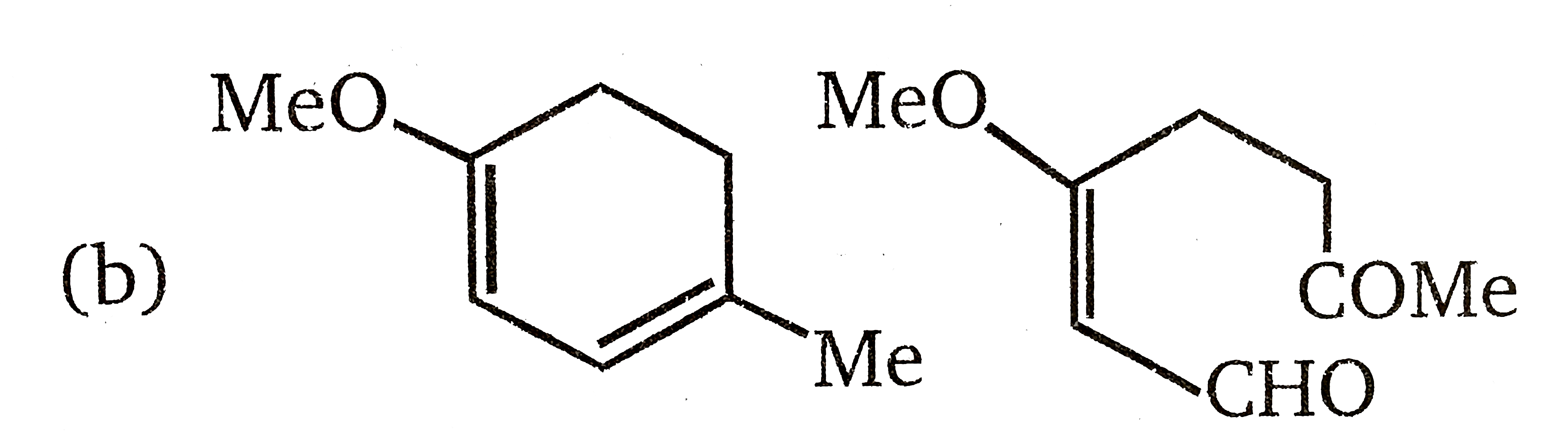Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The purple colour of which compound is in lassigne's solution in sulphur test? |
|
Answer» `Na_(4)[FE(CN)_(6)NOS]` |
|
| 2. |
The purity of H_(2)O_(2) in a given sample is 85%. The weight of impure sample of H_(2)O_(2) which required 10 ml of M//5 KMnO_(4) solution in a titration in acidic medium is 0.1 x.Find x? |
|
Answer» `10xx(1)/(5)xx5` = m eq of `H_(2)O_(2)` m. eq `H_(2)O_(2)` = 100 MEQ WEIGHT of `H_(2)O_(2)=10xx10^(-3)xx(34)/(2)=0.17gm` wt of imure `H_(2)O_(2)=(100)/(85)xx0.17=0.2gm` |
|
| 3. |
The purity of an organic solid is checked by its |
|
Answer» Sharp M.P |
|
| 4. |
The purity of an organic compound is determined by |
|
Answer» DENSITY |
|
| 5. |
The purity of an organic compound is determined by ............ . |
|
Answer» Chromatography |
|
| 6. |
The purification of the colloidal particles from crystalloid dimensions through semipermeable membrane is known as |
|
Answer» Coagluation |
|
| 7. |
The purification of aniline is occurred by which technique? |
|
Answer» Extraction by solvent |
|
| 8. |
The purification method of bauxite containing iron oxide as impurity is known as |
|
Answer» Hoope's PROCESS |
|
| 9. |
The purest form of carbon is |
|
Answer» BITUMINOUS COAL |
|
| 11. |
The pure hydrogen (99.9%) can be made by which of the following processes ? |
|
Answer» Mixing natural HYDROCARBONS of high molecular mass |
|
| 12. |
