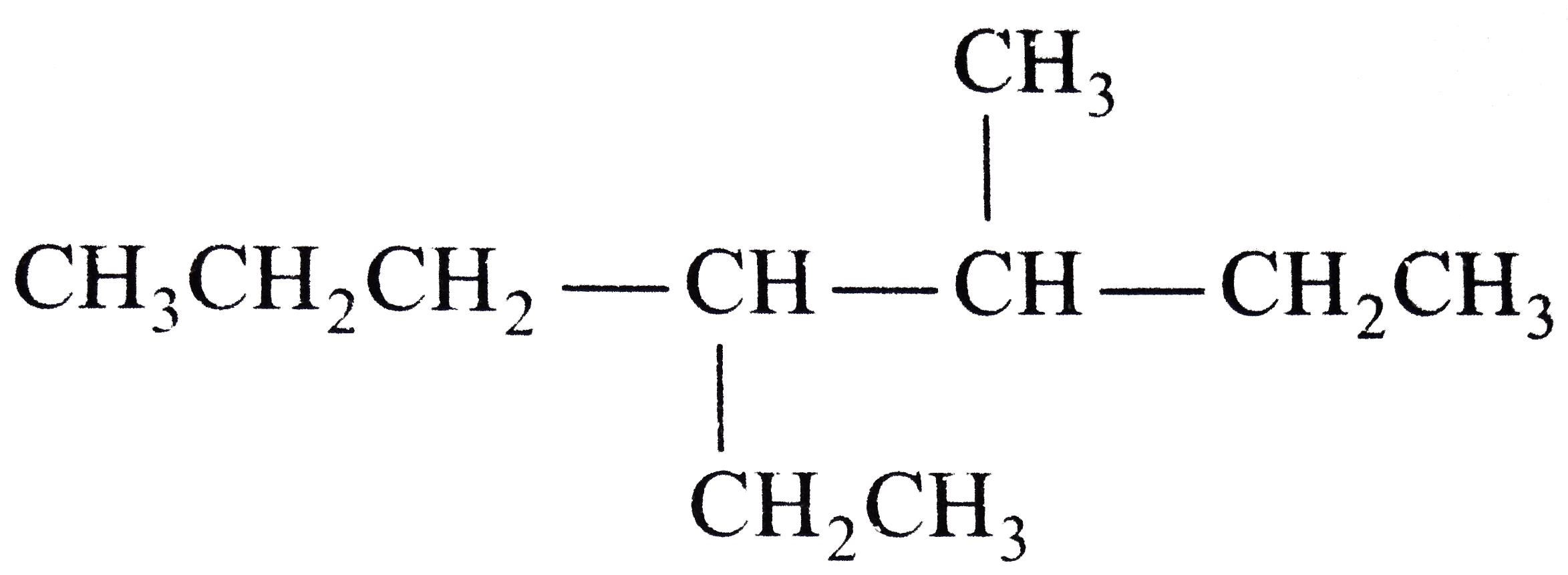Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is/are correct about the redox reaction? MnO_(4)^(ɵ)+S_(2)O_(3)^(2-)+H^(o+)toMn^(+2)+S_(4)O_(6)^(2-) |
|
Answer» 1 " mol of "`S_(2)O_(3)^(2-)` is oxidised by 8 " mol of "`MnO_(4)^(ɵ)` `2S_(2)O_(3)^(2-)toS_(4)O_(6)^(2-)+2e^(-)(n=(2)/(2)=1)` " Eq of "`MnO_(4)^(ɵ)-=" Eq of "S_(2)O_(3)^(2-)` `5xx` MOLES of `MnO_(4)^(ɵ)-=1xx` moles of `S_(2)O_(3)^(2-)` `therefore` 1 " mol of "`S_(2)O_(3)^(2-)=5 " mol of "MnO_(4)^(ɵ)` (b). pH changes from 4 to 10 (acidic to strongly basic) `e^(-)+MnO_(4)^(ɵ)toMnO_(4)^(2-)(n=1)` `S_(2)O_(3)^(2-)to2SO_(4)^(2-)+8e^(-)(n=8)` " Eq of "`MnO_(4)^(ɵ)=" Eq of "S_(2)O_(3)^(2-)` `therefore` 1 " mol of "`S_(2)O_(3)^(2-)=(1)/(8)" mol of "MnO_(4)^(ɵ)` Hence with change of pH from 4 to 10, will change the stoichiometry of reaction and also changes the product. (c). pH changes from 4 to 7 (acidic to neutral medium) `3E^(-)+MnO_(4)^(ɵ)toMnO_(2)(n=3)` `S_(2)O_(3)^(2-)to2HSO_(4)^(ɵ)+8e^(-)(n=8)` Hence it will also effect the stoichiometry of reaction and natural of product. (d). `At pH=7,S_(2)O_(3)^(2-)` is oxidised to `HSO_(4)^(ɵ)` ion. |
|
| 2. |
Which of the following is a square planner ? |
|
Answer» `[NiCl_4[^(2-)` |
|
| 3. |
Which of the following is/are correct matchings ? |
|
Answer» `CH_(3)-OVERSET(C)overset(||)(C)-OH` and `H-overset(O)overset(||)(C)-OCH_(3)` -Metamers |
|
| 4. |
The transition metal used as a catalyst is |
|
Answer» Nickel |
|
| 5. |
Which of the following is the coldest region ? |
|
Answer» Troposphere |
|
| 6. |
Which of the following order is /are incorrect ? |
|
Answer» |
|
| 8. |
Writenoteon decomposition reaction . |
|
Answer» `(##SUR_CHE_XI_VO2_S_MQP_02_E01_053_A01##)` |
|
| 9. |
Whichof these will not be oxidised by ozone |
|
Answer» KI |
|
| 10. |
Whichwerethe majorproblemsbeforethescientistsafter the discoveryof subatomicparticles ? |
|
Answer» Solution :The majorproblems beforethe scientistsat thattimewere to accountfor thestabilityof atom afterthediscoveryof SUBATOMIC particles . |
|
| 11. |
