Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is not a peroxide? |
|
Answer» `KO_(2)` |
|
| 2. |
What is the action of alkali metals with water? |
|
Answer» SOLUTION :Alakli metals reacts with water to form metal hydroxides and liberate hydrogen gas. `2M + 2H_(2) O to 2MOH + H_(2) UARR` [M = K, Rb, CS violent reaction] EX: `2Li + 2H_(2) O to 2LiOH + H_(2) uarr` [Slow reaction] `2Na + 2H_(2) O to 2NaOH + H_(2) uarr` (vigorous reaction) |
|
| 3. |
Which of the following species is not electrophilic in nature ? |
|
Answer» `CI^(+)` |
|
| 4. |
Whodiscoveredthe ratioof chargeto mass of electon and how ?Expalinwithdiagram |
|
Answer» SOLUTION :Valueof `e//m_(C ) : `in1897 J.J.Thomsonmeasuredthe ratioof electrical charge(e ) tothe massofelectron`e//m_(c ) = 1.758820 xx10^(11)C KG^(-1) (equ 2.1)` Where`m_(e ) = ` mass ofthe electronin kg the electronin coloumb  Expeiment- Dterminationmethodof `e//m_(e ) :`thomsonusedcathoderay tube toget the valueof `e//m_(s )`Thomsonappliedelectricalandmagneticfieldperpendicularto EACHOTHER on the pathelectron Observation : When onlyelectricfieldappliedthe electronsthedeviate from theirpath andand hitthecathoderayat point A.Simillarywhenonlymagneticfieldisapplied electronsirlkesthe cathoderaypoint C. bycarefullybalancingthe electricaladn magneticfieldstrengthit ispossibleto bringscreenat point B Bycrryingaccuratemeasurementsthomson wasable thdeterminethe valueof `e//m_(e )` |
