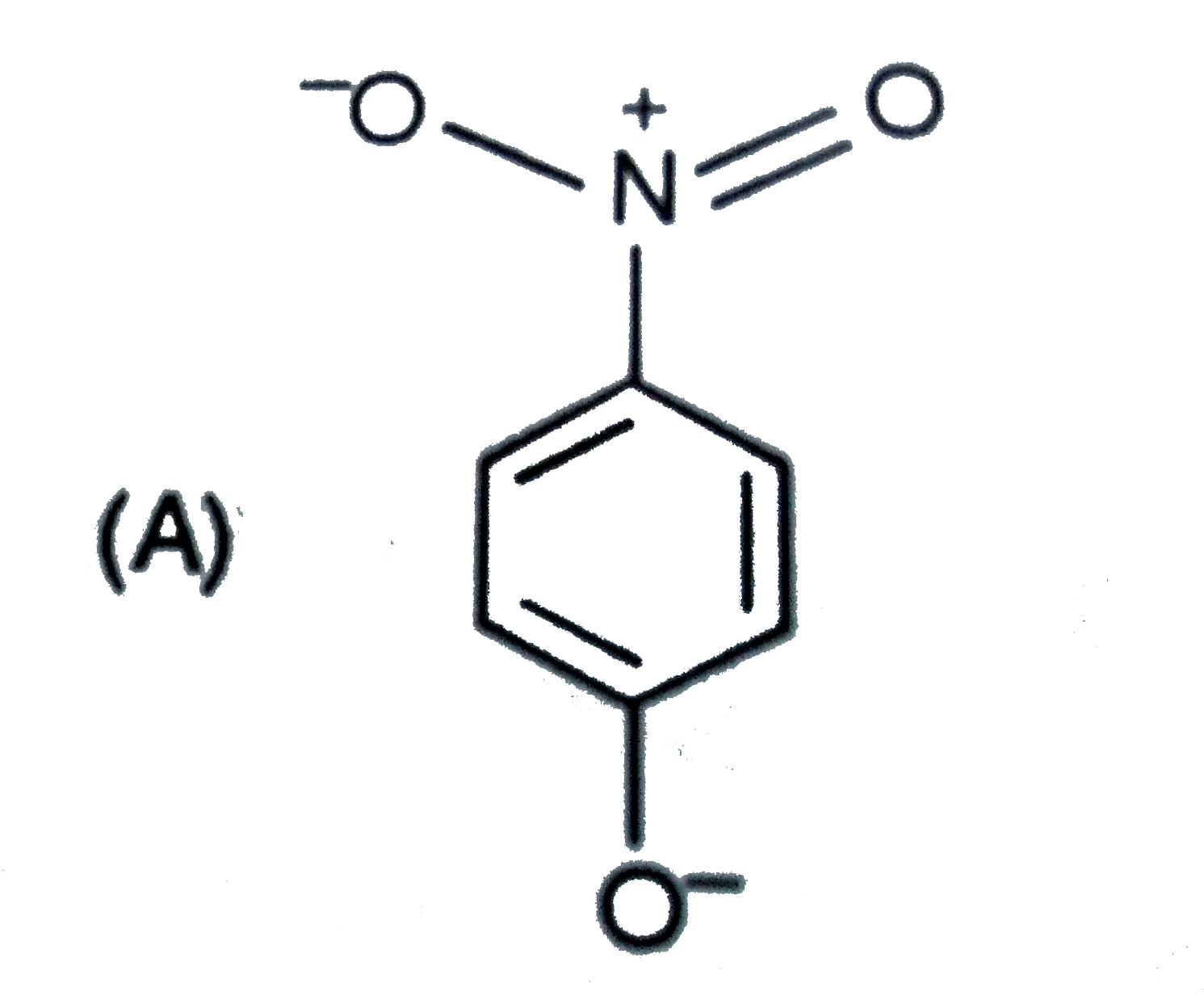
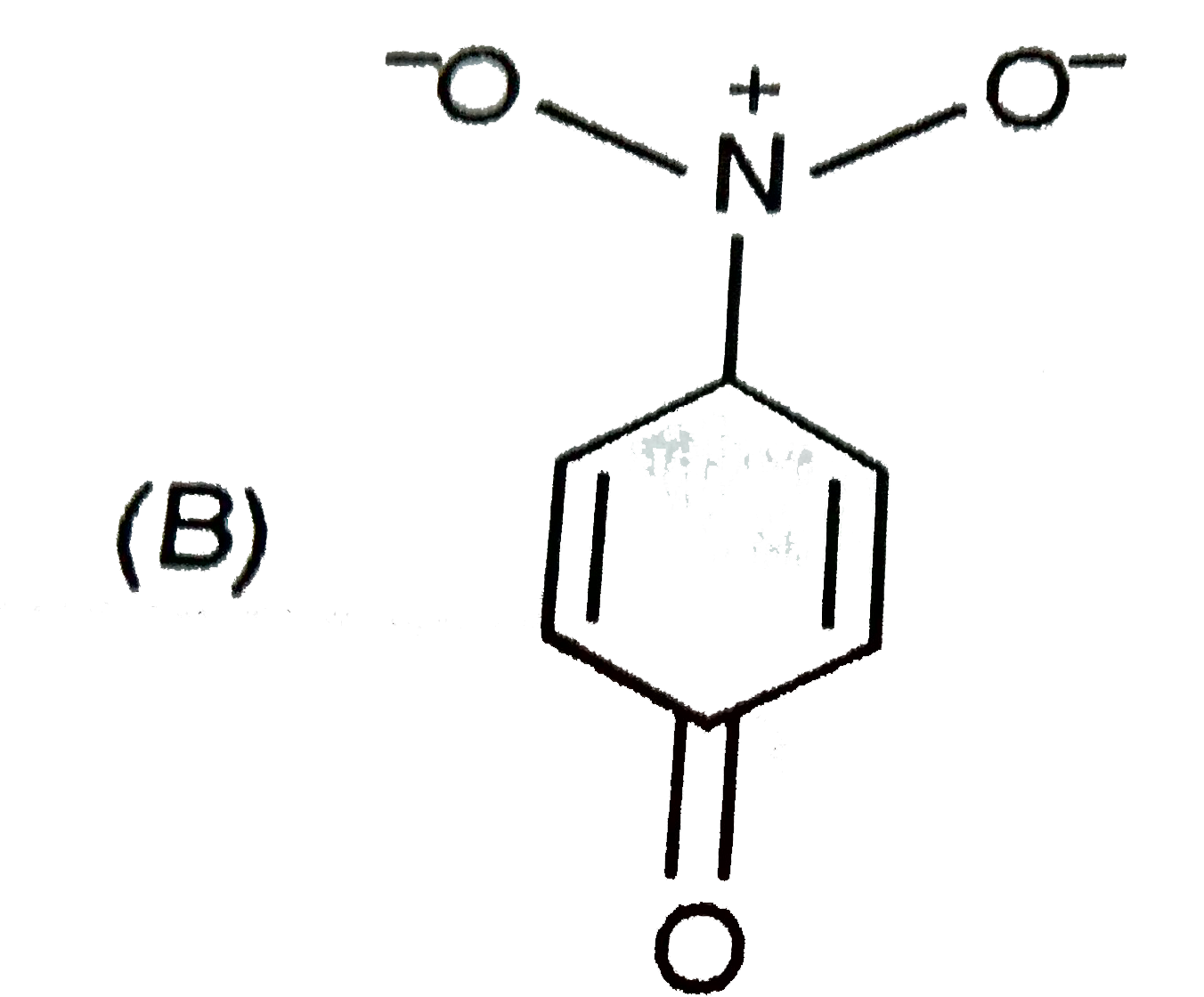
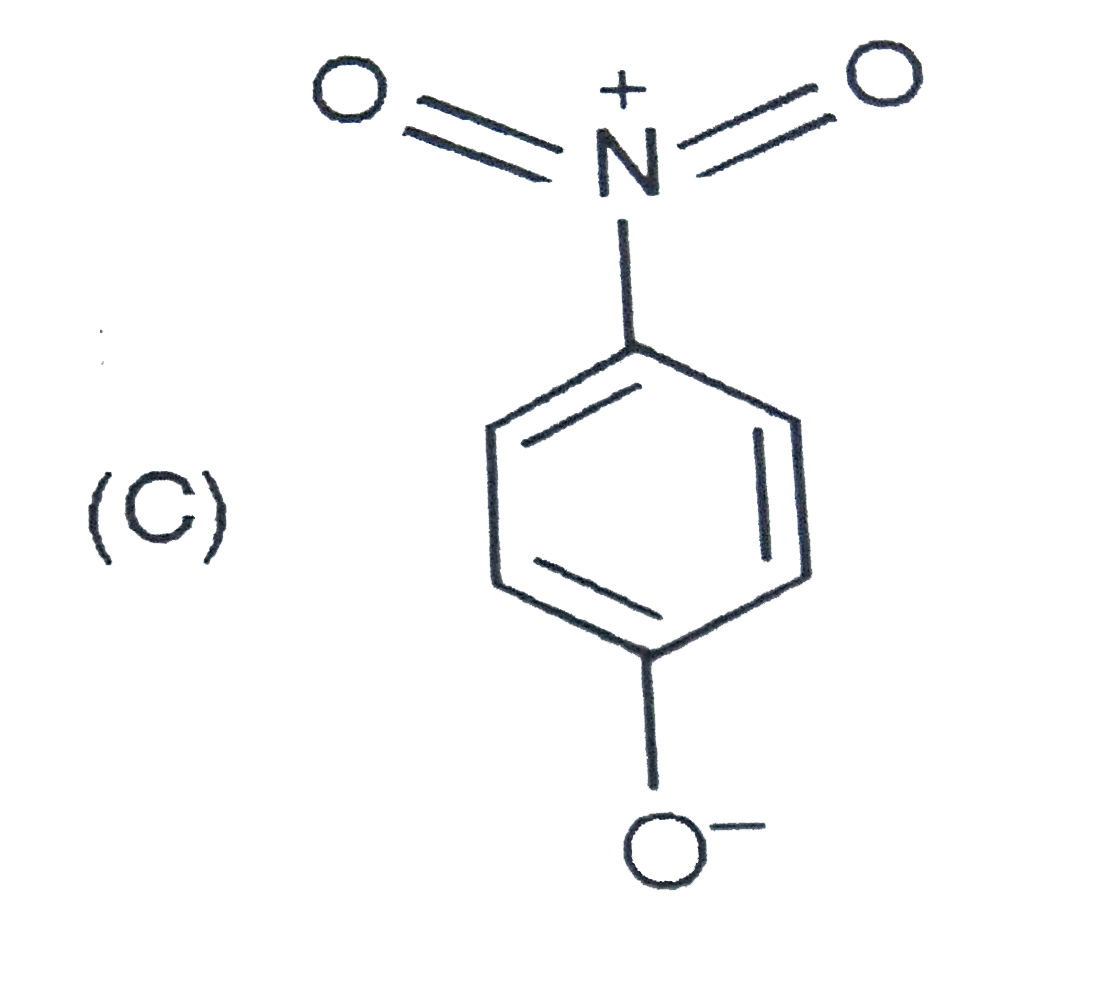
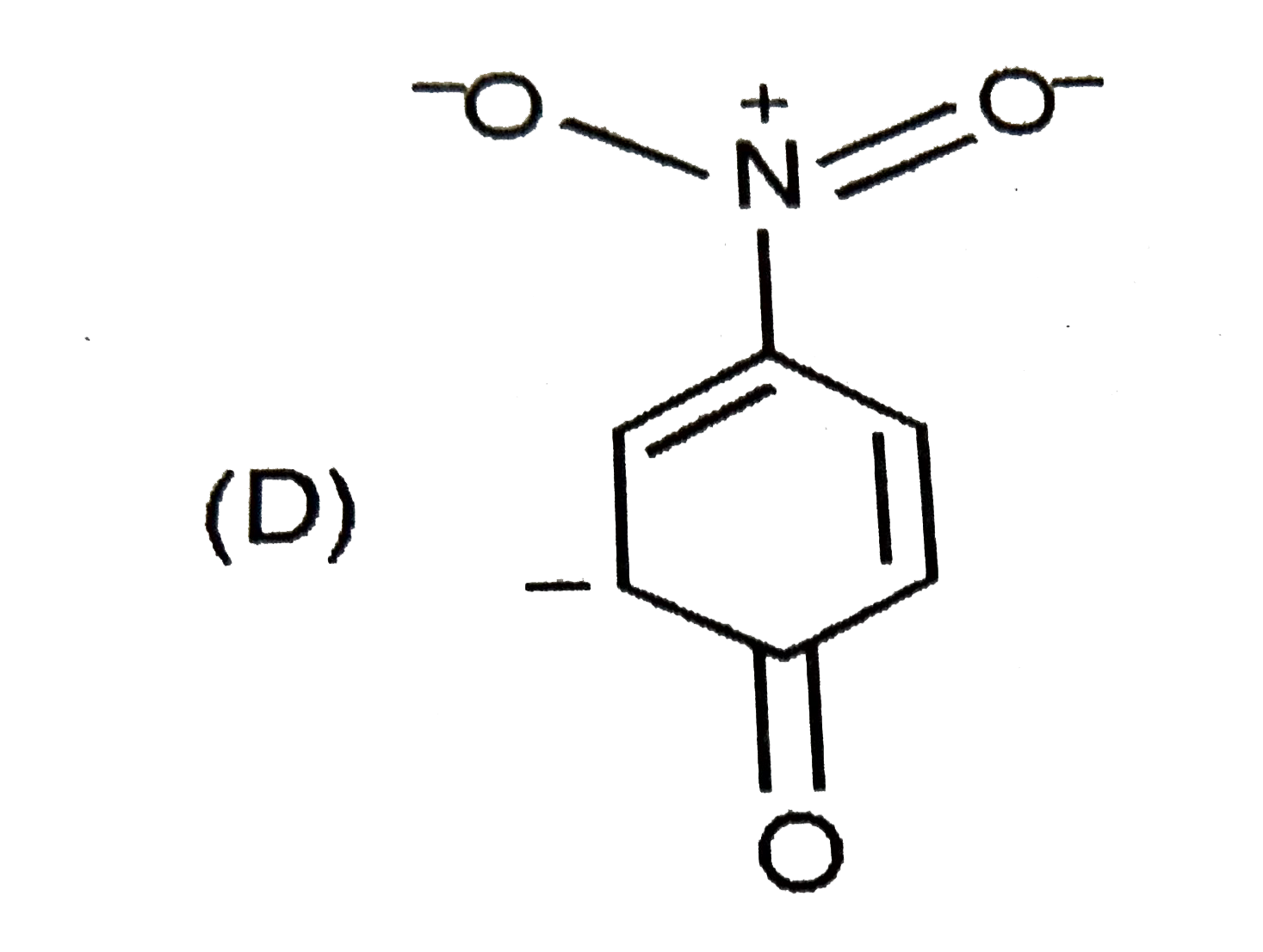
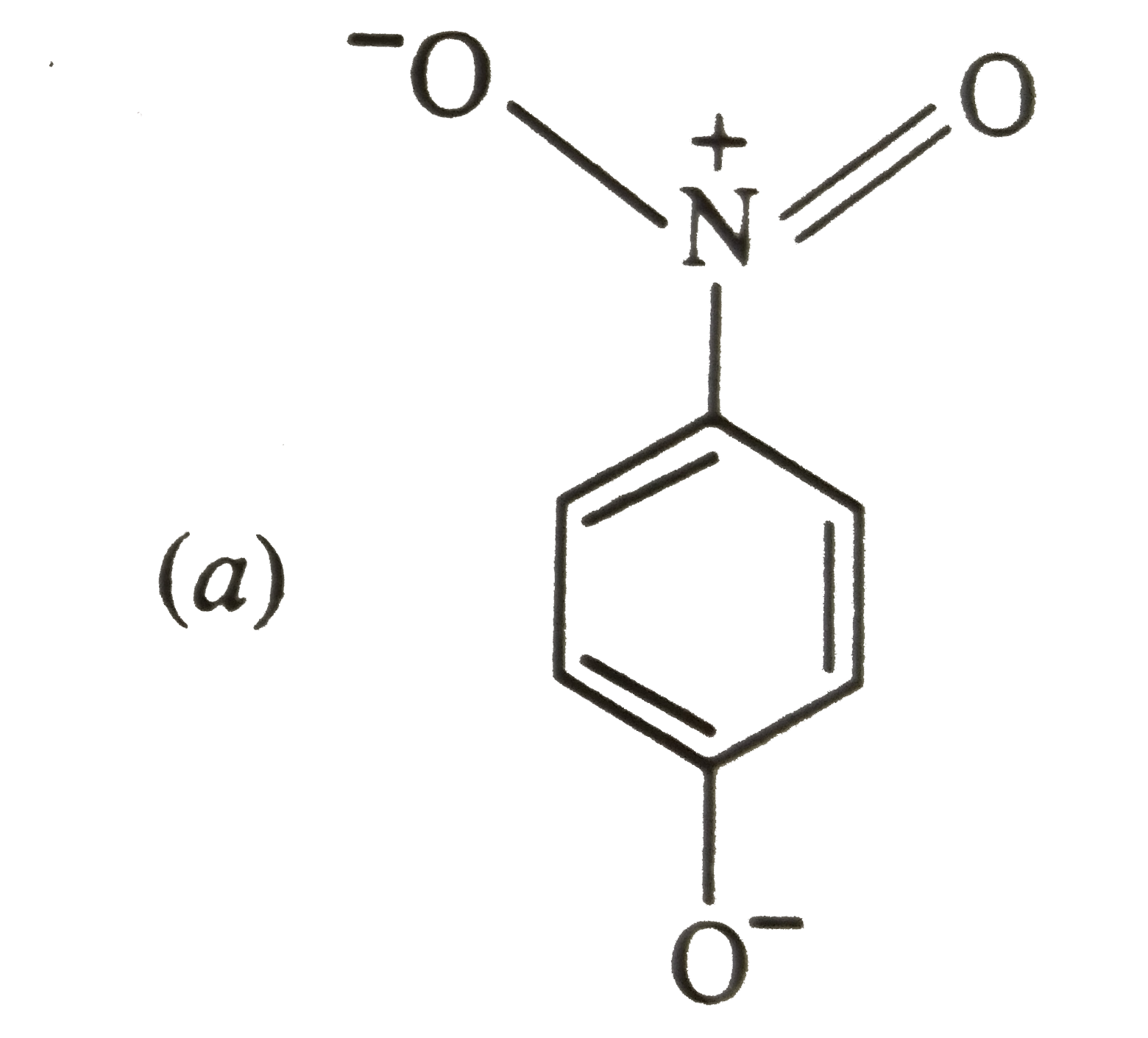
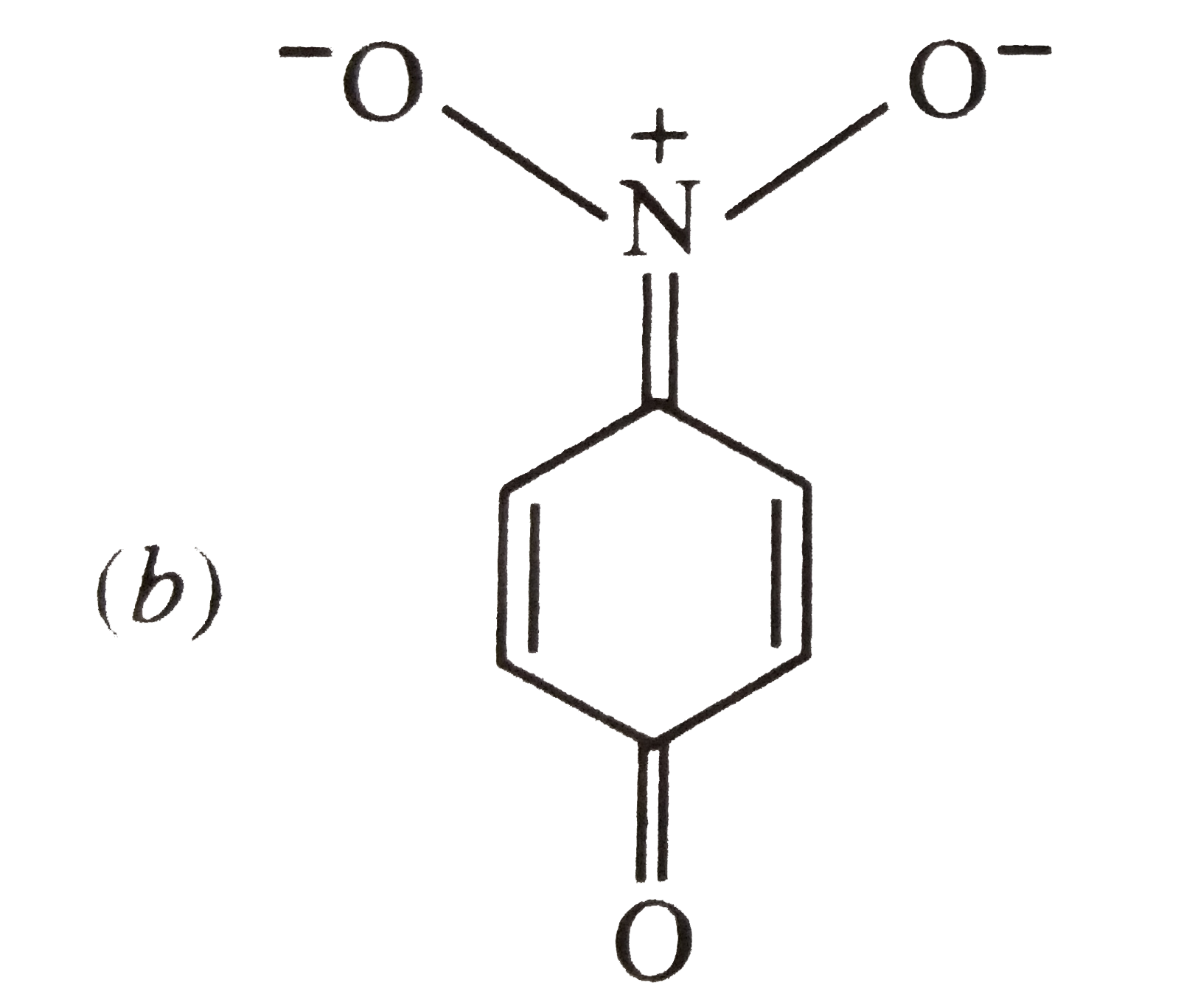
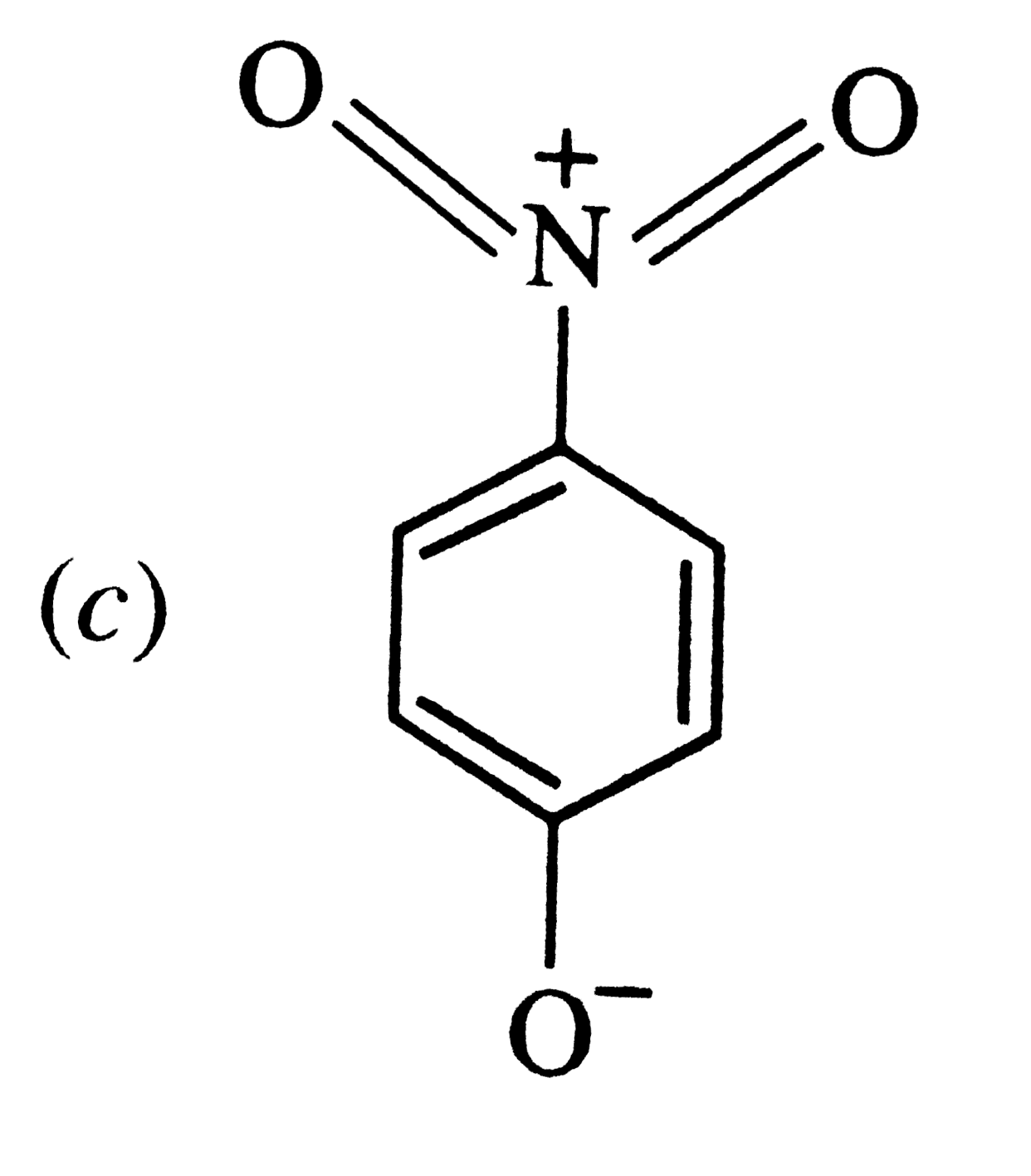
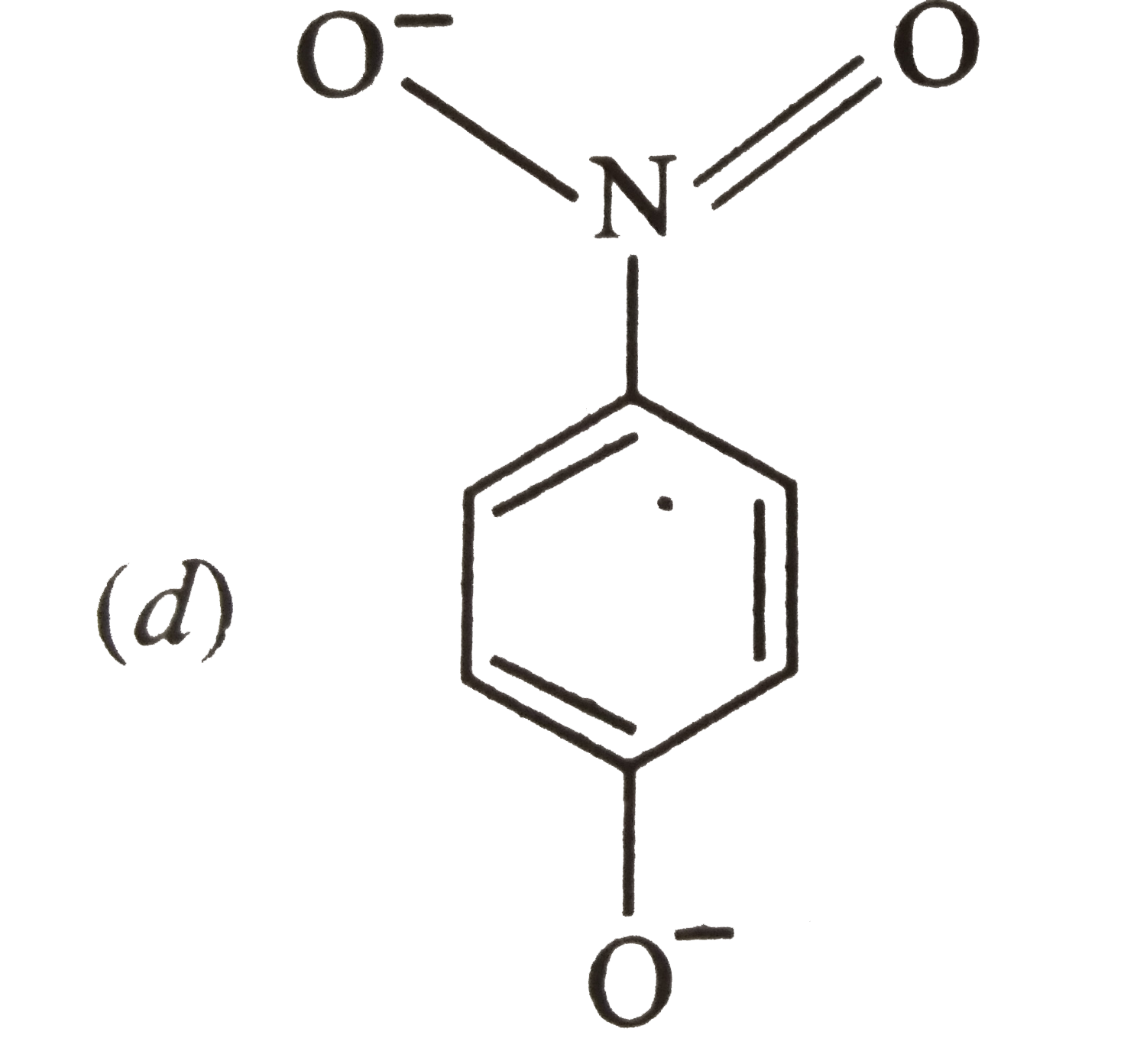
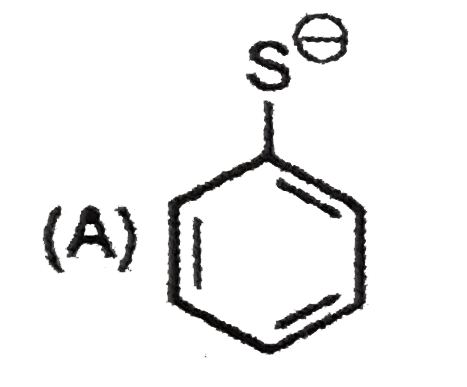
The most unlikely representation of resonance structures of p-nitrophenoxide ion is |
|
Answer»
|
|
| 6. |
The most unlikely representation of resonance structure of p-nitrophenoxide is |
|
Answer»
|
|
| 7. |
The most unlikely representation of resonance structures of p-nitrophenoxide ion is : |
|