The pure crystalline substance on being heated graduallyfirst forms a turbid liquid at constant temperature and still higher temperature turbidity completely disappears. The behaviour is a characteristic of substance forming |
|
Answer» ALLOTROPIC crystal |
|
| 13. |
The pupils of eyes are dilated with a very dilute solution of an alkaloid which is : |
|
Answer» adrenaline |
|
| 14. |
The Prussian blue colour obtained in the Lassaigne' s test for nitrogen is due to the formation of |
|
Answer» Iron (II) hexacyanoferrate (III) |
|
| 15. |
The prussian blue colour obtained during the test for nitrogen by Lassaigne.s test is due to the formationof |
|
Answer» `[FE(CN)_6]_3` |
|
| 16. |
The violet colour obtained during the test of sulphur Lassaigne's test is due to the formation of |
|
Answer» `Fe_(4)[Fe(CN)_(6)]_(3)` |
|
| 17. |
The prssure in bulb dropped from 2000 to 1500 mm of Hg in 47 minutes when the contained O_(2) leaked through a small hole. The bulb was then completely evacuated A mixture of O_(2) and another gas of mol wt79 in the molar ratio 1:1 at a total pressure of 4000 mm of Hg was introduced Find the mole ratio of two gases remaining in the bulb after a period of 74 minutes . |
|
Answer» |
|
| 18. |
The Prussian blue colour confirms the presence of nitrogen in an organic compound is due the formation of ............ . |
| Answer» SOLUTION :`Fe_(4)[FE(CN)_(6)]_(3)` | |
| 19. |
The proton is heavier than an electron by _______. |
|
Answer» 1850 times |
|
| 20. |
The property of hydrogen which distinguishes it from other alkali metals is |