|
| 5. |
Write the structures of two complex metal hydrides which are used as reducing agent is organic synthesis. |
| Answer» SOLUTION :`LiAlH_(4) and NaBH_(4)` | |
| 6. |
The value of Vander Waal's constant a' is maximum for |
|
Answer» HELIUM |
|
| 7. |
What happens when aluminium reacts with NaOH in presence of water molecules ? |
| Answer» | |
| 8. |
The volume of CO_(2) obtained by the complete decomposition of one mole of NaHCO_(3) at STP is |
|
Answer» `22.4L` |
|
| 9. |
Which one of the following is used as fibre -swelling agent in textile processing ? |
|
Answer» CHLOROBENZENE |
|
| 10. |
Which of the following pairs of substance on reaction will evolve H_2 gas ? |
|
Answer» IRON and dilute `H_2SO_4` |
|
| 11. |
The set of quantum numbers not applicable to an electron is |
|
Answer» 1,1,1,+1/2 |
|
| 12. |
Which one of the following orders is not in accordance with the property stated against it ? |
|
Answer» `HI gt HBR gt HCL gt Hf` :Acidic property in water The CORRECT order is : `[Cl_2 gt Br_2 gt F_2 gt I_2]` |
|
| 13. |
What is coordinate bond? |
| Answer» SOLUTION :In a chemical bonding both the electrons for sharing between atoms are CONTRIBUTED (donated) by one ATOM only. | |
| 14. |
Which of the following statement is incorrect about resonance? |
|
Answer» The most stable RESONANCE STRUCTURE EXPLAINS all the CHARACTERISTICS of a species. |
|
| 15. |
What would be the value of Delta n_(g) for the reaction NH_(4) Cl (s) hArr NH_(4) (g) + HCl (g) ? |
| Answer» Solution :`Delta n_(g) = n_(p) - n_(r) = 2 - 0 = 2` | |
| 16. |
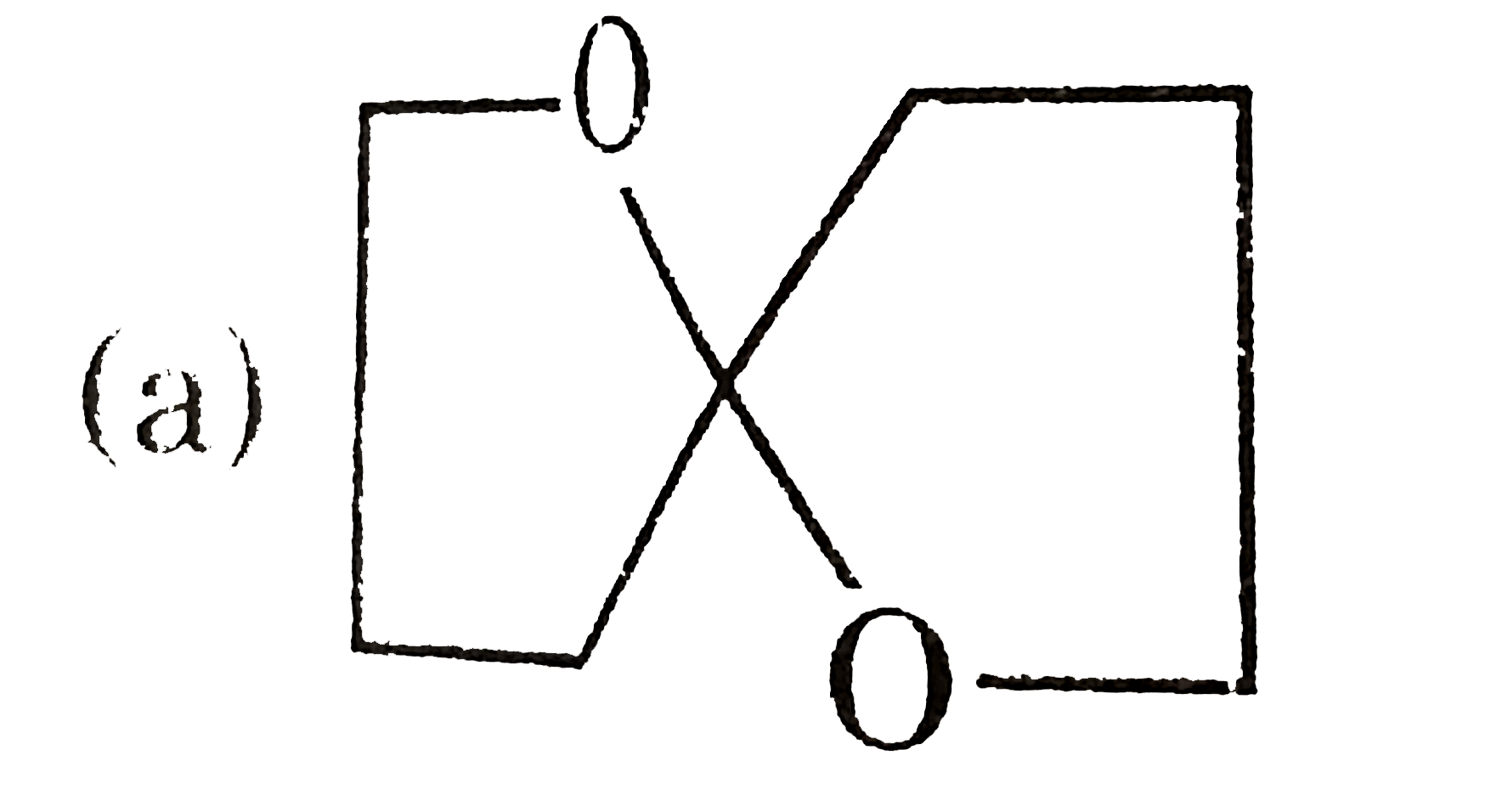
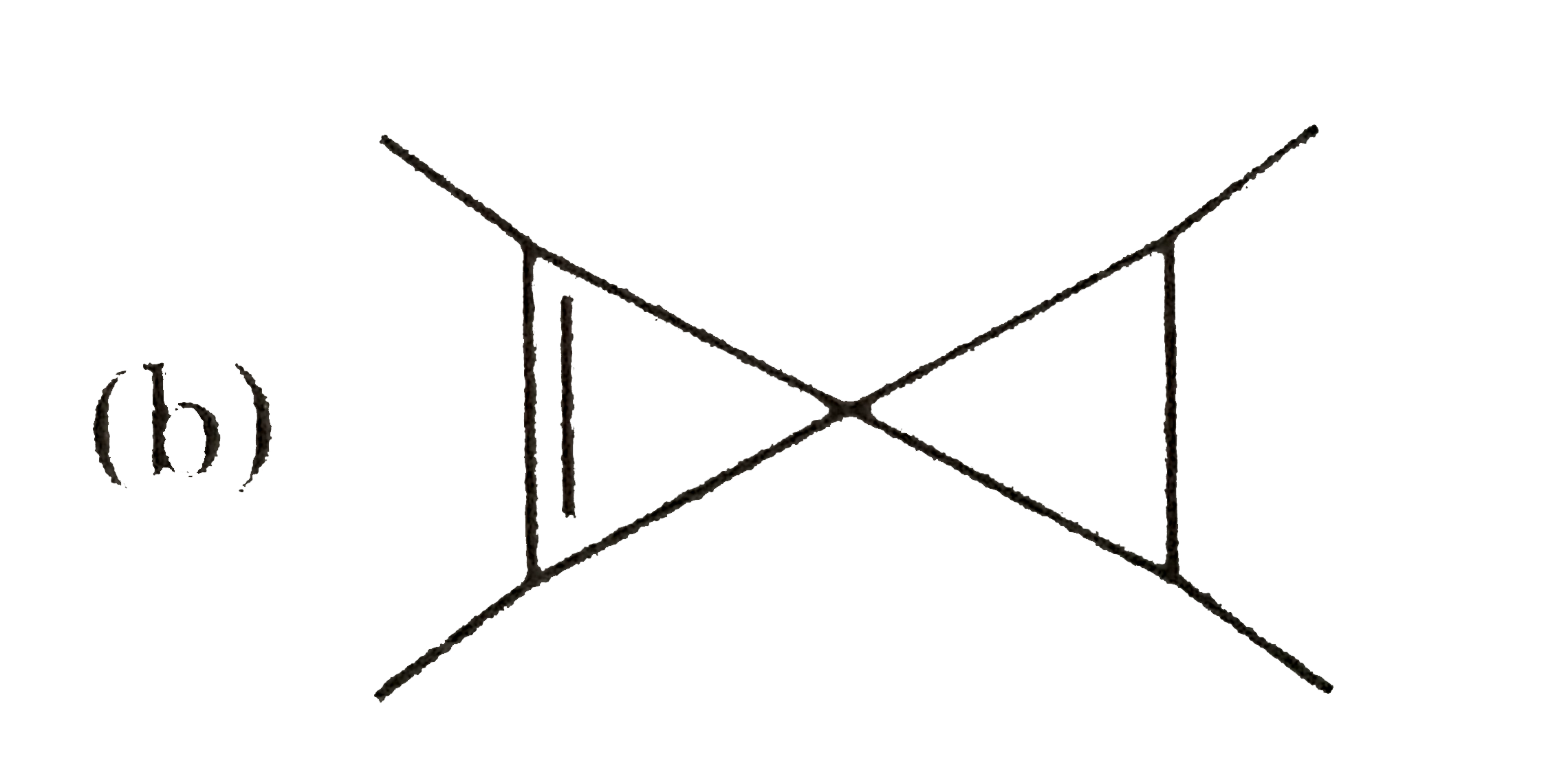
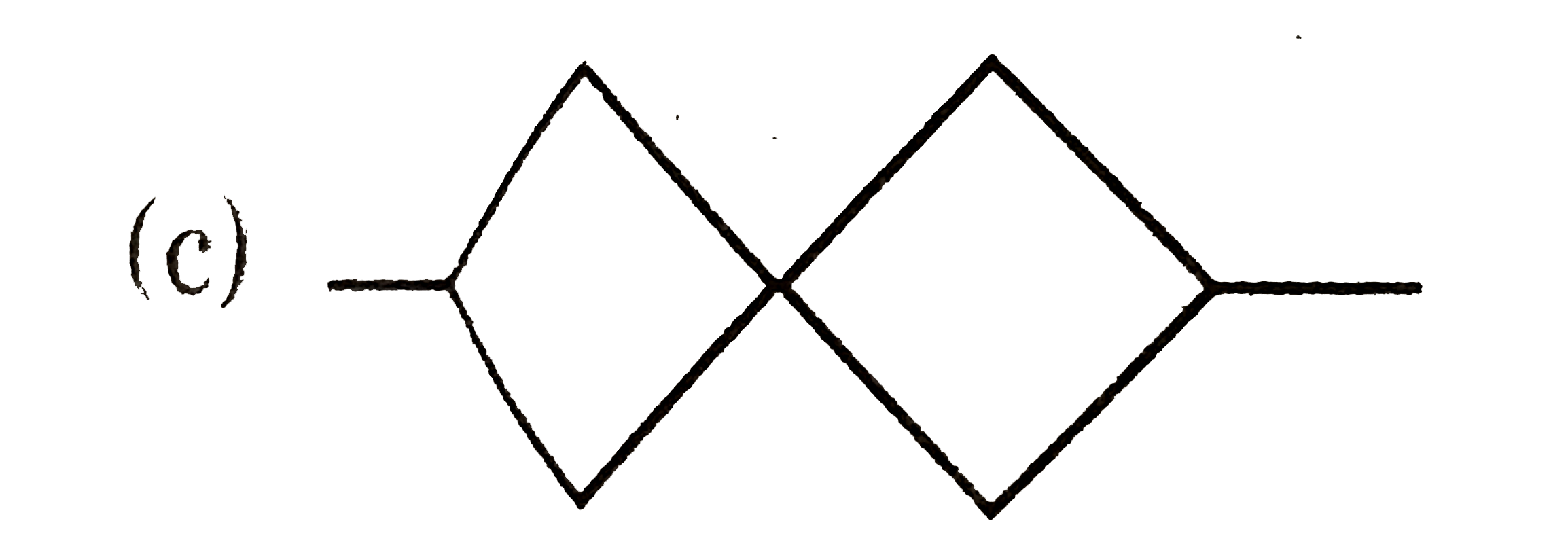
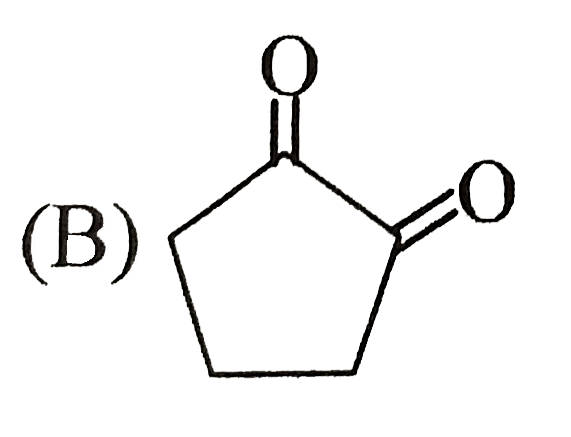
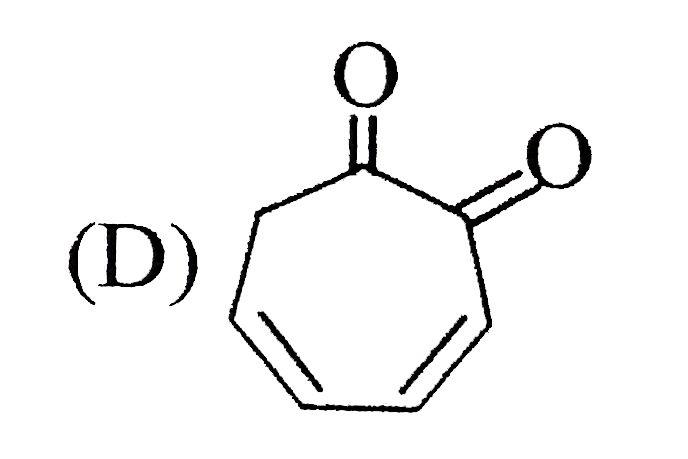
Which of the following structure cannot show optical isomerism? |
|
Answer»
|
|
| 17. |
Write structural formula for the following compounds (i) Cyclohexa-1, 4-diene (ii) Ethynyl cyclohexane |
Answer» SOLUTION :
|
|
| 18. |
which of the following are lyophilic in nature ? |
| Answer» Answer :A,C,D | |
| 19. |
Which of the following has a planar structure? |
|
Answer» `NH_(4)^(+)` |
|
| 20. |
What is resonance energy?Write about its value |
|