You have the following substances :NH_(3),O_(2),andH_(2)O. Write equations for the preparation N_(2)O from these substances. |
|
Answer» SOLUTION :`4NH_(3)(G)+5O_(2)(g)UNDERSET(1100K)overset(Pt)to4NO(g)+6H_(2)O(l):""2NO(g)+O_(2)(g)to2NO_(2)(g)` `3NO_(2)(g)+H_(2)O(l)to2HNO_(3)(aq)+NO(g),NH_(3)(g)+HNO_(3)(aq)toNH_(4)NO_(3)(aq)` `NH_(4)NO_(3)(aq)underset("at room temperature")overset("Vacuum evaporation")toNH_(4)NO_(3)(s),""NH_(4)NO(s)overset(523K)toN_(2)O(g)+2H_(2)O(l)` |
|
| 12. |
Write the possible metameric structure for C_4 H_(10) . |
|
Answer» SOLUTION :`C_4 H_(10)O `: `underset("1- methyoxypropane ")((i)CH_3 -O-CH_2 -CH_3)` `(ii )CH_3-O-underset( 2 "methoypropane ") underset(CH_3) underset( |)CH - CH_3` ` (iii )underset( "ethoxy ethane ")(CH_3-CH_2 -O- CH_2-CH_3)` |
|
| 13. |
Which of the following does not illustrate the anomalous property of Li ? |
|
Answer» The m.p. and b.p. of LI are compara-tively high |
|
| 14. |
The threshold frequency of a metal is 1.11 xx 10^(16) Hz. What is the maximum kinetic energy of the photo electron produced by applying a light of 15Å on the metal? |
|
Answer» Solution :`v=(C)/(lambda)=(3.0xx10^(8))/(15xx10^(-10)m)=2XX10^(17)Hz` `KE=hv-hv_(0)` `KE=[6.625xx10^(-34)Jsxx2xx10^(17)s^(-1)]-[6.625xx10^(-34)Jsxx1.11xx10^(16)s^(-1)]` `"Kinetic ENERGY "=1.25xx10^(-16)J` |
|
| 15. |
Which of the following is arranged in the increasing order of enthalpyof vaporisation ? |
|
Answer» `NH_(3). PH_(3). AsH_(3)` |
|
| 16. |
When a saturated solution of KCL is heated it becomes |
|
Answer» UNSATURATED |
|
| 17. |
Which reagent will bring about the conversion of carboxylic acids into esters ? |
|
Answer» `C_(2)H_(5)OH` |
|
| 18. |
Which one is not in IUPAC system ? |
|
Answer» `underset(("3-methyl-2-butanol"))(CH_(3)-underset(OH)underset(|)(CH)-underset(CH_(3))underset(|)(CH)-CH_(3))` |
|
| 19. |
What is the average distance between covalent molecules of two atoms called ? |
|
Answer» ATOMIC RADIUS |
|
| 20. |
What is the correct order of decreasing stability of the following cations? underset(I)(CH_(3)-overset(+)(C)H-CH_(3))""underset(II)(CH_(3)-overset(+)(C)H-OCH_(3)) underset(III)(CH_(3)-overset(+)(C)H-CH_(2)-OCH_(3)) |
