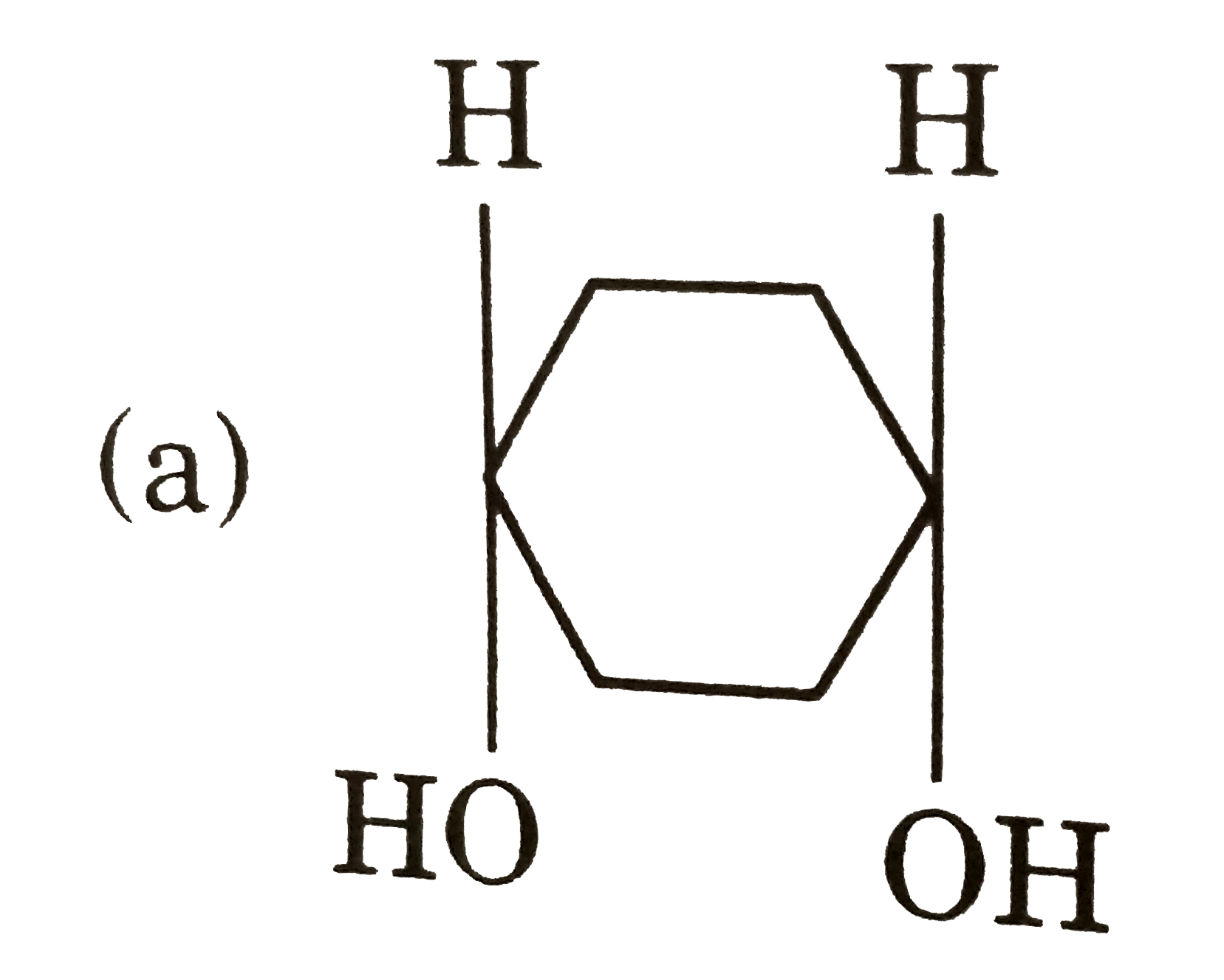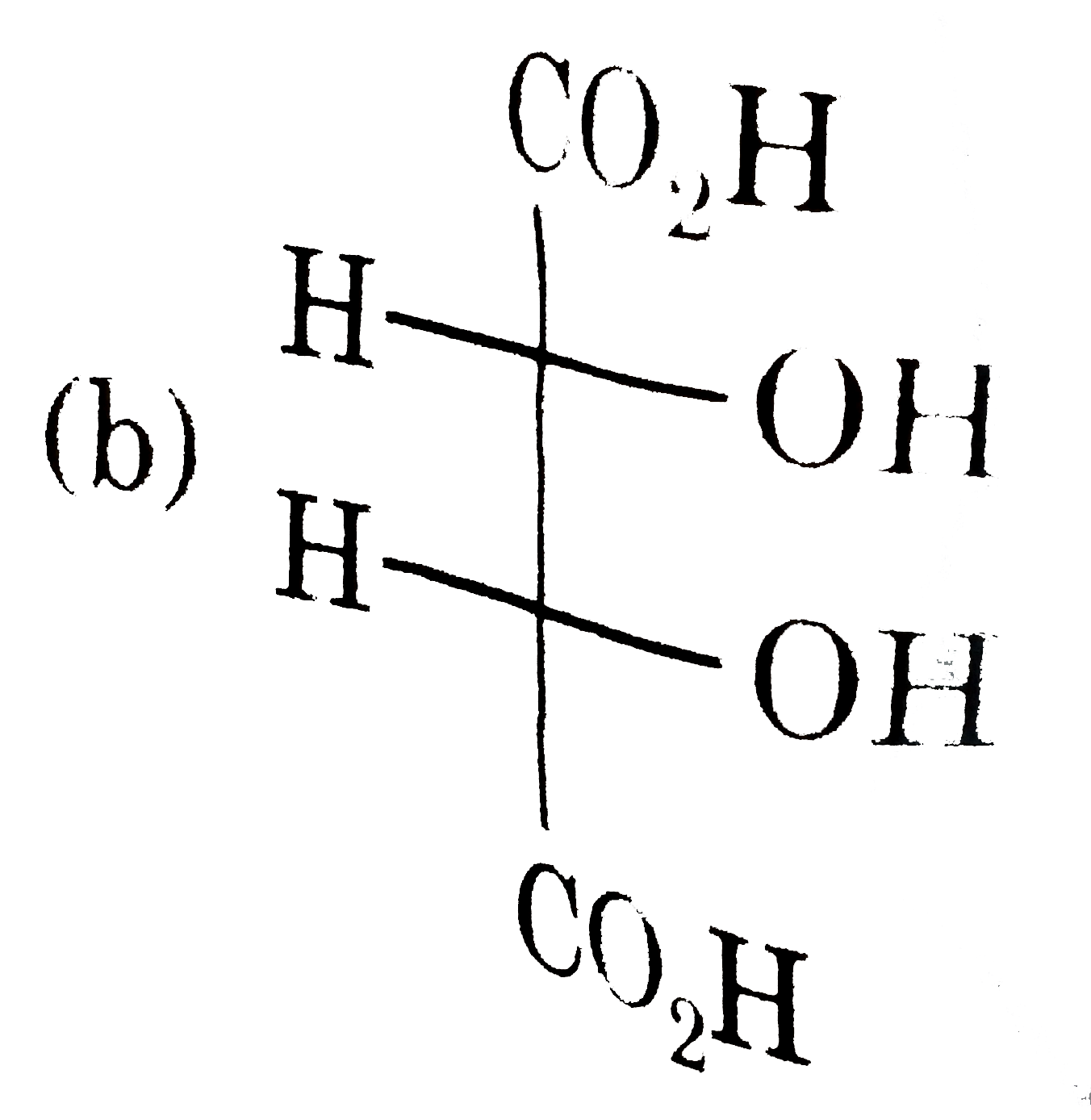Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the followjng statements are incorrect ? |
|
Answer» 0.2 moles of `KMnO_(4)` will oxide one mole of ferrous ions ferric ions in acidic MEDIUM. |
|
| 2. |
Describe the setting of plaster of Paris. |
|
Answer» monohydrate |
|
| 3. |
Which of the following statements about H_3BO_3 is not correct ? |
|
Answer» It is a strong tribasic acid. |
|
| 4. |
Which is (are) disproportionation reaction(s)? |
|
Answer» `2RCHOoverset("Al(OEt)"_3)rarrRCOOCH_2R` `2RCHO overset(Al("OEt")_3)rarr RCOOCH_2R` `4H_3PO_3 overset(Delta)rarr3H_(3)PO_(4)+PH_3` `RCHO overset(KOH)rarrRCOOK + RCH_2OH` In disproportionate reaction the same substance is simultaneously oxidised and reduced . While one atom of the substance acts as oxidant and ANOTHER atom of the substance acts as reductant. |
|
| 5. |
Which of the relation of K_p and K_c at equilibrium in N_2O_(4(g)) hArr 2NO_(2(g)) ? |
|
Answer» `K_p=K_c` |
|
| 6. |
Which ordering of compounds is accrding ot decreasing order of the oxidation state of nitrogen? |
|
Answer» `HNO_(3),NO,NH_(4)CL,N_(2)` |
|
| 7. |
Which of the following statements are true ? |
|
Answer» Mercury poisoning causes a disease called minamata. |
|
| 8. |
What islight ? |
|
Answer» SOLUTION :Inearlierdayslightwassupposedtobe madeof particles in 19the centry LIGTH isthe FORMOF radiationMaxwell wasagainthe firsttoreveal that ligh wavesareassociatedwithoscillating electricand magneticcharacter. |