Answer» Solution :The energy of actual STRUCTURE of the molecule (the resonance hybrid) is LOWER than that of any of the canonical structures. The difference in energy between the actual structure and the lowest energy resonance structure is CALLED the resonance stabilisation energy or simply the resonance energy VALUE of resonance energy: The more the number of important contributing structures, the more is the resonance energy. Reasonsenergy `PROP` No. of resonance structure. Reasons is particularly important when the contributing structures are equivalent in energy |
|
| 21. |
Under which of the following two conditions applied together, a gas deviates most from the ideal behaviour ? |
|
Answer» LOW pressure However, there is no gas which obeys the ideal gas equation under all conditions of pressure and temperature. Thus, the CONCEPT of ideal gas is only theoretical. These gases are here to OBEY the gas laws when the pressure is low or temperature is high. Such gases are known as .real gases.. Mostly all the gaes are real gases. Thus, at high pressure and low temperature, real gas is mostly DEVIATED from ideal behaviour. |
|
| 22. |
Whichof thefollowingis FALSE? |
|
Answer» sodiumion is themajorcationin THEBLOOD plasmaof vertebrates. |
|
| 23. |
Which one of the following is not correct |
|
Answer» Number of unshared electrons on the atom is also considered for calculation of FORMAL charge. |
|
| 24. |
What is Boyle temperature of Boyle point ? |
