Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Rain damages the monuments like Taj Mahal in Agra when industries are present nearby Why ? |
| Answer» SOLUTION :Industries produce a lot of OXIDES of nitrogen and sulphur which through reactions produce compounds which dissolve in RAIN water to form `H_2SO_4` and `HNO_3` . The rain THUS becomes acid rain . The marble `(CaCO_3)` of the monuments is attacked bythese ACIDS. | |
| 2. |
Rahul Dravid wants to wear 6.023xx10^(21) Ag atms in the form of a ring. His Silver Gold Copper alloy ring consists of 20% of Silver. The mass of the is 0.9 x. What is x? |
|
Answer» `6.023xx10^(21)` atoms `Ag-108g` `20gAg-100g Ag-Au` ALLOY `1.08g Ag = (1.08xx100)/(20)=5.4g` `0.9x = 5.4 implies x=6` |
|
| 3. |
Radius of the earth is 6.40 xx 10^6 m. Find the diameter of the earth. |
|
Answer» |
|
| 4. |
Radius ratio of an ionic compound is 0.93. The structure of the above ionic compound is of |
|
Answer» NaCltype In css, thecoordination number RATIO 8 : 8 |
|
| 5. |
Radius of 3^(rd) orbit of Li^(2+) ion is x cm then which is not the de Broglie wavelength of electrons in the first orbit is (in cm) |
|
Answer» `6pix ` `therefore 2 pi r = n LAMBDA` `therefore 2 pi xx x/9 = 1 xx lambda = (2pi x)/(9)` |
|
| 6. |
Radius of Cs^(+) ion is less than that of A) Fluoride ion B) Chloride ion C) Bromide ion |
|
Answer» A is CORRECT |
|
| 8. |
Radium disintegrates at an average rate of 2.24 xx 10^13 alpha-particles per minute. Each a-particle takes up two electrons from the air and becomes a neutral helium atom. After 420 days, helium gas collected was 0.5 rnL, measured at 27°C and 750 mmHg. Calculate the Avogadro constant. |
|
Answer» |
|
| 9. |
Radioactive (overset(82)(Br)-overset(82)(Br)) adds to 1-bromocyclohexene. The product is |
|
Answer» is 1,1,2-tribromocyclohexane |
|
| 10. |
Radioactive elements emit alpha, beta and gamma-rays and are characterised by their half-lives. The radioactive isotope of hydrogen is |
|
Answer» Protium |
|
| 11. |
Radioactive isotope of hydrogen is |
|
Answer» uranium |
|
| 12. |
Radioactive elements emit alpha, beta and gamma rays and are characterised by their half-lives. The radioactive isotope of hydrogen is |
|
Answer» <P>protium |
|
| 13. |
Radioactive elements emit alpha, beta" and "gamma rays and are characterised by their half - lives. The radioactive isotope of hydrogen is |
|
Answer» protium |
|
| 14. |
Radioactive elements emit alpha, beta and gamma are characterised by their half-lives. The radioactive isotope of hydrogen is |