Answer»
|
|
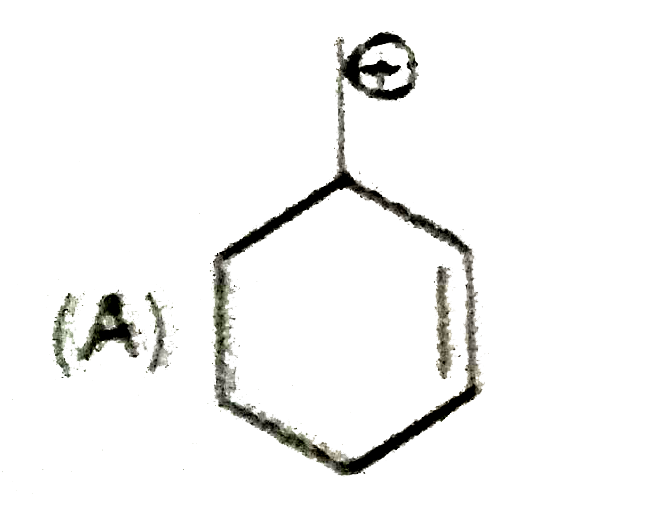
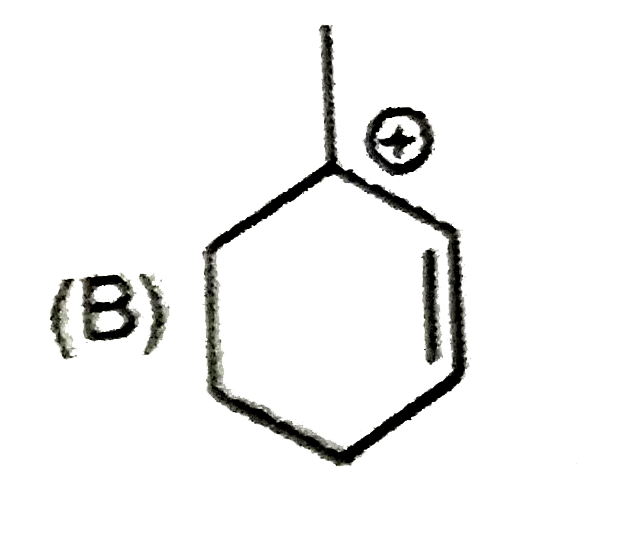
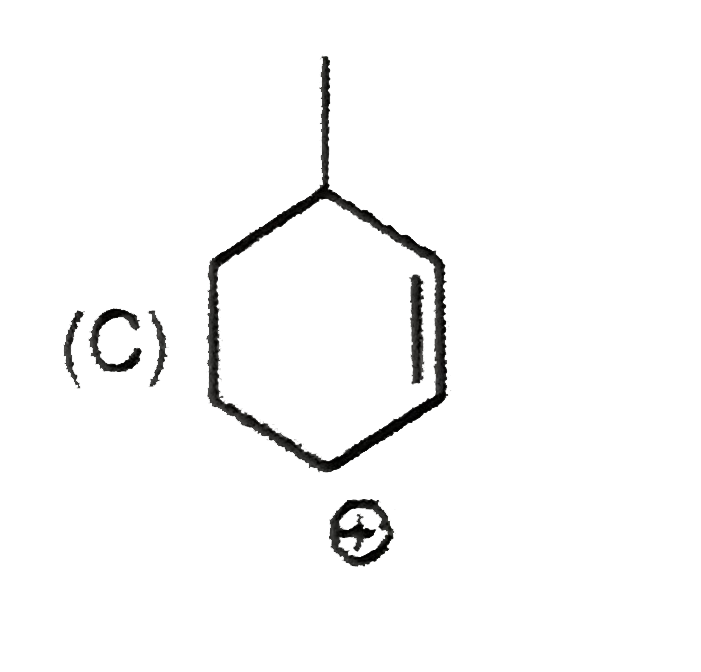
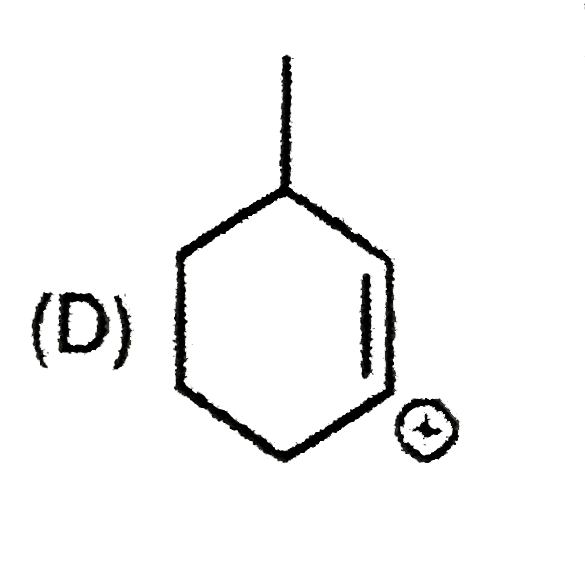
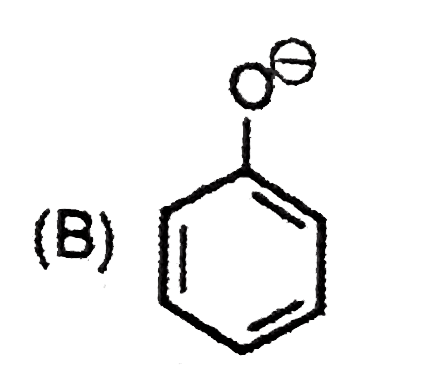
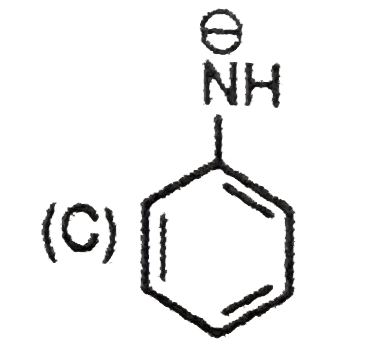
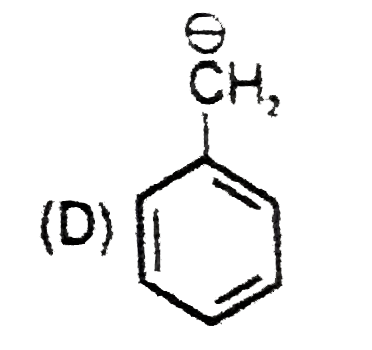
| 9. |
The most suitable reagent for the conversion ofRCH_(2)OH rarr RCHO is |
|
Answer» `KMnO_(4)` |
|
| 10. |
The most suitable reagent for the conversion of R-CH_(2)-OH rarr R-CHO is : |
|
Answer» `KMnO_(4)` |
|
| 11. |
The most suitable reagent for the conversion of primary alcohol into aldehyde with the same number of carbon atom is |
|
Answer» ACIDIFIED `K_(2)Cr_(2)O_(7)` |