| Answer» Solution : ..The temperature at which a real GAS obeys ideal gas LAW over an APPRECIABLE range of pressure is called Boyle.s temperature of Boyle POINT... | |
| 25. |
Why are solids rigid ? |
| Answer» Solution :In a SOILD, the constituent particles are very closely PACKED. HENCE, the FORCES of attraction AMONG these particles are very strong. Moreover , the constituent particles in solids have fixed positions and can only oscillate about their mean positions. Hence , they are rigid. | |
| 26. |
The volume of two moles of oxygen at STP |
|
Answer» 22.4 L |
|
| 27. |
X+H_(2)O overset("Heat, above" 30^(@)C)rarr Phenol as one of the product. X can be |
|
Answer» chlorobenzene 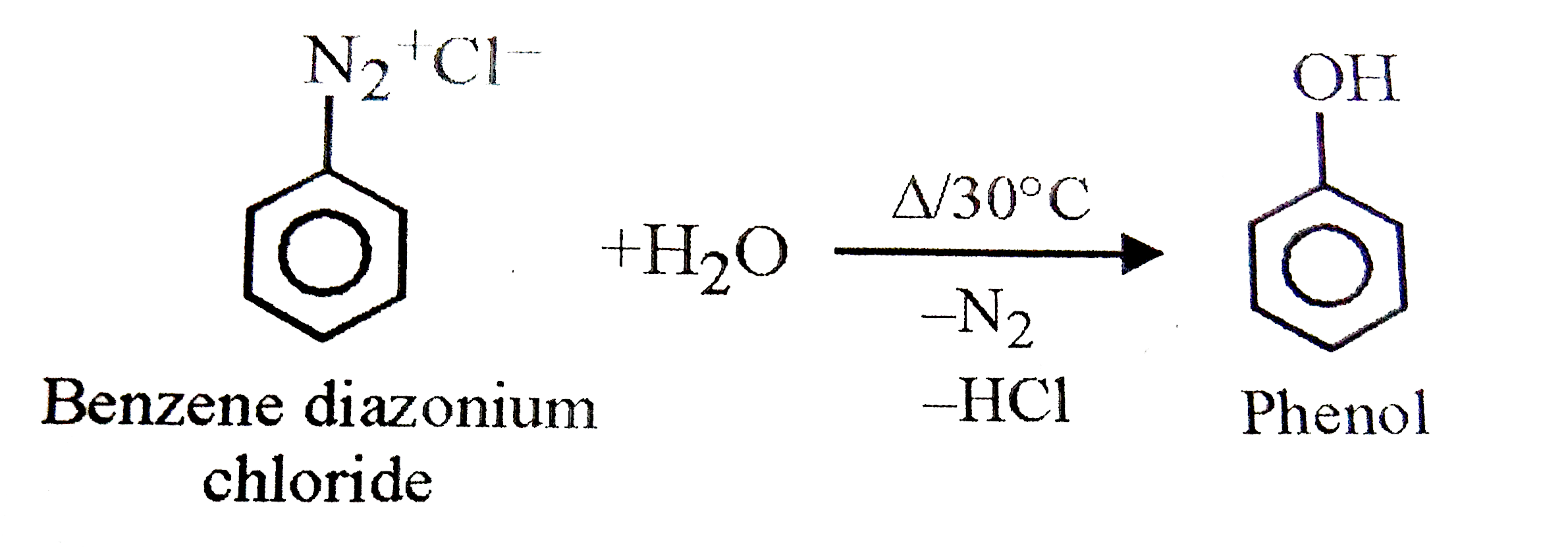
|
|
| 28. |
To prepare butan-2-ol using CH_(3)MgI what other chemical would you choose ? |
|
Answer» Propanal 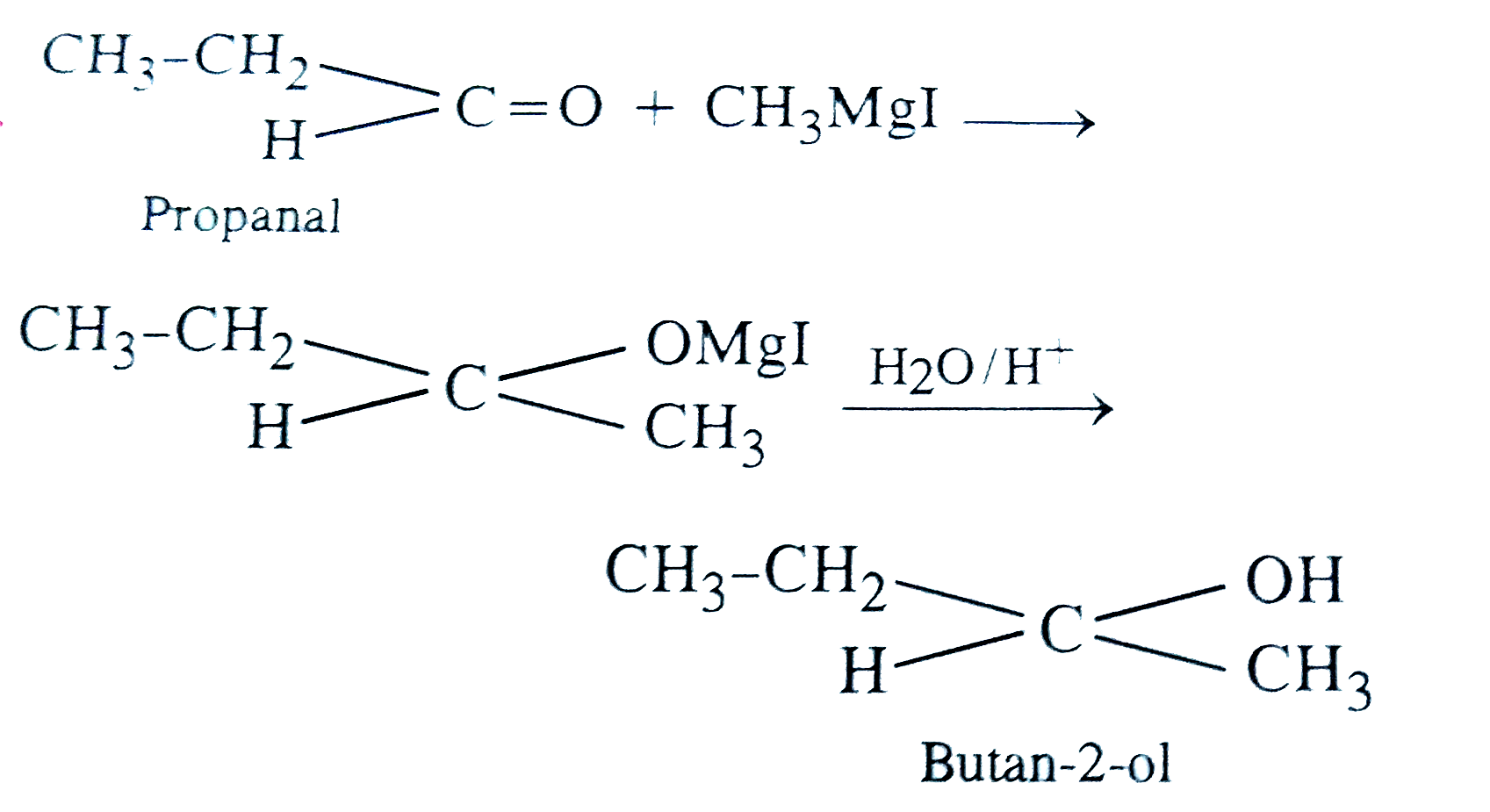
|
|
| 29. |
What type of multiple bonds are involved in the compounds containing C -=C, C-= O and -C-= N groups ? |
|
Answer» <P> SOLUTION :`P PI - P pi` |
|
| 30. |
Which of the following electrolyte will have maximum flocculation value for Fe(OH)_(3) sol ? |
|
Answer» NaCl |
|
| 31. |
Which of the following properties of atom could be explained correctly by Thomosn Model of atom ? |
|
