Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following statement are correct for an electronthat has n = 4 and m = - 27 |
|
Answer» The ELECTRON may be in a d-orbital `n = - 2` `s = + 1//2` |
|
| 2. |
What is the reason to pass bry air in combustion tube in estimation of C and H? |
| Answer» Solution :If the AIR is DRY than it contain `H_(2)O`. So correct WEIGHT of `H_(2)O` is not obtained | |
| 3. |
The value of K_(c) for the reaction 2AhArrB+C is 2xx10^(-3). At a given time, the composition of reaction mixture is [A]=[B]=[C]=3xx10^(-4)M. In which direction the reaction will proceed ? |
|
Answer» Forward DIRECTION |
|
| 4. |
What would be the IUPAC named for element with atomic number 120 ? |
|
Answer» Ununbium |
|
| 5. |
Which of the following arrangements represent increasing oxidation number of the central atom? |
|
Answer» Solution :Writing the O.N of Cr CI and Mn on each SPECIES in the foiur set of IONS we have (a) `CrO_(2)^(-) ,CIO_(3)^(-),CrO_(4)^(2-), MnO_(4)^(-)` (b) `CIO_(3)^(-),CrO_(4)^(2-),MnO_(4)^(-),CrO_(2)^(-)` (c ) `CrO_(2)^(-),CIOO_(3),MnO_(4)^(-),CrO_(4)^(2-)` (d) `CrO_(4)^(2-), MnO_(4)^(-), CrO_(2)^(-),CIO_(3)^(3)` only in arrangement (a) the O.N of CENTRAL ATOM incrases form left to right therefore OPTION (a) is correct |
|
| 6. |
Which combination is odd with respect to oxidation number ? |
|
Answer» `H_(2) SO_(5) , H_(2) S_(2) O_(8) ,K_(2)Cr_(2) , SF_6` |
|
| 7. |
Which of the following weighs less when weighed I magnetic field? |
|
Answer» `ScCl_(3)` |
|
| 8. |
Which of the following statements is INCORRECT for liquid ? |
|
Answer» Molecules in the bulb of liquid experience balanced forces and the resultant NET force is zero. |
|
| 9. |
Which one of the followingpairs of solutions is not an acidic buffer ? |
|
Answer» `H_(2)CO_(3)+Na_(2)CO_(3)` |
|
| 10. |
Which alkali metal salt is obtained as hydrated crystal ? |
|
Answer» Li |
|
| 11. |
The suspected carcinogeic water pollutant is |
|
Answer» MIC |
|
| 12. |
Which one of the following arrangements represent the correct order of least negative to most negative electron gain enthalpy |
|
Answer» `AlltOltClt CaltF` |
|
| 13. |
What is smog? How is classical smog different from photochemical smogs? |
|
Answer» Solution :The word smog is derived from smoke and fog. This is the most COMMON example of air pollution that occurs in many cities throughout the world. There are TWO types of smog: - (i) Classical smog : This type of smog occurs in cool humid climate. It is a mixture of smoke, fog and sulphur dioxide. It is ALSO called as reducing smog because it is chemically reducing mixture. (ii) Photochemical smog : This type of smog occurs in WARM, dry and sunny climate. It is produce due to the action sunlight on unsaturated HYDROCARBONS and nitrogen dioxide produced by automobiles and factories. It is also called as oxidising smog because it has high concentration of oxidising agent. |