|
| 9. |
Which one of the following compounds shows the presence of intramolecular hydrogen bond ? |
|
Answer» `H_(2) O_(2)` intermolecular hydrogen bonding whereas cellulose has intramolecular hydrogen BONDS |
|
| 10. |
The surface tension of which of the following liquid is maximum |
|
Answer» `H_(2)O` |
|
| 11. |
What will happen to volume of a bubble of air found under water in a lake where temperature is 15^(@)C and the pressure is 1.5 atm, if the bubble rises to the surface where the temperature is 25^(@)C and the pressure is 1.0 atm? |
|
Answer» Its VOLUME will become greater by a factor of 2.5 `(1.5"atm"xxV)/(288" K")=(1"atm"xxV_(2))/(298" K"):. V_(2)=1.6" V"` |
|
| 12. |
Which of the following pairs is correctly matched ? |
|
Answer» `{:("EXPERIMENTAL","Phenomenon"),("X-ray spectra","CHARGE on the nucleus"):}` |
|
| 13. |
The wavelength of line of sodium is 589.6 nm. Express it in Angstromunit. |
|
Answer» |
|
| 14. |
What type of bonds are formed due to orbital overlap? |
| Answer» Solution :`sigma`bond is due to HEAD on OVERLAP. `pi` bond is due to LATERAL, or sidewise overlap. Due to FACT that the extent of overlap in bond is GREATER than that in | |
| 15. |
The value of K_(c) = 4* 24 at 800 K for the reaction, CO (g) + H_(2)O (g) hArr CO_(2) (g) + H_(2) (g) Calculate equilibrium concentrations of CO_(2) , H_(2) , CO and H_(2) O " at 800 K, if only CO and " H_(2)Oare present initially at concentration of0*10 M each ? |