Answer» Overall neutrality of atom |
|
| 33. |
What is perhydrol ? |
| Answer» Solution :PERHYDROL is the trade name for `H_(2)O_(2)` which is USED as an ANTISEPTIC for WASHING wounds , TEETH and ears. | |
| 34. |
What does theelectronconfiguration 1s^(2) 2s^(2) 2p^(5) 3s^(1) indicate ? |
|
Answer» groundstateof FLUORINE |
|
| 35. |
What are the advantages of standard solution. |
|
Answer» Solution :1. The error due to weighing the SOUTE can be minimised by using concentrated stock solution that REQUIRED large quantities of solute. 2. We can prepare working standards of different CONCENTRATIONS by diliting the stock solution which is more efficient since consistency is maintained. 3. Some of the concentrated solutions are more stable and are LESS LIKELY to support microbial growth than working standards used in the experiments. |
|
| 36. |
Whichof the followingisFALSE ? |
|
Answer» LiCiis SOLUBLEIN ETHANOL |
|
| 37. |
Which one of the following is not correct ? |
|
Answer» Number of unshared electrons on the aom is also considered for calculation of FORMAL charge. |
|
| 38. |
Total number of elementsof group 13and 14 which form basicoxides is |
|
Answer» How many SILICON atoms are PRESENTIN the anionof a pyrosilicates ? |
|
| 39. |
What is the basicity of pyrophosphorous acid ? |