|
| 14. |
Three centred bond is present in |
|
Answer» `NH_3` |
|
| 15. |
Write the IUPAC name of the following compound. CH_(3) - underset(underset(CH_(3))(|))overset(overset(CH_(3))(|))(C )-CH_(2)-overset(overset(CH_(2)CH_(3))(|))(CH)-underset(underset(CH_(2)CH_(3))(|))(CH)-overset(overset(CH_(2)CH_(3))(|))(CH)- CH_(3) |
|
Answer» SOLUTION :The given compound can be written as `CH_(3)underset(CH_(3))underset(|)OVERSET(CH_(3))overset(|)C-CH_(2)-overset(CH_(2)CH_(3))overset(|)C-underset(CH_(2)CH_(3))underset(|)C-overset(CH_(3))overset(|)C-CH_(3)-CH_(3)` The IUPAC name is 4,5 - Diethy1-2,2,6-trimethylocatane. |
|
| 16. |
The starting materials for the formation of silicone polymers are |
|
Answer» Silicates |
|
| 17. |
Which of the following is a component of Portland cement? |
| Answer» Answer :D | |
| 18. |
Thermodynamically most stable form of carbon is: |
|
Answer» Diamond |
|
| 20. |
The shape of sulphate ion is |
|
Answer» Tetrahedral |
|
| 21. |
What is meant by chirality of a compound? Give one example. |
Answer» SOLUTION :A compound is said to be chiral if it exists in TWO stereoisomeric forms which are non-superimposable MIRROR images of each other. This property of non-superimposability is called chirality and the two COMPOUNDS which are non-superimposable mirror images of each other are called enantiomers. 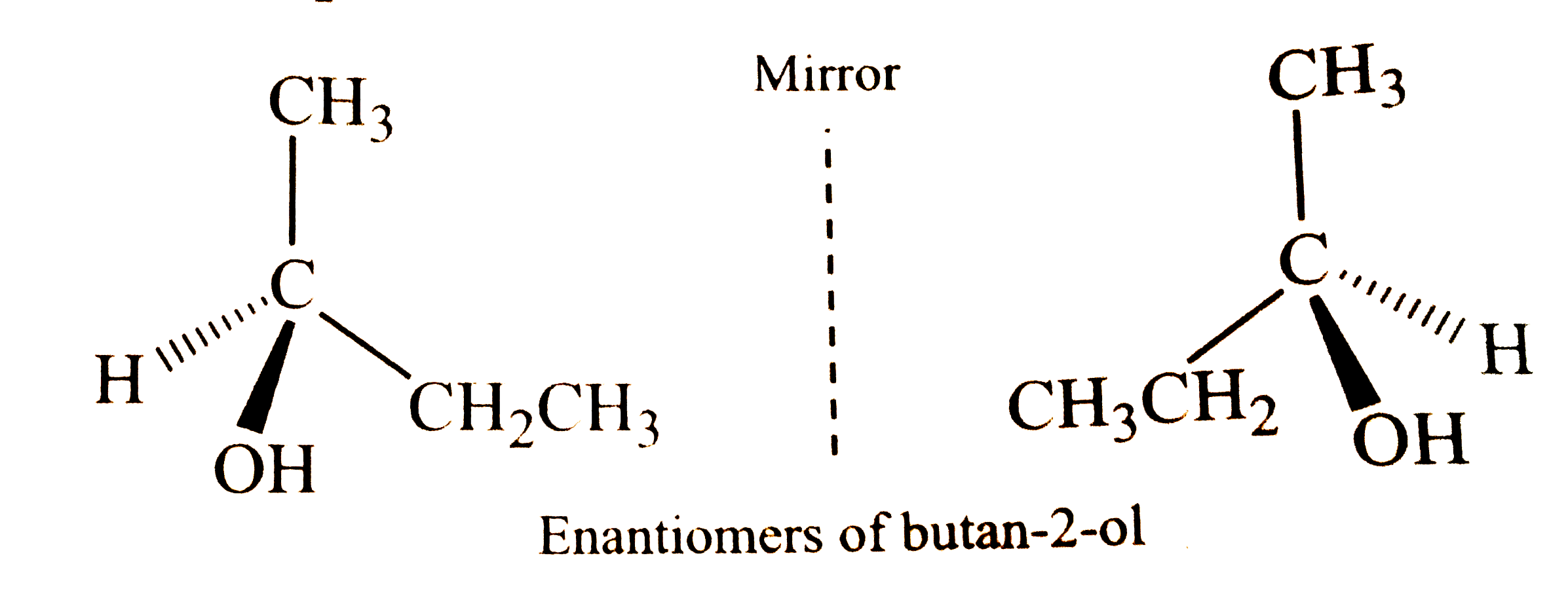 For example, butan-2-ol is a chiral compound because it exists in two STEREOISOMERS which are non-superimposable mirror images of each other. |
|
| 22. |
The volume of CO_(2) formed when 1 litre of O_(2) reacted with 2 lit of CO under the same condition is |
|
Answer» 1L |
|
| 23. |
When Na_2CO_3aqueous solution is being titrated with hydrochloric acid {:("Column-I " , "Column-II") ,((A) " At the start of titration", (P) " Buffer solution of " HCO_3^(-) and CO_3^(-2)),((B) "Before the first equivalent point " , (Q) "Buffer solution "H_2CO_3and HCO_3^(-) ), ((C) "At the first equivalent point",( R ) "Amphiprotic anion "pH =(1//2) (p^(ka_1) +p^(Ka_2))), ((D) "Between first and second equivalent points" , (S) "Hydrolysis of "CO_3^(-2)) :} |
|
Answer» ` CO_3^(2-) + H^(+) to HCO_3^(-) ` until ` 1^(ST) ` equation pt, both are present i.e., buffer of ` HCO_3^(-)and CO_3^(2-) ` At Ist equation pt only`HCO_3^(-) `present amphiprotic anion after Ist eq pt, untill 2nd equation pt ` HCO_3^(-)+HCl to H_2CO_3` i.e.,buffer of `H_2CO_3 and HCO_3^(-) ` |