|
Answer» Protium |
|
| 15. |
Radioactive alkali metal is ………. And its outer electronic configuration is …………. . |
| Answer» SOLUTION :FRANCIUM , `7S^(1)` | |
| 16. |
Radio (overset(82)(Br)-overset(82)(Br)) adds to 1-bromocyclohexene. The product is |
|
Answer» is 1,1,2-tribromocyclohexane |
|
| 17. |
What is the correct order of radii of Fe^(2+), Fe^(3+) and neutral Fe atom. Why? |
|
Answer» |
|
| 18. |
Radial wave function for an electron in hydrogen atom is Psi = (1)/(16 sqrtpi) ((1)/(a_(0))^(3//2)) [(x -1) (x^(2) - 8x + 12)] e^(-x//2) where x = 2r//a_(0), a_(0) = radius of first Bohr orbit. Calculate the minimum and maximum positions of radial nodes in terms of a_(0) |
|
Answer» Solution :`Psi = (1)/(16 sqrtpi) ((1)/(a_(0))^(3//2)) [(x -1) (x^(2) -8x - 12)] e^(-x//2)` At radial node, `Psi = 0`. Hence, `(x -1) = 0 or (x^(2) -8x - 12) = 0` If `x -1 = 0`, then `x = 1`, i.e., `(2r)/(a_(0)) = 1 or r = (a_(0))/(2)` If `x^(2) -8x + 12 = 0`, i.e.,` (x -6) (x -2) = 0`, then `(x -6) = 0 or (x -2) = 0` If `(x -2) = 0`, then `x = 2 or (2r)/(a_(0)) = 2 or r = a_(0)` If `(x - 6) = 0`, then x = 6 or `(2r)/(a_(0)) = 6 or r = 3 a_(0)` Thus, nodes exist at `(a_(0))/(2), a_(0) and 3a_(0)`. Minimum is at `(a_(0))/(2)` and maximum at `3a_(0)` |
|
| 19. |
Radial probability distribution curve is shown for s-orbital. The curve is: |
|
Answer» 1s |
|
| 20. |
Radial part of the wave function depends on quantum numbers |
|
Answer» n and s |
|
| 21. |
Racemnic mixture is formed by mixing two |
|
Answer» Isomeric compounds |
|
| 22. |
R, u, T can be |
|
Answer» HCOONa,NaOH,HCHO |
|
| 23. |
R-OH+HX rarr RX+H_(2)O In the above reaction, the reactivity of alcohols is |
|
Answer» Tertiary `gt` secondary `gt` primary |
|
| 26. |
R is : |
|
Answer» But-1-ene 
|
|
| 27. |
R - CH_(2) - CH_(2)OH can be converted into RCH_(2)CH_(2)COOH. The correct sequence of reactants is |
|
Answer» `PBr_(3), KCN, H^(+)` |
|
| 28. |
R-CH_(2)-CH_(2)-OH can be converted into RCH_(2)CH_(2)COOH by the following sequence of steps |
|
Answer» `PBr_(3), KCN, H_(3)O^(+)` 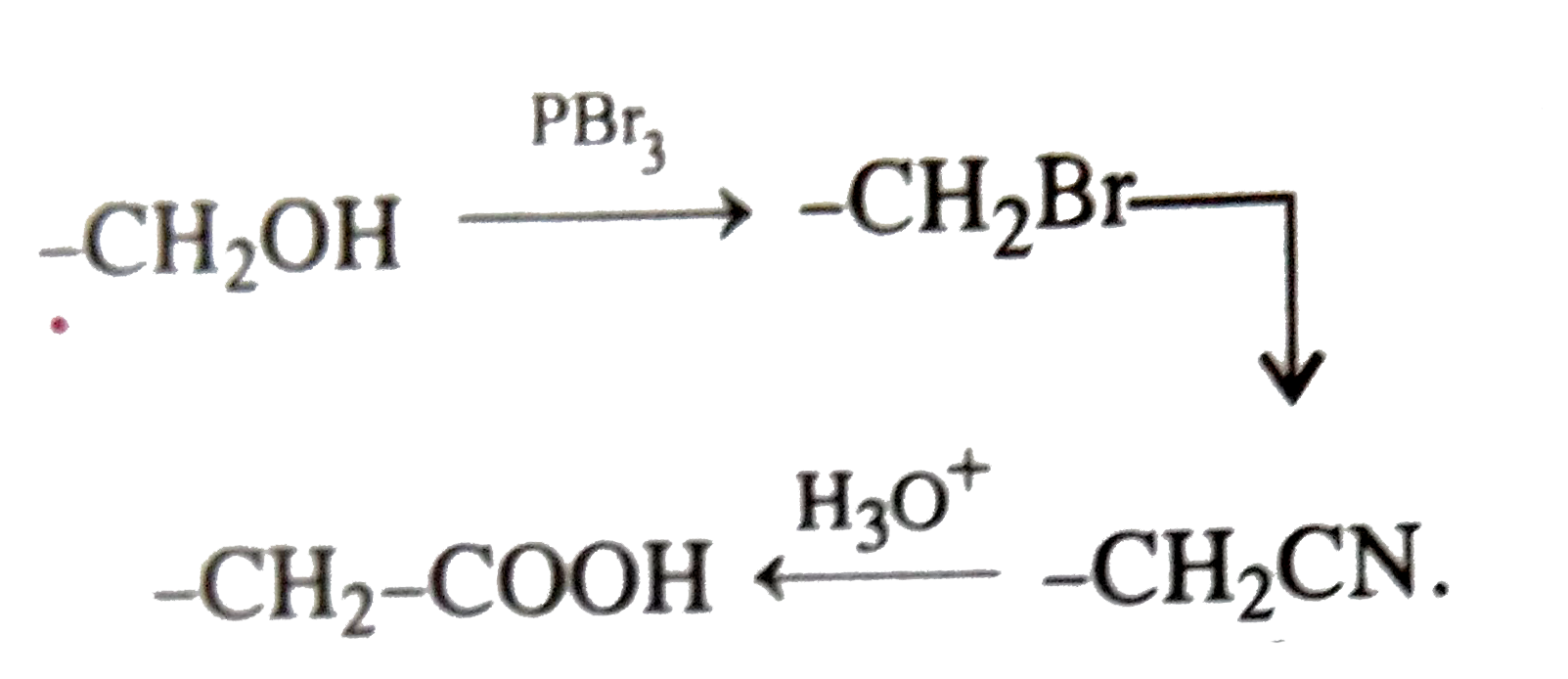
|
|
| 29. |
R-CH = CH_2 overset(NOCl)to X, hence X is |
|
Answer» `R-UNDERSET(NO)underset(|)CH-CL` |
|
| 30. |
R-C-=N underset(-40^(@)C)overset(DIBAL+H,H_(@)O)rarrProduct The product formed is : |
|
Answer» `R-CO-NH_(2)` |
|
| 31. |
R-alanine is represented by |
|
Answer»

|
|
| 33. |