|
Answer» IIgtIgtIII 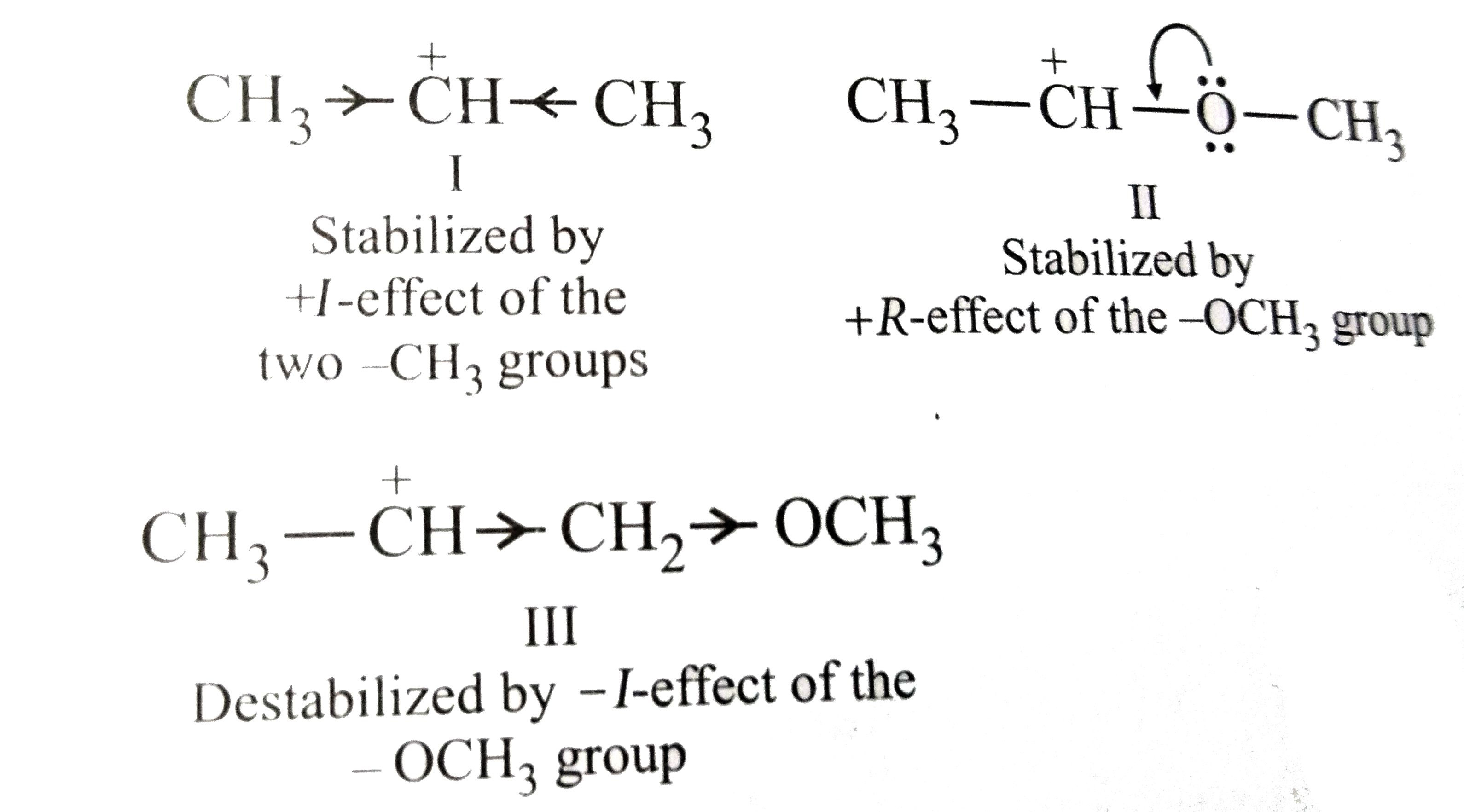 THUS, the stabiloity of CARBOCATIONS DECREASES in the ORDER: IIgtIgtIII. |
|
| 21. |
Which of the following is second most electronegative element ? |
|
Answer» CHLORINE |
|
| 22. |
Which of the following elements form peroxides when heated in excess of air ? |
| Answer» Solution :K FORMS superoxide , `KO_(2)` while others FORM peroxides . | |
| 23. |
Washing soda is |
|
Answer» `Na_(2)CO_(3) . 7H_(2)O` |
|
| 24. |
Which among the following statement(s) describe an element ? (i) It is pure substance which could be split into two or more simpler substance. (ii) It is a pure substance which cannot be split into simpler substance (iii) It's composition is not uniform (iv) All the above |
| Answer» | |
| 25. |
What is meant by .demineralised. water and how can it be obtained ? |
|
Answer» Solution :Permanent hardness is due to the presence of soluble salts of magnesium and calcium in the form of chlorides and sulphates in water. Permanent hardness is not REMOVED by BOILING. It can be removed by the following methods. (i) Treatment with washing soda (sodium carbonate) : Washing soda reacts with soluble calcium and magnesium chlorides and sulphates in hard water to form insoluble CARBONATES. `MCl_2 + Na_2CO_3 to MCO_3 darr + 2NaCl (M=Mg,Ca)` `MSO_4 + Na_2CO_3 to MCO_3 darr + Na_2SO_4` (ii) Calgon.s method: Sodium hexametaphosphate `(Na_6P_6O_18)` , commercially called .calgon., when added to hard water, the following reactions take place. `Na_6P_6O_18 to 2Na^(+)+ Na_4 P_6O_18^(2-)` `M^(2+) + Na_4P_6O_18^(2-) to [Na_2MP_6O_18]^(2-) + 2Na^(+)` (M=Mg, Ca) The complex anion keeps the `Mg^(2+)` and `Ca^(2+)` ions in solution. (iii) Ion-exchange method : This method is also called zeolite/permutit process. Hydrated sodium aluminium silicate is zeolite/permutit. For the sake of simplicity, sodium aluminium silicate `(NaAlSiO_2)` can be written as NaZ. When this is added in hard water, exchange