|
| 12. |
The most suitable method of separation of 1:1 mixture of ortho- and para-nitrophenols is |
|
Answer» chromatography |
|
| 13. |
The most stable resonating structure of CH_(3) - O - CH = CH is |
|
Answer» `CH_(3) - UNDERSET(* *)overset(* *)O - Coverset(Theta)H-overset(OPLUS)CH_(2)` |
|
| 14. |
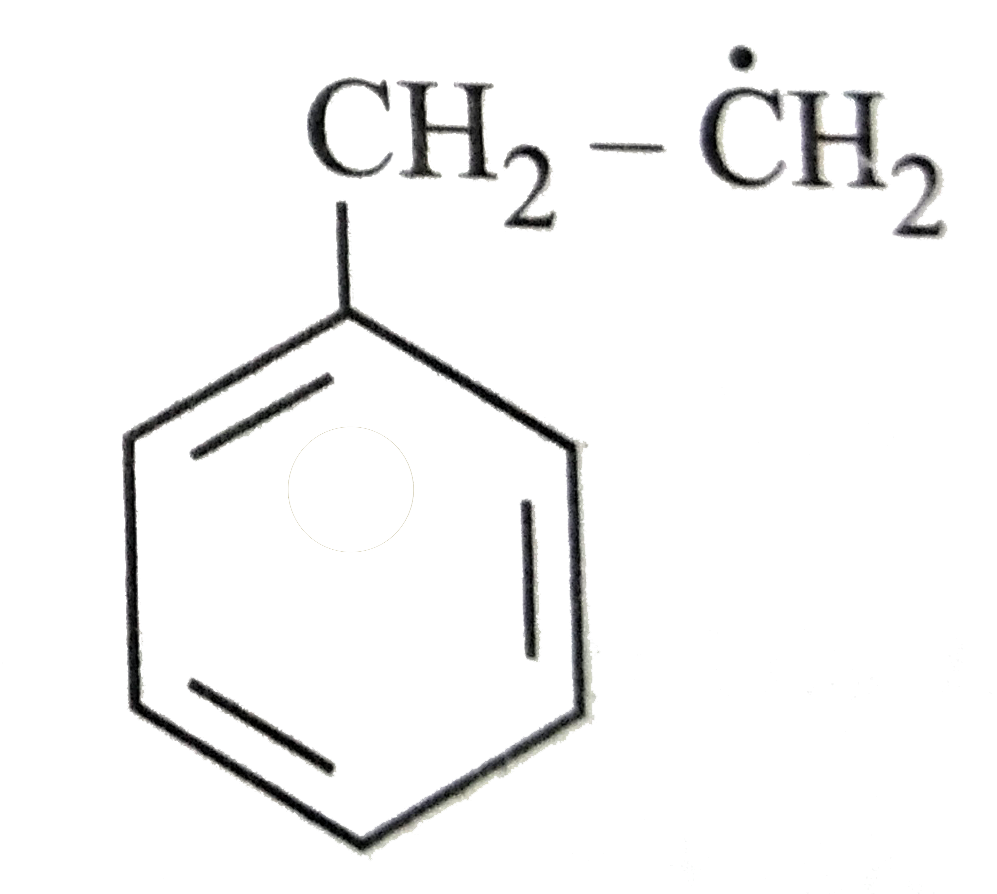
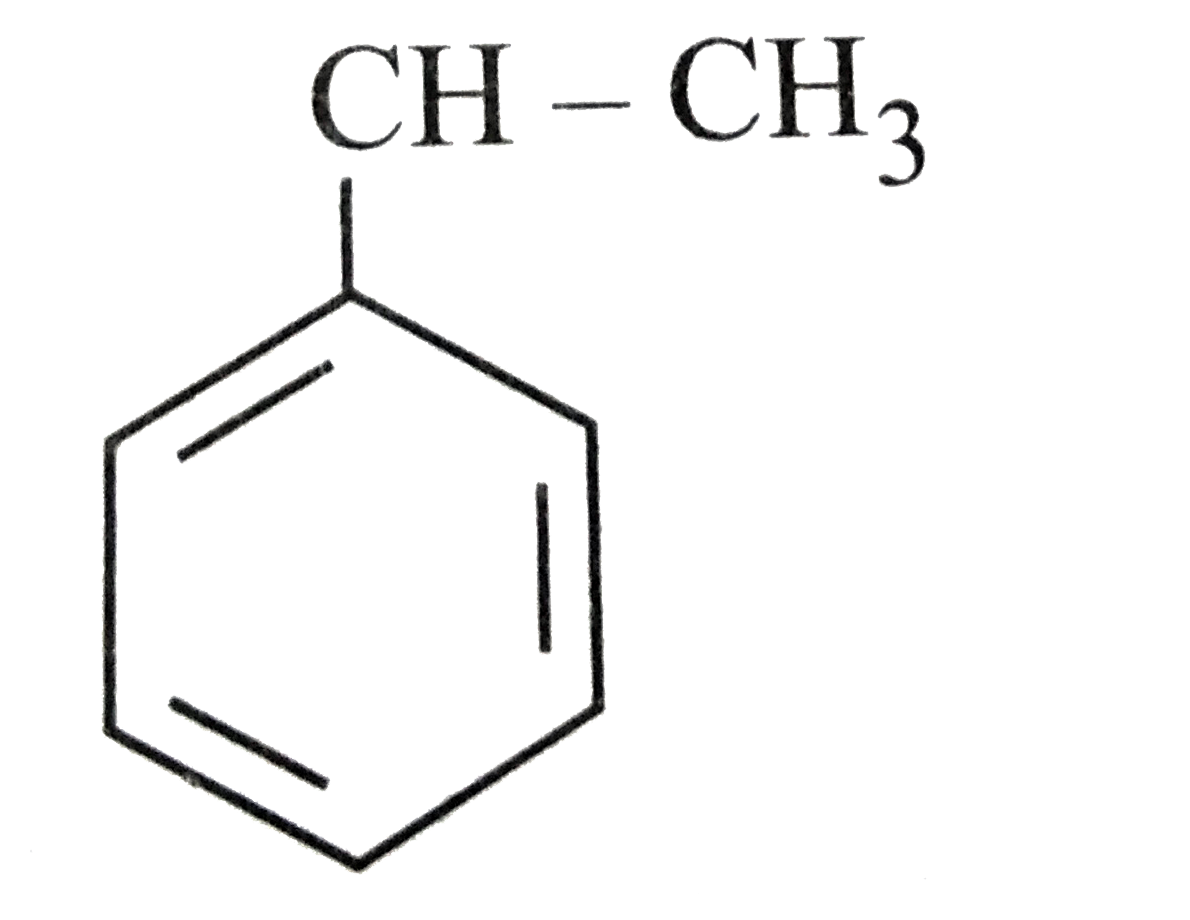
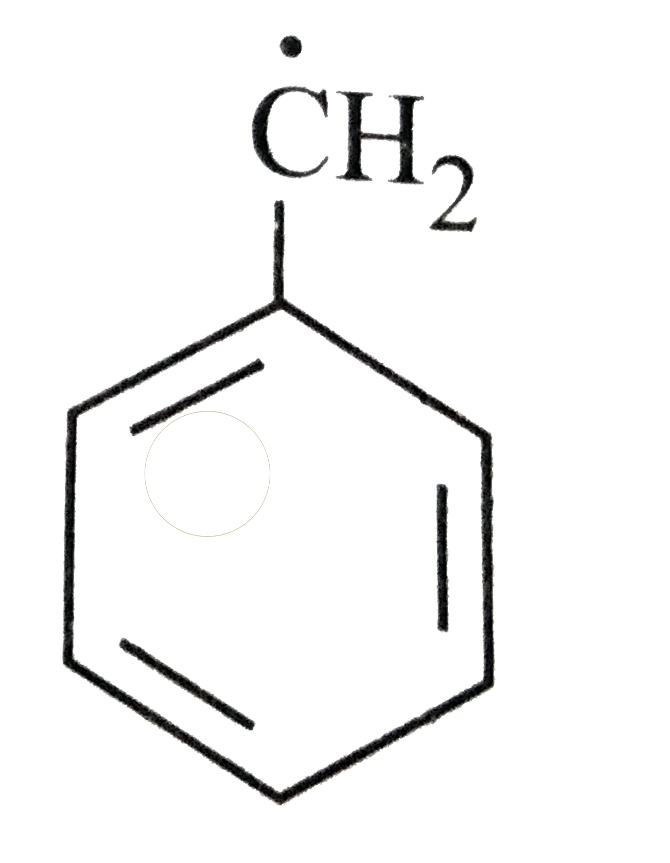
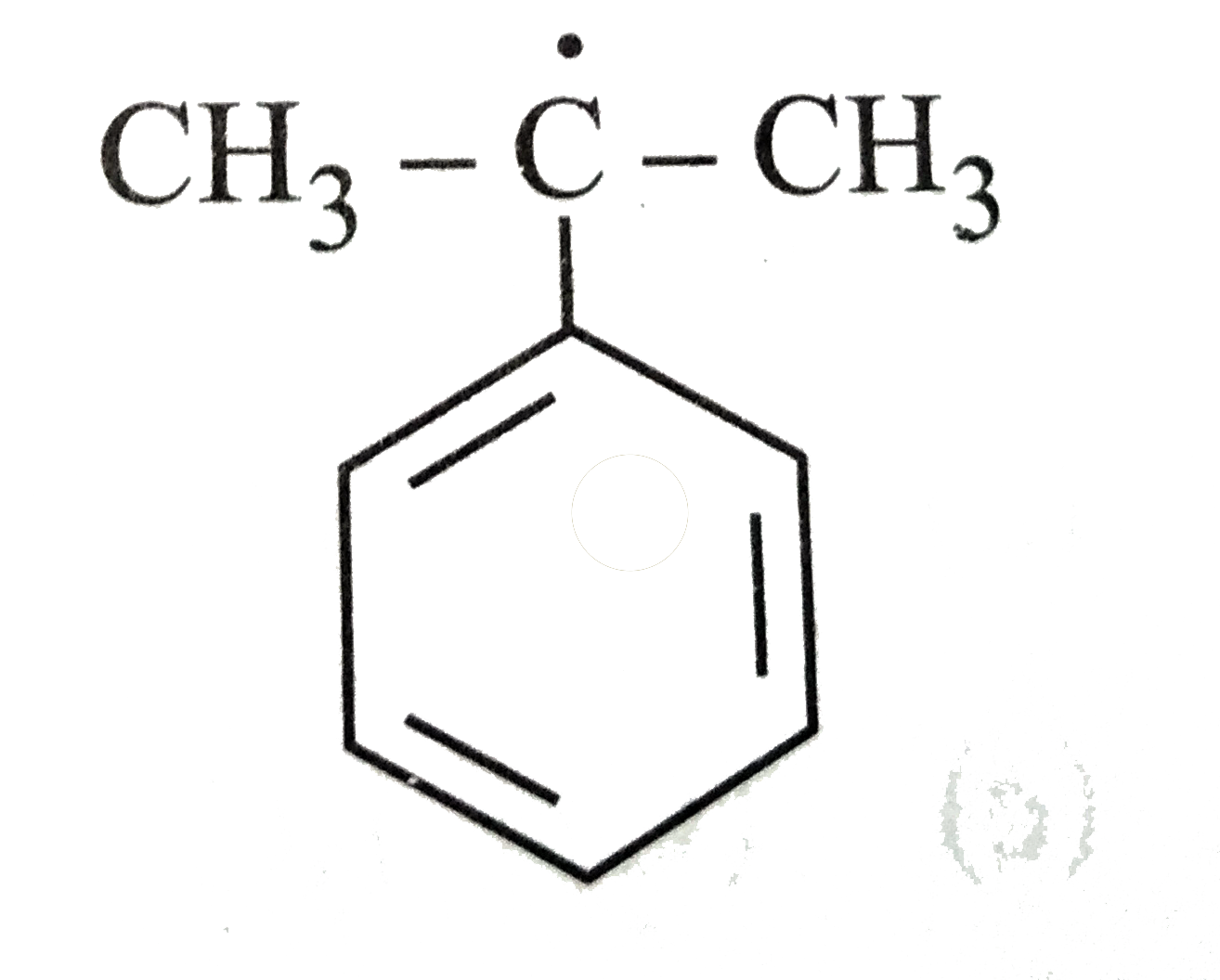
The most stable radical among the following is |
|
Answer»
|
|
| 15. |
The moststableoxidation state of aluminum is "……………."while that of thallium is "…………..". |
|
Answer» |
|
| 16. |