|
Answer» SOLUTION :` {:(,CO(G),+,H_(2)O(g),hArr,CO_(2)(g),+,H_(2)(g)),("Intial conc.",0*1 M,,0*1M,,,,),(" AT . eqm.",0*1-x,,0*1-x,,x,,x):}` `K_(c) = ([CO_(2)][H_(2)])/([CO][H_(2)O]) :. 4* 24 = x^(2)/(0*1-x)^(2)` or `x/(0*1 - x)= 2*06 or x = 2*06 (0*1-x) or x= 0*206 - 2*06 x or 3*06 x = 0*206 or x = 0*067` `:. [CO_(2)]_(eq) = [H_(2)O]_(eq) = 0*067 M, [CO]_(eq) = [H_(2O] _(eq) = 0*1 - 0*067 = 0*033 M` |
|
| 16. |
What do you mean by "inversion temperature" in different regions of the atmosphere ? |
| Answer» SOLUTION :When we GO from one region of the atmosphere to the next adjoining region, the trend of temperature changes from increase to DECREASE or vice versa. This is CALLED inversion temperature. | |
| 17. |
To measure the quantity of MnCl_(2) dissolved in an aqueous solution, it was completely converted to KMnO_(4) using the reaction. |
|
Answer» `MnCl_(2) +K_(2)S_(2)O_(8)+H_(2)O to KMnO_(4)+H_(2)SO_(4)+HCl` Balanced ionic equation will be : `2Mn^(2+)+5S_(2)O_(8)^(2-)+8H_(2)O to 2MnO_(4)^(2-) +10SO_(4)^(2-)+16H^(+)` Ionic equation involved in the oxidation of oxalic acid by `KMnO_(4)` in acid medium is : `2MnO_(4)^(-)+6H^(+)+5{:(COO^(-),),(|,),(COO^(-),):}to2Mn^(2+)+8H_(2)O+10CO_(2)` Number of moles of `MnCl_(2)=("Number of moles of oxalic acid")/(2.5)` `=(0.225//90)/(2.5)=1xx10^(-3)` Mass of `MnCl_(2)=0.001xx126g` =0.126 g =126 mg |