|
| 24. |
When 4.2 g of sodium bicarbonate are added to a solution of acetic acid weighing 10.0 g, it is observed that 2.2 g of carbon dioxide is released into the atmosphere. The residue left is found to weigh 12.0 g. Show that these observations are in agreement with the law of conservation of mass. |
|
Answer» Solution :Sodium bicarbonate `(NaHCO_3)` reacts with acetic acid (`CH_3COOH`) according to the FOLLOWING equation. `UNDERSET(4.2 g)(NaHCO_(3)) + underset(10.0g)(CH_(3)COOH) to underset("Residue" 12.0g)(CH_(3)COONa+ H_(2)O) + underset(2.2 g)(CO_(2))` The total mass of the reactants `(NaHCO_(3) + CH_(3)COOH) = 4.2+ 10.0 = 14.2 g` The total mass of products `(CH_(3)COONa + H_(2)O + CO_(2)) = 12.0 + 2.2 = 14.2 g` Since, the total mass of the products is the same as that of the reactants, the observations are in agreement with the LAW of conservation of mass. |
|
| 25. |
Zinc is not considered as a transition metal because |
|
Answer» It is DIAMAGNETIC |
|
| 26. |
Whyare noble gases chemically inert? |
| Answer» Solution :NOBLE gaseshaving completely FILLED ELECTRONIC configuration so neiither accept nor lose theirelectronreadily and hence they are CHEMICALLY inert in NATURE. | |
| 27. |
Which compound show tautomerism : |
|
Answer» Ph-NO |
|
| 28. |
When alkyl substituted acetylene undergoes addition with hydrogen in presence of Lindlar's catalyst, the alkene formed is |
|
Answer» A mixture of CIS and TRANS isomers |
|
| 29. |
Which of the following is hcp crystal structure ? |
|
Answer» NaCl |
|
| 30. |
Which of the following are inoic earbides ? |
|
Answer» `CaC_(2)` |
|
| 31. |
What is the use of alloy prepared by mixing lithium and lead (white metal) ? |
| Answer» Solution : Alloy prepared by MIXING LITHIUM and lead (White metal) is USED to prepare bearings for motor ENGINES. | |
| 32. |
Thermodynamics is not concerned about |
|
Answer» CHANGE in energy INVOLVED in a chemical reaction. |
|
| 33. |
Which is the most acidic oxide of group 14 elements? |
| Answer» SOLUTION :With INCREASE in non-metallic CHARACTER of an element, the ACIDIC character of its oxide increases. Hence carbondioxide is more acidic among GROUP 14 elements. | |
| 34. |
How many protons, neutrons and electrons are present in an atom of tritium ? |
|
Answer» |
|
| 35. |
Which of the following compounds may give reaction with acidic KMnO_(4) ? |
|
Answer»
|
|
| 36. |
Which of the following phenols is most soluble in aqueous sodium bicarbonate ? |
|
Answer» 2,4-dihydrozyacetophenone |
|
| 37. |