The most stable oxidation state of chromium is |
|
Answer» `+5` |
|
| 17. |
The most stable methylated alkene is |
|
Answer» `UNDERSET(underset(H_(3)C)(|))OVERSET(overset(H_(3)C)(|))(C )= underset(underset(CH_(3))(|))overset(overset(CH_(3))(|))(C )` |
|
| 19. |
The most stable free radical which can be isolated is |
|
Answer» Trityl radical |
|
| 20. |
The most stable free radical is |
|
Answer» `C_6H_5-overset(C_6H_5)overset(|)UNDERSET(C_6H_5)underset(|)(C^(@))` |
|
| 21. |
The most stable free radical among the following is |
|
Answer»
|
|
| 22. |
The most stable form of carbon at high temperature is X. The C - C bond length in diamond is Y while C - C bond length is graphite is Z. What are X,Y and Z respectively? |
|
Answer» Graphite `1.42 Å, 1.54 Å` |
|
| 23. |
The most stable form of trans 1,2 dimethyl cyclo hexane? |
|
Answer» (1E, 2E) |
|
| 24. |
The most stable electrophile is |
|
Answer» `R_(3)overset(OPLUS)(C)` |
|
| 26. |
The most stable conformation of n-butane is |
|
Answer» Gauche |
|
| 27. |
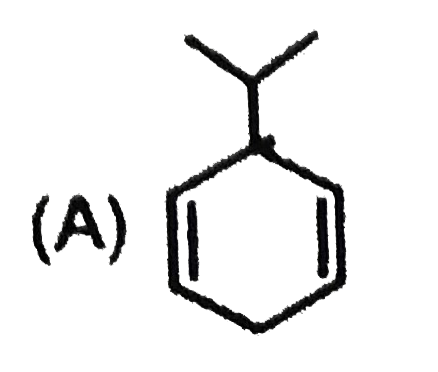
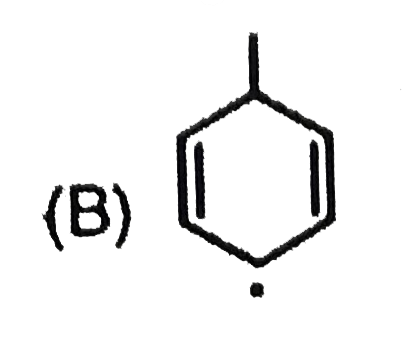
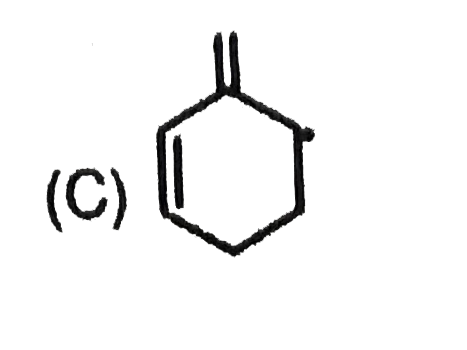
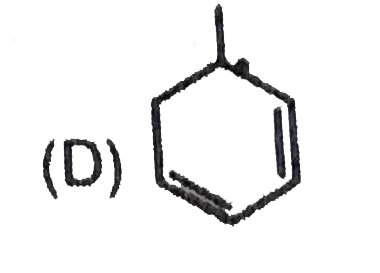
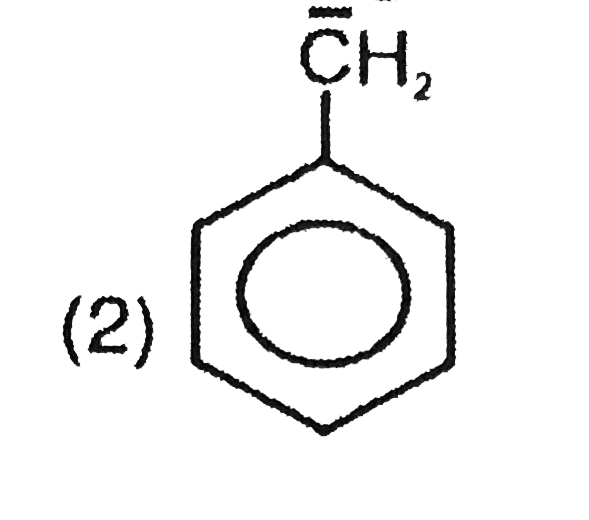
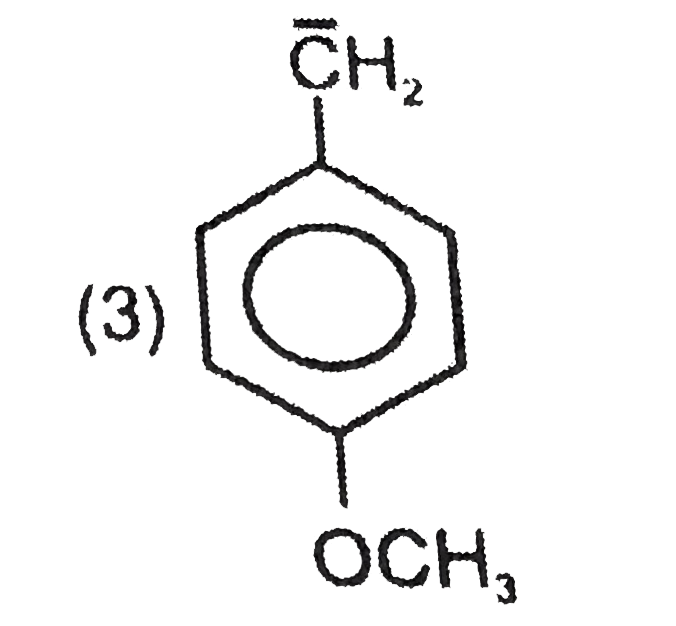
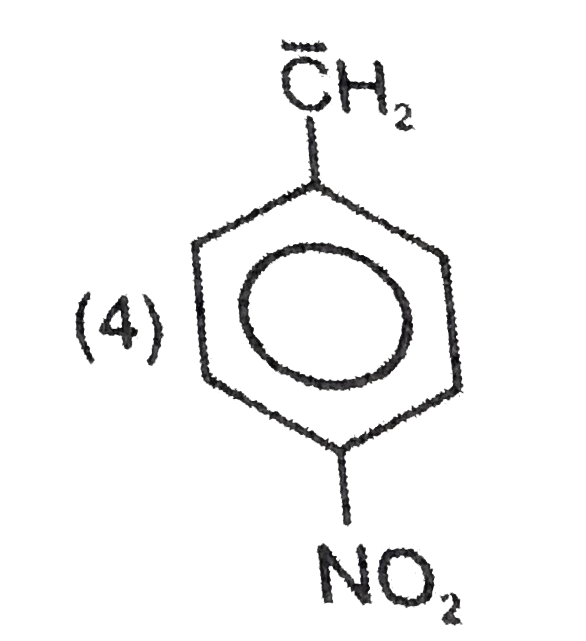
Most stable carbocation among the following is |