|
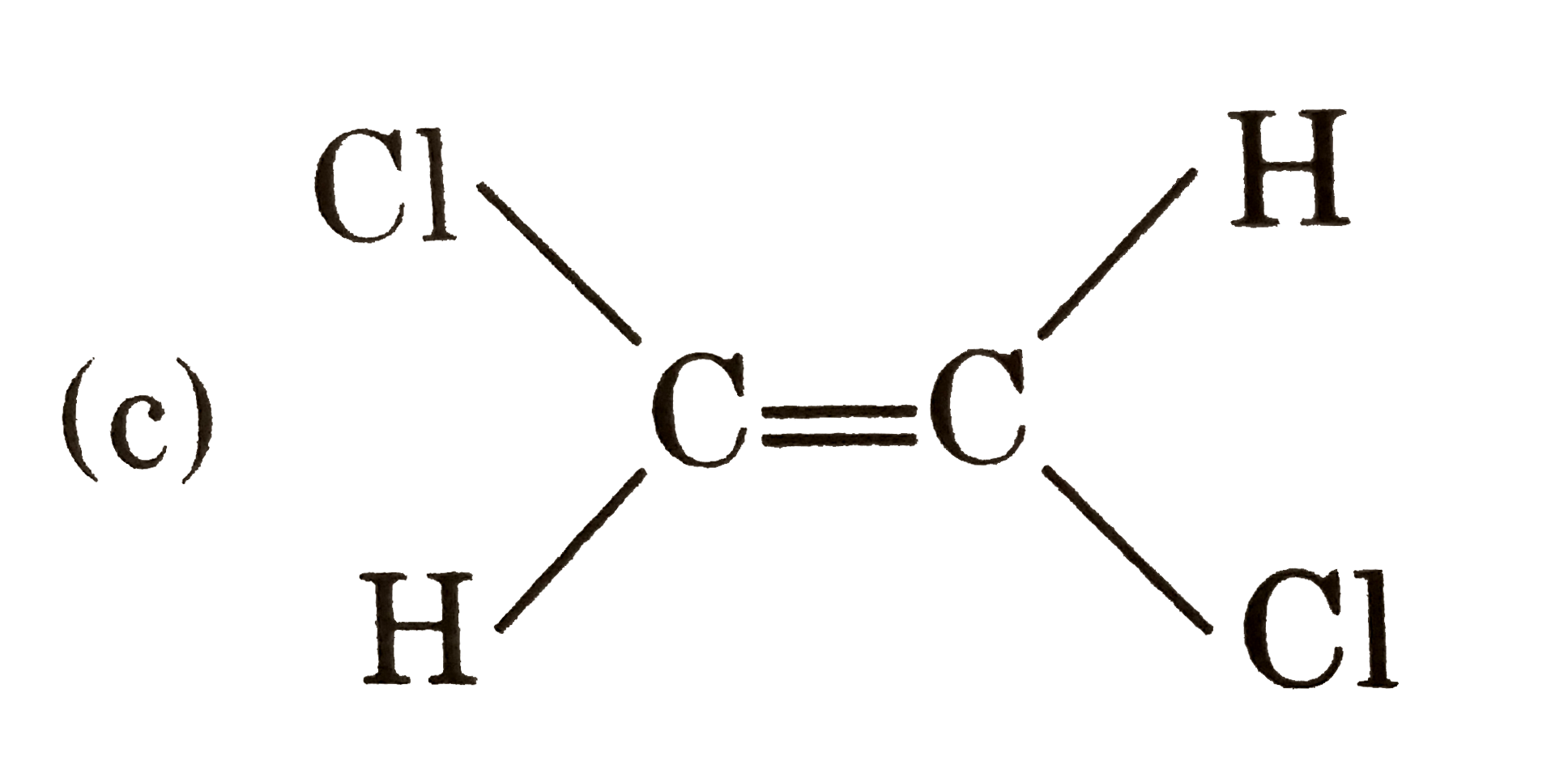
| 18. |
Which of the following pair is disteremoere? |
|
Answer»
|
|
| 19. |
Which of the following statement(s) is (are) correct ? |
|
Answer» The co-ordination number of each TYPE of ion in CsCl crystal is 8 A unit cell of an IONIC crystalshares some of its IONS with other unit cells. |
|
| 20. |
whichof thefollowinghashighestmeltingand boilingpoint? |
|
Answer» BA |
|
| 21. |
What is anoxia or asphyxiation ? |
| Answer» SOLUTION :Acute oxygen starvation in the BODY (due to CO poisoning) is called ANOXIA or ASPHYXIATION. | |
| 22. |
Which akyl groups can originate from propane ? |
| Answer» SOLUTION :n-propyl and sec-propyl | |
| 23. |
Which of the following is very much volatile ? |
|
Answer» Diamond |
|
| 24. |
Whichone of the following is a polynuclear aromatic hydrocarbon |
|
Answer» anthraceen |
|
| 25. |
What is the reason, why the degree of hydrolysis is not present in CH_3COONH_4 solution ? |
|
Answer» Solution :It is a salt of WEAK ACID `CH_3COOH` and weak base `NH_4OH` solution is neutral then `h=sqrt((K_w/(K_a+K_b)))` |
|
| 26. |
Which of the following arrangements represent increasing aoxidation number of the central atom? |
|
Answer» `CrO_(2)^(-),CIO_(3)^(-),CrO_(4)^(2-),MnO_(4)^(-)` `O.N of CI "in" CIO_(3)^(-)=+5` `O.N of Cr "in" CrO_(4)^(2)=6` `O.N of MnO_(4)^(2)=+7` |
|
| 27. |