reactions take place. `2NaZ_((s)) + M_((aq))^(2+) to MZ_(2+(aq)) to NZ_(2(s)) + 2Na_((aq))^(+)` (M=Mg, Ca) Permutit/zeolite is said to be exhausted when all the sodium in it is used up. It is regenerated for further use by treating with an aqueous sodium chloride solution. `MZ_(2(s)) + 2NaCl_((aq)) to 2NaZ_((s)) + MCl_(2(aq))` (iv) Synthetic resins method : Nowadays hard water is softened by USING synthetic cation EXCHANGERS. This method is more efficient than zeolite process. Cation exchange resins It contain large organic molecule with -`SO_3H` group and are water insoluble. Ion exchange resin `(RSO_3H)` is changed to RNa by treating it with NaCl. The resin exchanges `Na^+` ions with `Ca^(2+)` and `Mg^(2+)` ions present in hard water to make the water soft. Here R is resin anion. `2RNa_((s))+ M_((aq))^(2+) to R_2M_((s)) + 2Na_((aq))^(+)` The resin can be regenerated by adding aqueous NaCl solution. Pure de-mineralised (de-ionized) water free from all soluble mineral salts is obtained by passing water successively through a cation exchange in the `H^+` form) and an anion exchange in the `OH^-` form) resins. `2RH_((s)) + M_((aq))^(2+) hArr MR_(2(s)) + 2H_((aq))^(+)` In this cation exchange process, H+ exchanges for `Na^(+), Ca^(2+), Mg^(2+)` and other cations present in water. This process results in proton release and thus makes the water acidic. Anion exchange process : `RNH_(2(s)) + H_2O_((l)) hArr RNH_(3)^(+) . OH_((s))^(-)` `RNH_3^(+). OH_((s))^(-) + X_((aq))^(+) hArr RNH_(3)^(+) . X_((s))^(-) + OH_((aq))^(+)` `OH^-` exchanges for anions like `Cl^(-), HCO_3^(-), SO_4^(2-)`etc. present in water. `OH^-` ions, thus, liberated neutralise the `H^+` ions set free in the cation exchange. `H_((aq))^(+) + OH_((aq))^(-) to H_2O_((l))` The exhausted cation and anion exchange resin beds are regenerated by treatment with dilute acid and alkali solutions respectively. |
|
| 27. |
The wavelength of spectral line for electron transition is inversely related to |
|
Answer» NUCLEAR charge |
|
| 28. |
