Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is Hydrides ? Give its types and Explain. |
|
Answer» Solution :Dihydrogen, under certain REACTION conditions, combines with almost all elements, except noble gases, to FORM binary COMPOUNDS, called hydrides. If .E. is the symbol of an element then hydride can be EXPRESSED as `EH_X` (e.g., `MgH_2`) or `E_mH_n` (e.g., `B_2H_6`). The hydrides are classified into three categories: (i) Ionic or saline or saltlike hydrides (ii) COVALENT or molecular hydrides (iii) Metallic or non-stoichiometric hydrides |
|
| 2. |
Which of the following is amphoteric |
|
Answer» `Li_(2)O` `underset(("Basicoxide"))(BeO+2HCl) to BeCl_(2)+H_(2)O`, `underset(("Acidicoxide"))(BeO+2NaOH) to Na_(2)BeO_(2)+H_(2)O` |
|
| 3. |
Which of the following formulae represent an alkane? |
|
Answer» `C_10H_20` |
|
| 4. |
Which of the following statement is/are correct ? |
|
Answer» METALLIC hydries are deficient in hydrogen |
|
| 5. |
Which of the following alkynes can be identified and distinguished from the rest of the alkynes on reaction with ammoniacal silver nitrate to gave a white presipitate? |
|
Answer» `CH_(3)C-=C-CH_(3)` |
|
| 6. |
The values that are useful in writing chemical formulae and in calculation of oxidation states are |
|
Answer» IONISATION potential |
|
| 7. |
Which molecular out of the following does not contain unpaired electrons ? |
|
Answer» `N_(2)^(+)` `O_(2) = KK sigma_(2s)^(2) sigma_(2s)^(**2) sigma_(2p_(z))^(2) pi_(2_(p_(x)))^(2) pi_(2_(p_(y)))^(2)pi_(2_(p_(x)))^(**1)pi_(2_(p_(y)))^(**1) ` `O_(2)^(2-) = KK sigma_(2s)^(2) sigma_(2s)^(**2) sigma_(2p_(z))^(2) pi_(2_(p_(x)))^(2) pi_(2_(p_(y)))^(2)pi_(2_(p_(x)))^(**2)pi_(2_(p_(y)))^(**2) ` `B_(2) = KK sigma_(2s)^(2) sigma_(2s)^(**2)pi_(2_(p_(x)))^(1)pi_(2_(p_(y)))^(1) ` thus , `O_(2)^(2-)`has no unpaired electrons. |
|
| 8. |
What is the value of pH and pK_b of mix solution of ammonium hydroxide and ammonium chloride ? Why ? |
|
Answer» Solution :`PH APPROX pK_b approx` 9.25 because in it taken `[NH_4^+] approx [NH_3]` So, LOG `(["conjugate acid", NH_4^+])/(["base", NH_3])`=log 1 = ZERO So, pH=`pK_b` |
|
| 9. |
When Zoclite which is hydrated Sodium Aluminium Silicate, is treated with hard water, the sodium ions are exchanged with |