|
Answer» 2 |
|
| 40. |
What is Lattice enthalpy ? Give example. |
|
Answer» Solution :The lattice enthalpy of an ionic SOLID is defined as the energy required to COMPLETELY separate ONE mole of a solid ionic compound into GASEOUS constituent ions. Eaxmple : The lattice enthalpy of NaCl 788 kJ`mol^(-1) `. This means that 788, kJ of energy is required to separate one mole of solid NaCl into one mole of `Na_((G))^(+)` and one mole of `Cl_((g))^(-)` to an infinitie distance. |
|
| 41. |
Write the equilibrium constant (K_c) expression for the following reactions. (i) Cu^(2+)(aq)+2Ag(s) hArr Cu(s)+2Ag^+(aq) (ii) 4Hcl(g)+O_(2)(g) hArr 2Cl_(2)(g)+2H_(2)O(g) |
|
Answer» Solution :`log""(K_2)/(K_1)=(DeltaH^@)/(2.303R)[(T_2-T_1)/(T_2T_1)]` (II) `K_(C) = ([Cl_(G)]^2[H_2O(g)]^2)/([HCl(g)]^4[O_2(g)])` |
|
| 43. |
Why B_(2) is paramagnetic in nature while C_(2) is not? |
|
Answer» Solution :The molecular ORBITAL ELECTRONIC configuration of both `B_(2) and C_(2)` are: `B_(2):[sigma1s]^(2)[sigma^(**)1s]^(2)[sigma^(**)2s]^(2)[sigma^(**)2s]^(2)[pi2p_(x)]^(1)[pi2p_(y)]^(1)` `B_(2):[sigma1s]^(2)[sigma^(**)1s]^(2)[sigma^(**)2s]^(2)[sigma^(**)2s]^(2)[pi2p_(x)]^(2)[pi2p_(y)]^(2)` SINCE `B_(2)` has two unpaired electrons, therefore, `B_(2)` is paramagnetic. `C_(2)` has no unpaired electron, therefore, `C_(2)` is diamagnetic. |