Quicklime and slaked lime are the cheapest and the most widely used bases for neutralising unwanted acids. Lime is used to neutralise acidic soils. A important application of quicklime is in air pollution control for the removal of SO_(2) in electric power plants. Slaked lime is used in the manufacture of other alkalis and bleaching powder, in sugar refining, in tanning hides and in water softening The drying agent which absorbs CO_(2) and reacts violently with water is |
|
Answer» `CaCO_(3)` `Ca(OH)_(2) + Ca(HCO_(3))_(2) rarr 2CaCO_(3) darr + 2H_(2)O` |
|
| 35. |
Quick lime reacts with water to form |
|
Answer» `CaCO_(3)` |
|
| 36. |
Quicklimeisa/ an_______. |
|
Answer» Basicoxide |
|
| 39. |
Quartz Is extensively used as a piezoelectricmaterial, it contains "………..". |
|
Answer» Pb |
|
| 40. |
Quartz is extensively used as a piezoelectric material, it contains ………. |
|
Answer» PB |
|
| 42. |
Quantum numbers are assigned to get complete information of electrons regarding their energy, angular momentum, spectral lines etc. Four quantum numbers are principal quantum numbers which tell the distance of electron from nucleus , energy of electron in a particular shell and its angular momentum. Azimuthal quantum number tells about the subshells in a given shell and of course shape of orbital. Magnetic quantum number is the study of orientations or degeneracy ofa subshell. Spin quantum number which defines the spin of electron designated as +1/2 or -1/2 represented by respectively. Electrons are filled in orbitals following Aufbau rule, Pauli's exclusion principal and Hund's rule of maximum multiplicity. On the basis of this answer the following questions. Two unpaired electrons present in carbon atom are different with respect to their |
|
Answer» AZIMUTHAL QUANTUM number |
|
| 43. |
Quartz is an example of |
|
Answer» CHAIN silicate |
|
| 44. |
Quantum numbers l=2 and m=0 represent which orbital? |
|
Answer» `d_(XY)` |
|
| 45. |
Quartz is |
|
Answer» PIEZOELECTRIC but not FERROELECTRIC |
|
| 47. |
Quantum number value for 2p sub shell are ? |
|
Answer» n=2, l=0 |
|
| 48. |
Quantity of electricity liberated during the formation of 1 mole water in a fuel cell is |
|
Answer» 1F |
|
| 49. |
Quantitative estimation of C, H, and extra elements (e.g., N,S,P and halogens) is carried out by Liebig.s combustion, Carius, Dumas, and Kjeldahl.s method. In the quantitative estimation of oxygen by using I_2O_5,the formula used is: |
|
Answer» PERCENTAGE of `O = 44/32 XX (W xx 100)/(w)` |
|
| 50. |
Quantitive measurements of nitrogen in an organic compounds is done by the method. |
|
Answer» Barthelot method |
|