Which of the following statement(s) is are not correct with reference to ferrous and ferric ions |
|
Answer» `Fe^(3+)` GIVES brown colour with POTASSIUM ferricynide |
|
| 29. |
Which of the following is a Bronsted-Lowry acid but not an Arrhenius acid? |
|
Answer» HCL |
|
| 30. |
Total number of molecules having three centered two e^(-) bonds among the following is B_(2)H_(6),Al_(2)Cl_(6),BeCl_(2)(S), BeH_(2)(S), Al_(2)H_(6), [Be_(2)(CH_(3))_(2)]n,C_(2)H_(6),Al_(2)(CH_(3))_(3),C_(2)(CH_(3))_(6) |
|
Answer» |
|
| 31. |
Three isotopes of an element have mass numbers m, (m + 1) and (m + 2). If the mean mass number is (m + 0.5) then which of the following ratios may be accepted for m,(m + 1) (m + 2) in that order? |
|
Answer» `1:1:1` |
|
| 32. |
Which of the following is the most stable form of the mentioned compound? |
|
Answer» Eclipsed |
|
| 33. |
The soliders of Napolean army while at Alps during freezingwinter suffereda serious problem as regards the tin buttongs of theiruniforms.White metallictin buttons got convertedinto grey powder. This transformationis related to |
|
Answer» an INTERACTIONWITH nitrogenof the air at very low temperatures |
|
| 35. |
Which one of the following sets of compounds correctly illustrate the law of of reciprocal proportions. |
|
Answer» `P_(2)O_(3), PH_(3), H_(2)O` |
|
| 36. |
Write the expression for the equilibrium constant, K_c for each of the following reactions : (a)2NOCl(g) hArr 2NO(g)+Cl_2(g) (b)2Cu(NO_3)_2(s) hArr 2CuO(s) +4NO_2(g) (c )CH_3COOC_2H_5 hArr CH_3COOH(aq)+C_2H_5OH(aq) (d)Fe^(3+)(aq) +3OH^(-) (aq) hArr Fe(OH)_3(s) (e) I_2(s) + 5F_2(s) hArr 2IF_5(g) |
|
Answer» Solution :(a)`2NOCl(g) hArr 2NO(g)+Cl_2(g)` `K_c=([NO]^2 [Cl_2])/([NOCl]^2)` (b)`2Cu(NO_3)_2(s) hArr 2CuO(s) +4NO_2(g) +O_2(g)` `K_c=[NO_2]^4[O_2]` (c ) `CH_3COOC_2H_5(aq)+H_2O(L) hArr CH_3COOH(aq)+C_2H_5OH(aq)` `K_c=([CH_3COOH][C_2H_5OH])/([CH_3COOC_2H_5])` (d)`FE^(3+)(aq)+3OH^(-)(aq) hArr Fe(OH)_3(s)` `K_c=1/([Fe^(3+)][OH^-]^3)` (e) `I_2(s) + 5F_2(g) hArr 2IF_5(g)` `K_c=([IF_5]^2)/([F_2]^5)` |
|
| 37. |
Which of the following is the most suitable method for removing the traces of water from ethanol? |