|
Answer» Its ELECTROPOSITIVE CHARACTER |
|
| 21. |
The property of hydroge which distinguishes it from all metal is |
|
Answer» Its electropositive CHARACTER |
|
| 22. |
The property of crystalline solid is not |
|
Answer» anisotropic |
|
| 24. |
What are the two properties of carbon that make it capble of forming a large number of compounds ? |
|
Answer» |
|
| 25. |
The property of an element that is not normals determined directly but is obtained indirectly using Born-Haber cycle |
|
Answer» IONISATION potential |
|
| 26. |
The property of a system which depends only on the nature of the substance contained in the system but not on the amount of the substance is called "…................" property. |
| Answer» SOLUTION :INTENSIVE | |
| 27. |
The propertiesas of alkali meterls are mentioed in List-I Match tyhier order of change as Present inList_II The correct match is |
| Answer» Answer :1 | |
| 28. |
The properties of the elements are the periodic function of their atomic number. The statement is given by |
| Answer» Solution :Mosely. | |
| 29. |
The proper value of significant figures in 38.0 +0.0035+0.00003 is. |
|
Answer» 38 |
|
| 30. |
The progress of the reaction A nB with time t is shown in Fig. From this information answer the following questions The equilibrium constant K_C is |
|
Answer» 1.2 |
|
| 31. |
The progress of the reaction A harr nB with time t is shown in Fig. From this information answer the following question The value of n, is |
|