Which of the following statement is not true about NaCl structure ? |
|
Answer» Each unit CELL CONTAINS 4 NACL MOLECULES |
|
| 28. |
Which of the following statements best explain the greater acidity of terminal alkynes (RC-=CH) compared with monosubstituted alkenes (RCH=CH_(2))? |
|
Answer» The sp-hybridized carbon atoms of the alkynes are LESS ELECTRONEGATIVE than the `sp^(2)` CARBONS of the alkene. |
|
| 29. |
Whatwould be theIUPACnamesand symbolsfor elementswithatomicnumbers 122, 127, 135 149and 150 ? |
|
Answer» Solution :FROMTHE Table3.6page`3//12` therootsfor 2, 7 , 5 , 9and oare biseptpentennand nilrespectively Thereforetheirnamesand symbolsare : `{:(Z,," NAME ",," Symbol "),(122,,"Unbibium ",,UBB),(127,,"Unbiseptium ",,Ubs),(135,,"Untripentium ",,UTP),(149,,"Unquadennuium ",,Uqe),(150,,"Unpentnilium ",,Upn):}` |
|
| 31. |
Which carbocation is the most stable ? |
|
Answer»
|
|
| 32. |
What is covalent bond ? Givesuitable examples to represent single , double and triple covalent bonds. |
Answer» Solution :The type of mutual sharing of one or more PAIRS of electrons between TWO combining ATOMS results in the formation of a chemical bond called a covalent bond. If two atoms SHARE just one pair of electron a single covalent bond is formed as in the case of hydrogen molecule . If two or THREE electron pairs are shared between the two combining atoms, then the covalent bond is called a double bond or a triple bond, respectively. 