|
Answer» Anti 
|
|
| 28. |
The most stable conformation of ethylene glycol is |
|
Answer» anti 
|
|
| 29. |
The most stable conformation of Butane is |
|
Answer» Gauche-form |
|
| 30. |
The most stable carbonium ion is |
|
Answer»
|
|
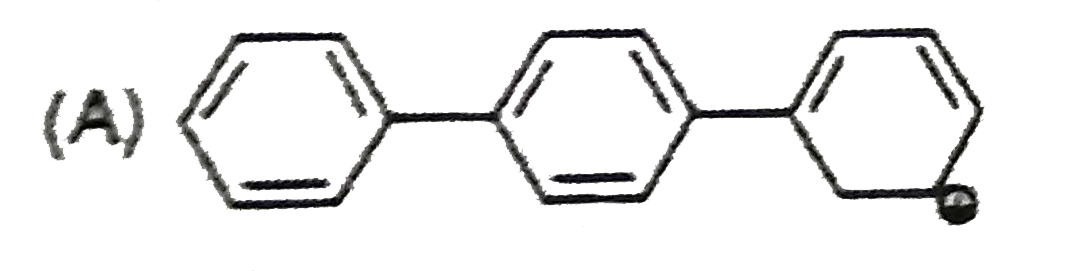
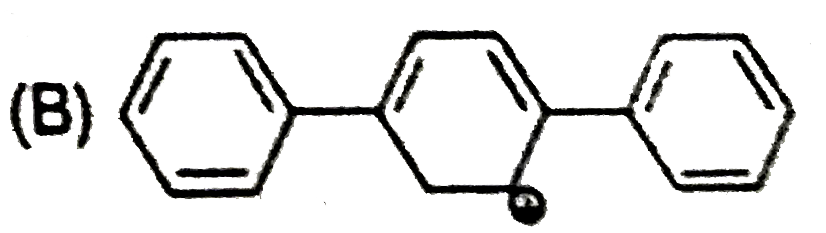
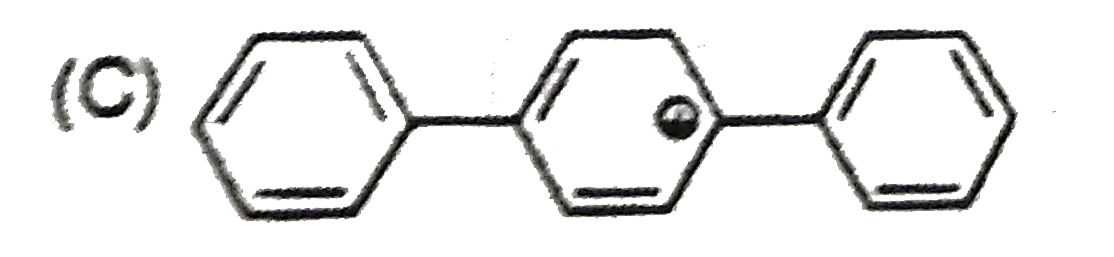
| 32. |
The most stable carbocation is: |
|
Answer»
|
|
| 33. |
The most stable carbanion is |
|
Answer» `CH_(3)-CH_(2)^(-)` |
|
| 34. |
The most stable carbanion among the following is |
|
Answer»
|
|
| 35. |
The most stable anion is: |
|
Answer»
|
|
| 36. |
The most stable alkene is |
|