|
Answer» REACTING with Na metal `Mg(OC_(2)H_(5))_(2)+H_(2)O rarr MgO+2C_(2)H_(5)OH`. |
|
| 38. |
This reaction is classified as |
|
Answer» Electrophilic substitution reaction |
|
| 39. |
why are alkali metals strong reducing agents? |
| Answer» SOLUTION :It is because of low IONISATION energy. They lose electrons EASILY, that is why they are STRONG reducing agents. | |
| 40. |
The structure of H_2O_2 is ….. |
|
Answer» planar |
|
| 41. |
What value are permitted for the angular momentum quantum number l for an electron with principal quantum number n = 4 ? |
| Answer» SOLUTION :0, 1, 2, 3 | |
| 42. |
When alpha- rays strike a thin gold foil then, |
|
Answer» most of the `alpha` - rays do not PASS through the GOLD FOIL. |
|
| 43. |
When a light of frequency v_(1) is incident on a metal surface the photoelectrons emitted have twice the kinetic energy as did the photoelectron emitted when the same metal has irradiated with light of frequency v_(2). What will be the value of threshold frequency? |
|
Answer» `v_(0)=v_(1)-v_(2)` |
|
| 44. |
Write the possible isomers for the formula C_(5)H_(10) with their name and type of isomerism present in it. |
|
Answer» Solution :`C_(5)H_(10)`: (i) `CH_(3)-CH_(2)-CH_(2)–CH = CH_(2)` (Pent-1-ene) (II) `CH_(3)-CH_(2)-CH=CH-CH_(3)` (Pent-2-ene) The TYPE of isomerism present above is POSITION isomerism. |
|
| 45. |
Which of the following does possess acidic character?(i) Butane (ii) But-1-ene (iii) But-1-yne (iv) But-2-yne |
| Answer» SOLUTION :But-1-yne | |
| 46. |
Which of the following when dissolved in water forms a solution which is nonconducting ? |
|
Answer» Chile salt PETRE |
|
| 47. |
Which sets of quantumnumber are consitentwith the theory ? |
|
Answer» `n = 2,L = 1,m = 0,s = -1//2` |
|
| 48. |
When citric acid is heated at 525 K, the main product formed is |
|
Answer» Acetone 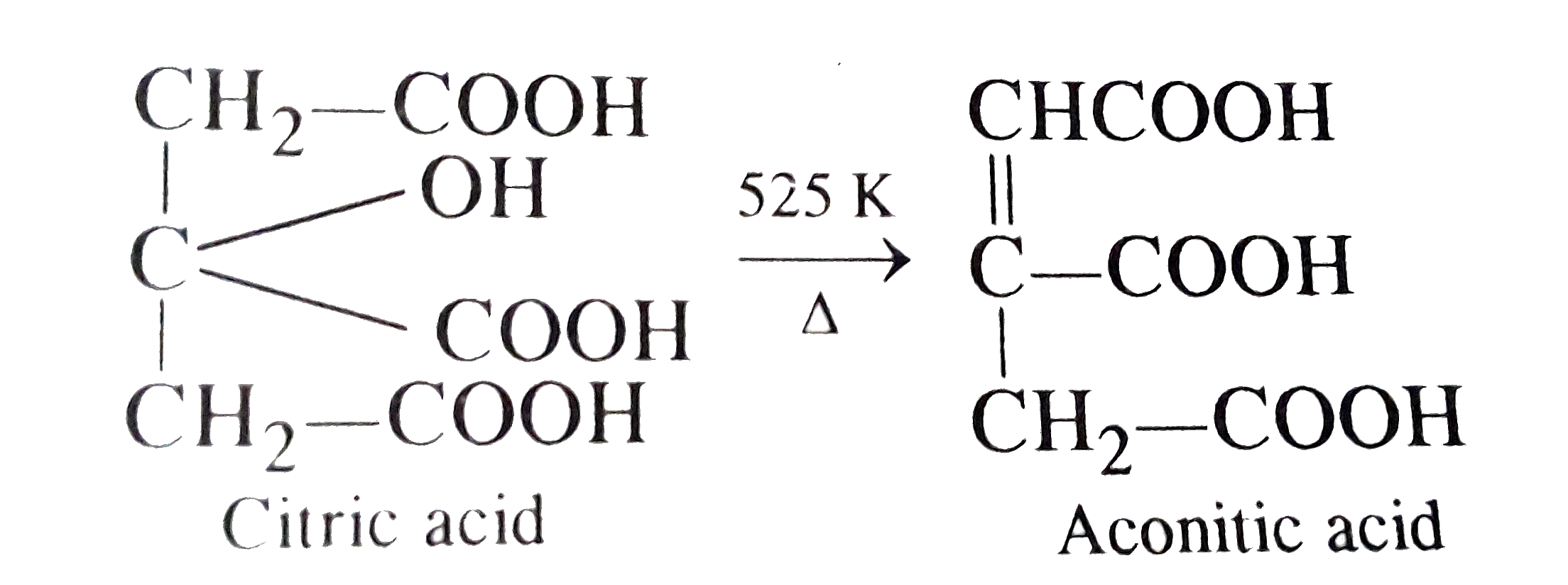
|
|
| 49. |
Which of the following alkali metals forms only, the normal oxide , M_(2)O on heating in air ? (where M is the alkali metal). |
|
Answer» Rb |
|