|
|
| 34. |
Total number of following reagents, which can be used to distinguish with visile change between SO_(2) and CO_(2) gases: FeCI_(3)-solution, Br_(2)-water, CI_(2)-wate, H_(2)O_(2), (KMnO_(4)+H_(2)SO_(4)), (strach-IO_(3)^(-)),(K_(2)Cr_(2)O_(7)+H_(2)SO_(4)),Ca(OH)_(2) |
|
Answer» |
|
| 35. |
Which set of quantum numbers is possible for the last electron of Mg^(+) ion |
|
Answer» n=3,l=2,m=0,s=+1/2 |
|
| 36. |
Which of the following molecule deviates from Octet rule with respect to central atom |
|
Answer» `SF_6` |
|
| 37. |
Which of the following orbital overlapping is not possible according to VBT? |
|
Answer»
|
|
| 38. |
Whichis / arecorrect statement? |
|
Answer» Insimplecubic closepackedarrangementno octahedralvoidis present at edgecentre. |
|
| 40. |
The percentage of CH_(2)O in CuSO_(4)5H_(2)O is |
|
Answer» zero |
|
| 41. |
When Cu rod is kept in AgNO_(3) solution, give redox reaction. |
| Answer» SOLUTION :`Cu_((s))+2Ag_((aq))^(+)toCu_((aq))^(+2)+2Ag_((s))` | |
| 42. |
The source of energy of Red giant Stars, which are cooler than sun is |
|
Answer» Fission |
|
| 43. |
Write which one out of O_(2)^(+)and O_(2)^(2-)is more stable on the basis of bond order calculations . |
| Answer» Solution :Bondorder of `O_(2)^(+) = 2.5 `, BOND order of `O_(2)^(-)m = 1.5 . Hence, O_(2)^(+)` is more STABLE than `O_(2)^(-)` . | |
| 44. |
Write the lewis do structure for H_(2) and oxygen molecule. |
|
Answer» SOLUTION :`H_(2)` molecule. HYDROGEN atom share one electron pair to acquire octet, then the BOND formed is a single COVALENT bond. `H..HorH-H` (b) `O_(2)` molecule Oxygen atom share two electron paire to acquire octet then the bond formed is a double covalent bond. `:overset(..)O::overset(..)O:or:overset(..)O=overset(..)O:` |
|
| 45. |
What is meant by environmental pollution? |
| Answer» Solution :Any undesirable change in our environment that have harmful effects on PLANTS, animals and human BEINGS is called ENVIRONMENTAL POLLUTION | |
| 46. |
What is the correct bond order for given electron configuration ? (sigma_(1s))^(2) (sigma_(1s))^(2) (sigma_(2s))^(2) (sigma_(2s)^(*))^(2) (sigma_(2p_(z)))^(1) |
|
Answer» 0.5 |
|
| 47. |
The values of the van der Waals' constant a for some gases are given below. H_2 = 0.245 " atm " L^2 mol^(-2) O_2 = 1.360 " atm " L^2 mol^(-2) CO_2 = 3.590 " atm " L^2 mol^(-2) NH_3 = 4.170 " atm " L^2 mol^(-2) Arrange these gases in the decreasing order of their liquefaction tendencies. |
| Answer» SOLUTION :`NH_3 GT CO_2 gt O_2 gt H_2` | |
| 48. |
Under hydrolytic condition, the compounds used for preparation of linearpolymerand forchaingterminationrespectively are |
|
Answer» `CH_(3)SiCl_(3)` and `Si(CH_(3))_(4)` |
|
| 49. |
Which among the following is a Pseudoalum |
|
Answer» `(NH_(4))_(2) SO_(4).Fe_(2)(SO_(4))_(3).24H_(2)O` |
|
| 50. |
The test used for identifying carbon-carbon double fond in an alkene is |
| Answer» Answer :A | |