Answer» 1  at equilibrium 0.3 MOLES of A REACTED to GIVE 0.6 moles of B `implies n=(0.6)/(0.3)=2` |
|
| 32. |
The product(s) via-oxymercuration (HgSO_4+H_2SO_4) of 1-Butyne would be : |
|
Answer» `CH_3-CH_2-oversetoverset(O)(||)C-CH_3` |
|
| 33. |
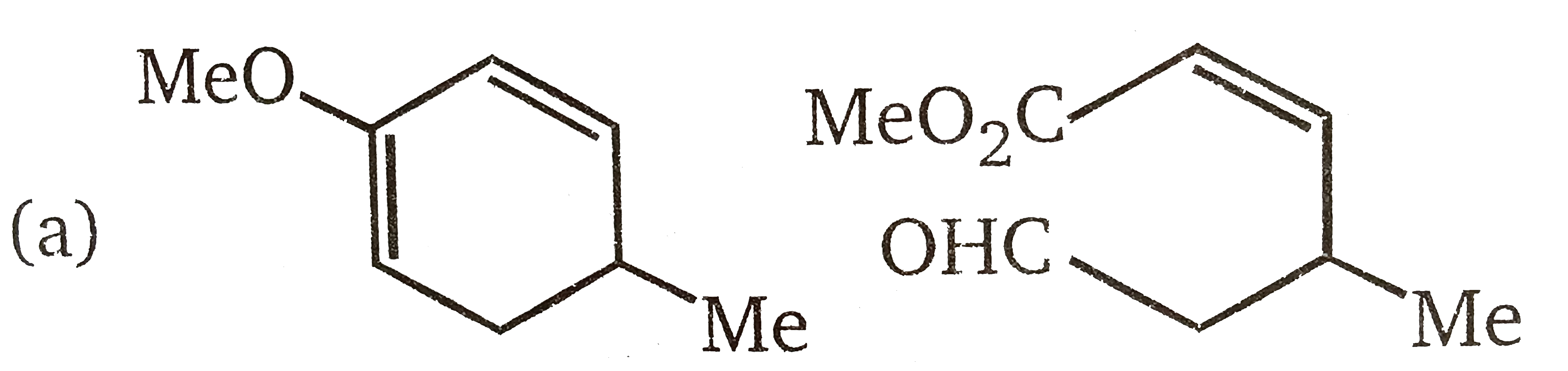
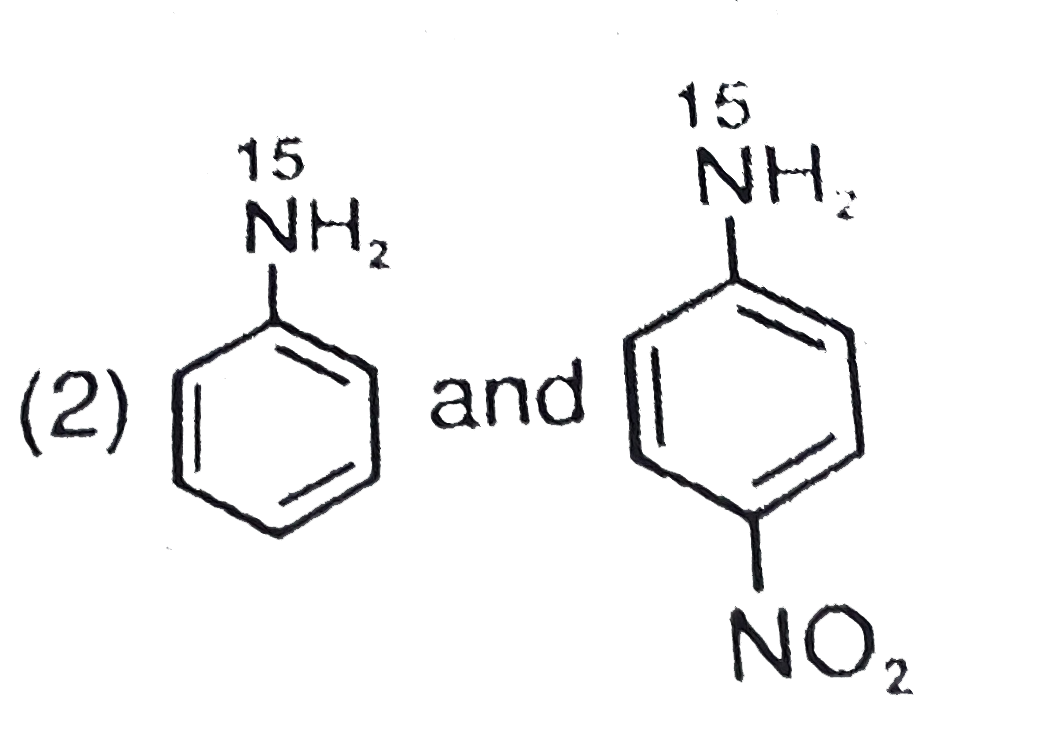
The products P and Q in the following sequence of reactions, are |
|
Answer»
 More NUCLEOPHILIC ALKENE WIL ATTACK. |
|
| 34. |
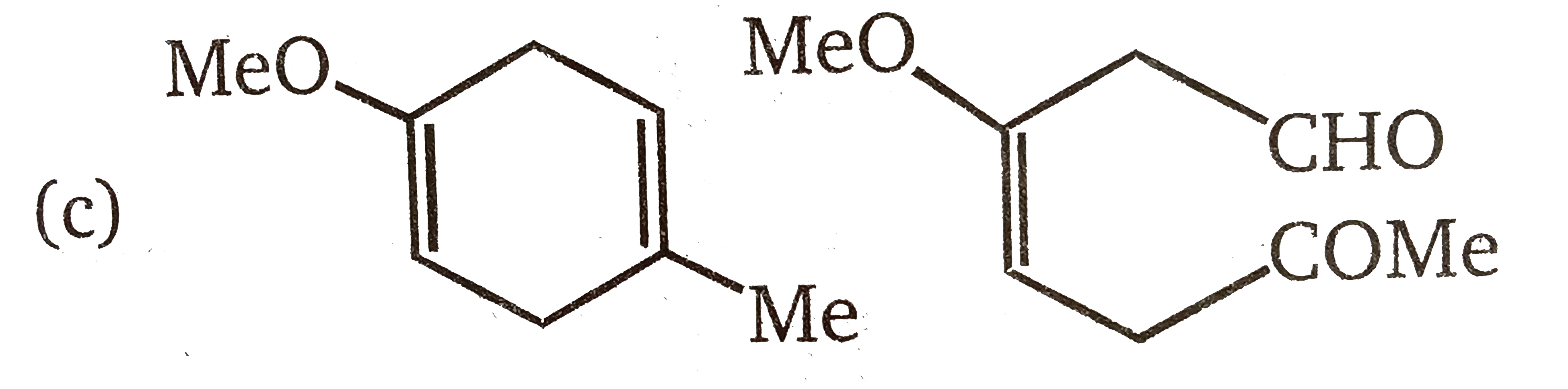
The product(s) of the following reactions sequence is(are) |
|
Answer»
|
|
| 35. |
The products of the following reactions is C_2H_5Br + 2Na+ CH_3Br underset"Propane"overset"dry ether"to? |
| Answer» Solution :Ethane, propane and butane | |
| 36. |
The products of the following reaction are ______________, respectively. i. C_(2)H_(5)OH overset("hot. "Al_(2)O_(3))rarr ii. C_(2)H_(5)OH overset("hot Cu")rarr |
|
Answer» ACETALDEHYDE, ethene |
|
| 37. |
The products of the following reaction are, SiO_(2)+Coverset(Delta)to |
|
Answer» Si,C and `CO_(2)` |
|
| 38. |
The products of ozonolysis of an alkene are ethanl and methanal, then that alkene is |
|
Answer» `CH_(3)-C-=C-CH_(3)` |
|
| 39. |
The products of oxidation of phopshrous acid by hot concentrated sulphuirc acid are………..and………… . |
|
Answer» |
|
| 40. |
The products of combustion of an aliphatic thiol (RSH) at 298 K are |
|
Answer» `CO_(2)(g), H_(2)O(g)` and `SO_(2)(g)` |
|
| 41. |
The products of electrolysis of concentrated common salt solution are |
|