Which of the following changes decrease the vapour pressure of water kept in a sealed vessel ? |
|
Answer» Decreasing the QUANTITY of water However, vapour pressure also decreases with DECREASE in temperature as kinetic energy of the molecules decrease. |
|
| 38. |
Which has strongest H-bond ? |
|
Answer» S - H ..... O `overset(+ DELTA )(H) - overset(- delta)(F) ` BOND is most POLAR . |
|
| 39. |
Which of the following reactions leads the formation of colloidal solution ? |
|
Answer» `CU + HgCl_(2) to CuCl_(2) + Hg` |
|
| 41. |
What is dipole moment (mu) of bond ? Explain by example. |
|
Answer» Solution :Polar bonding : As a result of polarization, the molecule possesses the dipole MOMENT. The dipole moment is defined as the product of the magnitude of the charge (Q) and the distance between the centres of POSITIVE and negative charge. " Mathematically : `mu = Q xx r ` Where, Q = charge on atom in coulomb r = Distance between to centre in METER. `mu` = Dipole moment in Debye units and ` D = 3.33564 xx 10^(-30)` cm Bond dipole moment express method : Dipole moment is a vector quantity and by convention. It is depicted by a small arrow `rarr` with tail on the negative centre `LARR` and head pointing towards the positive centre. In chemistry presence of dipole moment is represented by the CROSSED arrow `(rarr)` put on Lewis structure of the molecule. the cross in on positive and find arrow head is on negative end. For example the dipole moment of HF may be represented as e.g., `underset("Polarity in bond")(H^(delta+)) - F^(delta-) "So"Hunderset("polarity in bond") overset(rarr)(-)underset(..)overset(..)(F):` 
|
|
| 42. |
What are the important features of lattice enthalpy? |
|
Answer» Solution :Higher LATTICE ENERGY shows greater electrostatic attraction and therefore a stronger bond in the solid. 2. The lattice enthalpy is greater for IONS of higher charge and smaller RADII. |
|
| 43. |
Which of the following orders of acid strength is correct |
|
Answer» `RCOOH gt ROH gt HOH gt HC-= CH` 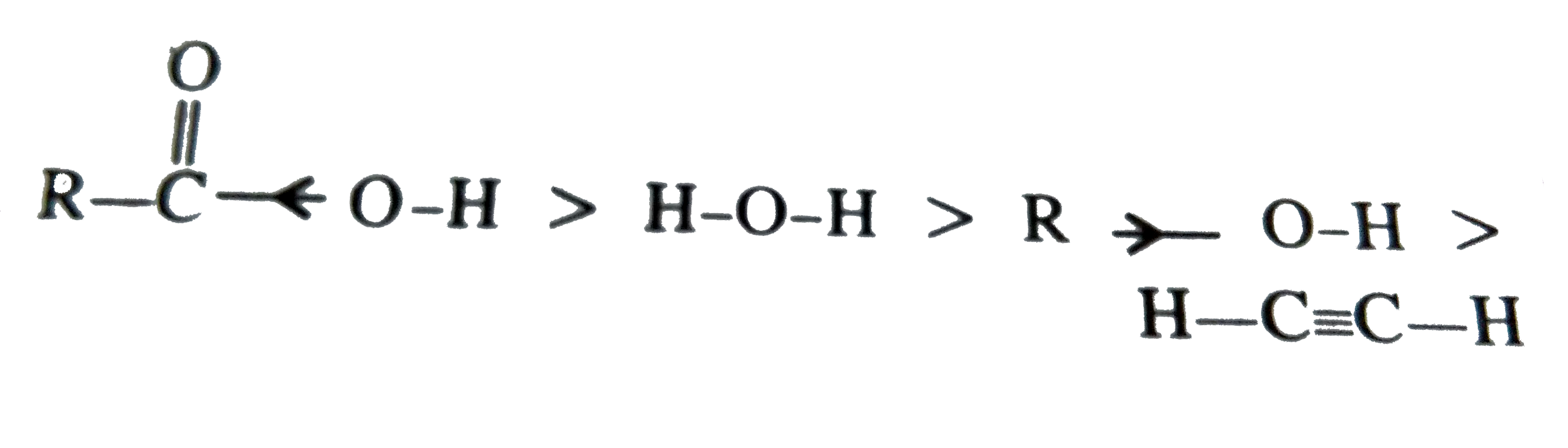
|
|
| 44. |
Which of the following elements is the hardest? |
|
Answer» BORON |
|
| 45. |
The vicimal diols can be detected by the reaction of the compound with |
|
Answer» `NaOH` and `I_(2)` The vicinal diols can be detected with `HIO_(4)` or`PB(CH_(3)COO)_(4)` |
|
| 47. |
Which compound will have highest boiling point ? |
|
Answer» `CH_(4)` |
|
| 48. |
Which of the following compounds will not be obtained as a product in the above reaction? |
|
Answer»
|
|
| 49. |
Which of the following does not contain fused benzene ring? |
|
Answer» Naphthalene |
|