|
| 44. |
The size of the following species increases in the order |
|
Answer» `Mg^(2+) LT NA^(+) lt F^(-) lt AL^(3+)` |
|
| 45. |
Which of the following compounds have higher enolic content than Keto content: |
|
Answer» `CH_(3)-overset(O)overset(||)(C)-overset(O)overset(||)(C)-CH_(3)` |
|
| 46. |
What happened to work when (i) gasexpands against external pressure (ii) gas is compressed (iii) gas expands into vacuum (iv) an ideal gas expands reversibly and isothermally . |
|
Answer» Solution :(i) When a gas expands against external pressure, work is done by the system. (ii) When a gas is COMPRESSED , Work is done on the system. (iii) When the gas expands into VACUUM no work is done becomes is zero. (iv) When the gas isallowed to expand under REVERSIBLE condition, work done by the gas is MAXIMUM. |
|
| 47. |
Which of the following correctly represents Boyle's Law ? |
|
Answer»
|
|
| 48. |
The tendency of forming M_("aq")^(3+)is highest for which IIIA group elements ? |
|
Answer» B |
|
| 49. |
Which compound have maximum value of bond energy ? |
|
Answer» HBr |
|
| 50. |
The structural formula of 3-ethyl-2-methyl hexane is |
|
Answer» `CH_(3)-CH(CH_(3))-CH(C_(2)H_(5))-CH_(2)-CH_(2)-CH_(3)` |
|