Answer» `Na+Cl_(2)` |
|
| 42. |
The product(s) obtained via oxymercuration (HgSO_4 + H_2SO_4) of 1-butyne would give |
|
Answer» `CH_3CH_2-oversetoversetO(||)C-CH_3` |
|
| 43. |
The products obtained on heating LiNO_(3) will be |
|
Answer» `LiNO_(2) + O_(2)` |
|
| 44. |
The products formed when acetic anhydride is treated with ammonia in excess is/are |
|
Answer» `2CH_(3)COONH_(4)` |
|
| 45. |
The products formed when boron trichloride is reduced with lithium aluminium hydride are |
|
Answer» `B_(2)H_(6) and HCl` |
|
| 46. |
The products formed when a mixture of the following two amides( I and II) are treated with bromine and aqueous KOH are |
|
Answer»
|
|
| 47. |
The products formed are : |
|
Answer» `CH_(3)-OVERSET(O)overset(||)(C)-CHO` |
|
| 48. |
The products formed during the cracking long chain hydrocarbon C_(6)H_(12), are ....... |
| Answer» SOLUTION :`C_(6)H_(6)+3H_(2)` | |
| 49. |
The products for the following reactions are (i) CH_(3)-underset(H)underset(|)overset(Br)overset(|)(C)-CH_(2)-CH_(3)+"alc. KOH"to X (ii) CH_(3)-underset(CH_(3))underset(|)(C)H-CH=CH_(2) overset(O_(3))to Y+Z |
|
Answer» `X=(CH_(3))_(2)C=CH_(2),Y=CH_(3)CH_(2)CHO,Z=CH_(3)CH_(2)CHO` 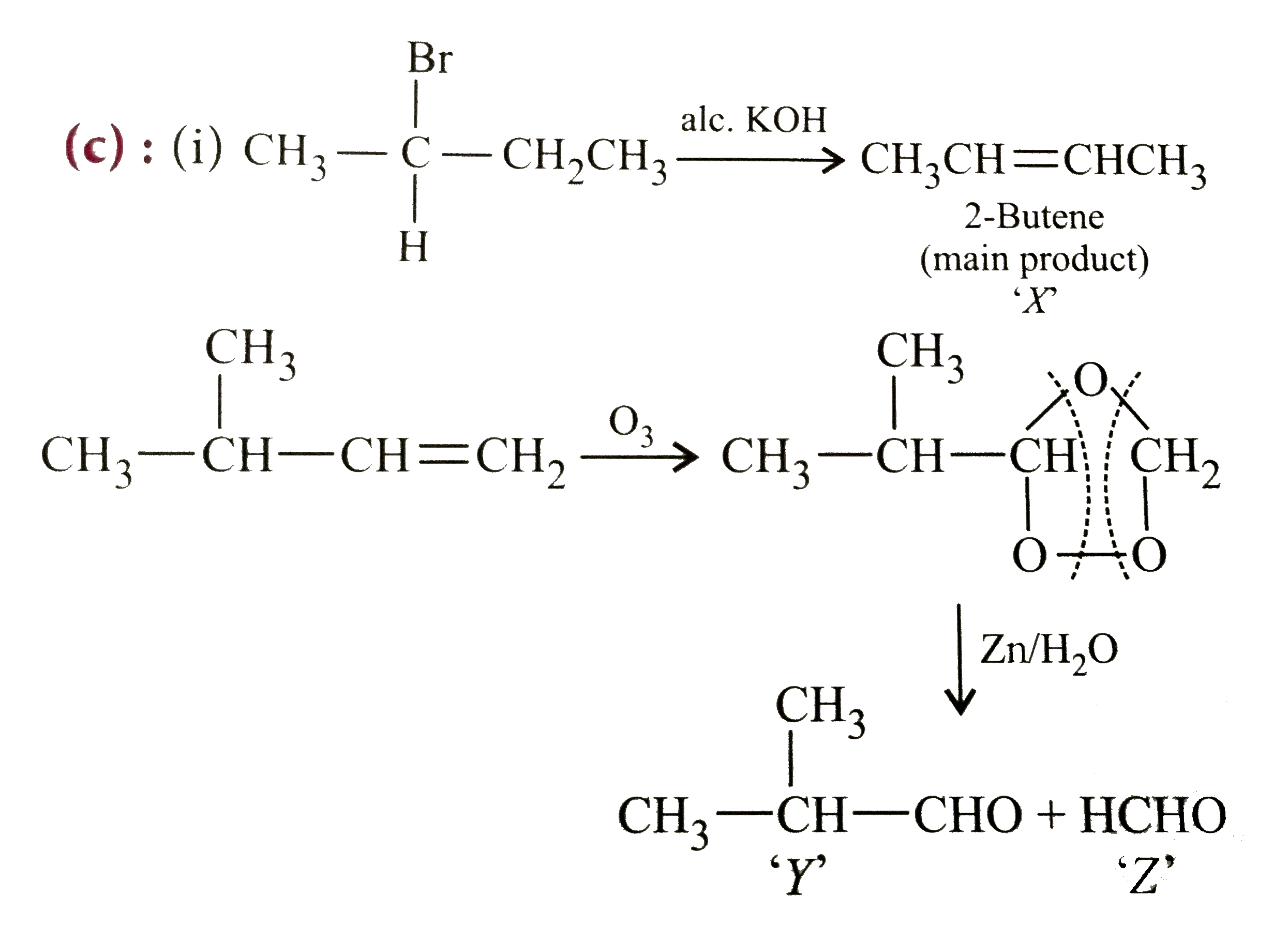
|
|
| 50. |
The products expected to be formedin the Wurtz reaction of a mixture of neopentyl bromide and isobutyl bromide are (i)2,2,4-trimethylpentane (ii)2,2,5,5-tetramethylhexane (iii)2,2,4,4-tetramethylhexane (iv)2,5-dimethyl hexane (v)2,2,5-trimethylhexane |
|
Answer» (ii),(iii) and (v) |
|