Answer» `CH_(2)=CH-CH_(2)-CH_(3)` |
|
| 37. |
The most stable alkene among the following is |
|
Answer»
|
|
| 38. |
The most satisfactory method of separating sugars from each other is: |
|
Answer» fractional CRYSTALLIZATION |
|
| 39. |
The most reactive nucleophile among the following is |
|
Answer» `CH_(3)O^(-)` |
|
| 40. |
The most reactive state of hydrogen is |
|
Answer» Atomic hydrogen |
|
| 41. |
The most reactive nucleophile among the following is : |
|
Answer» `CH_(3)O^(-)` |
|
| 42. |
The most recive isotope of H is |
|
Answer» `""_(1)H^(1)` |
|
| 43. |
The most reactive compound towards electrophilic substitution is |
|
Answer» Benzene carbaldehyde |
|
| 44. |
The most probable speed of O_(2) molecules at T(K) is |
|
Answer» `sqrt((RT)/(4 pi))` |
|
| 45. |
The most probable velocity of the molecules is given by (m is mass of a molecule) |
|
Answer» `SQRT((3KT)/(m))` |
|
| 46. |
The mostprobableradius(in pm) for findingthe electronin He^(+) is |
|
Answer» 0 where `a_(0) =52.9` pm forheliumionz=2 `r_(map) = (52.9)/( 2) = 26.45 pm` |
|
| 48. |
The most probable radius (in pm) for finding the electron in He^(+) is |
|
Answer» `0.0` `:.` For `H^(+), r_(1) = (52.9 XX1^(2))/(2) = 26.5` pm |
|
| 49. |
The most probable velocity of the molecules of a gas is 1 km/sec. The R.M.S velocity of the molecules is |
|
Answer» `1.128km//SEC` |
|