|
Answer» `H^(+)` IONS d) `Mg^(2+) + 2H^(+) -" Resin" to Mg("Resin")_2 + 2H^(+)` |
|
| 10. |
What is oxidation number of S in H_(2)SO_(5) ? |
| Answer» SOLUTION :The oxidation number of S in `H_(2)SO_(5) is +6. | |
| 11. |
Which of the following decreases the dissolved oxygen in water? |
| Answer» Solution :Organic matter | |
| 12. |
What is the correct order of acid catalysed hydation of following alkenes ? (I)CH_2=CH_2(II)CH_3-CH=CH_2(III)(CH_3)_2C=CH_2(IV)CH_3-CH=CH-CH_3 |
|
Answer» III GT I gt II gt IV |
|
| 13. |
The reagent that can be used to distinguish between phenol and ethanoic acid is |
|
Answer» AMMONIACAL silver NITRATE SOLUTION |
|
| 14. |
Which is the actual order of ions with respect to ionic radius? |
|
Answer» `N^(3-) gt O^(2-) gt F^(-) gt NA^(+)` |
|
| 15. |
Whether the following redox reaction is oxidation and which is reduction? Zn to Zn^(2+)+2e^(-) |
| Answer» SOLUTION :OXIDATION | |
| 16. |
The structure of 4-methylpent-2ene is |
|
Answer» `(CH_(2))_(2)CH-CH_(2)CH=CH_(2)` |
|
| 17. |
What is the number of photons of light with a wavelength of 4000 pm that provide 1J of energy ? |
|
Answer» Solution :`lamda = 4000" pm " = 4000 xx 10^(-12) m = 4XX 10^(-9) m` `E = Nhv = N h(c)/(lamda) :. N = (E xx lamda)/(h xx c) = ((1J) xx (4xx 10^(-9) m))/((6.626 xx 10^(-34) Js) (3.0 xx 10^(8) MS^(-1))) = 2.012 xx 10^(16)` photons |
|
| 18. |
Which one is an example of gel |
|
Answer» Soap |
|
| 19. |
Which of the following species is non-linear ? |
|
Answer» `ICI_(2)^(-)` angular due to `sp^(3)` hybridization of Cl-atom 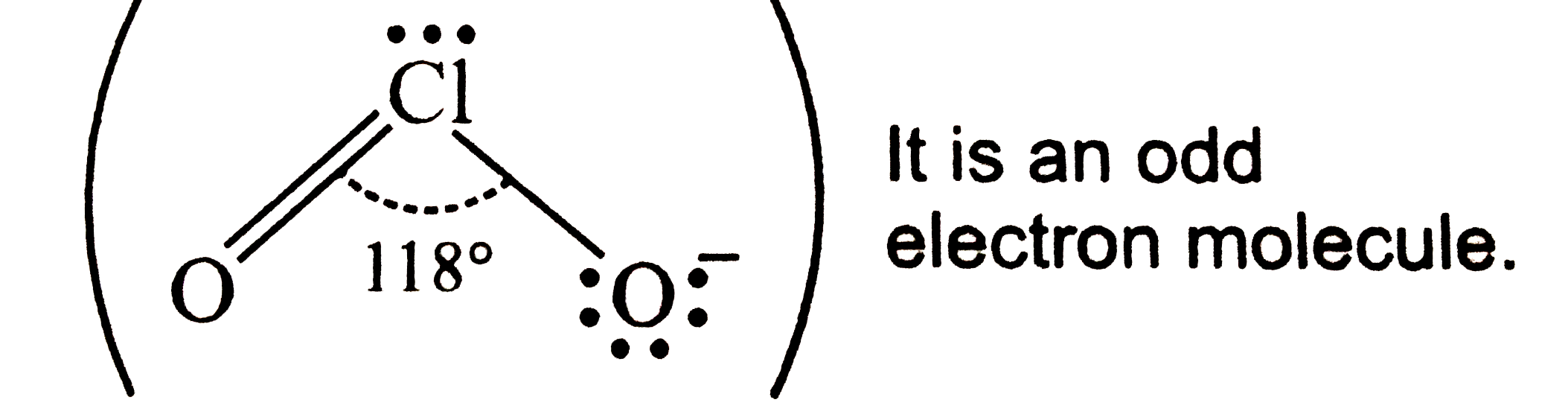
|
|
| 20. |
Which of the following reactions does occur as shown (i.e., forward is favoured) |
|
Answer» `CH_3 -= C^(-) Na^(+) + CH_3CO_2 H to CH_3CO_2^(-) Na^(+) + CH_3C -= CH` `CH_3 COOH GT CH_3 - OH gt CH_3 - C -= CH gt NH_3` |
|
| 21. |
Which of the following carbanion is most stable |
|
Answer» `CH_(3)CH_(2)^(-)` |
|
| 22. |
What is SI units of coefficient of viscosity ? How is it related to poise ? |
|
Answer» SOLUTION :SI unit=kg `m^(-1)s^(-1)` Poise =1 g `CM^(-1)s^(-1)"" :. "" 1 "Poise"=10^(-1) kg ^(-1)s^(-1)=(1)/(10)`th of SI unit. |
|
| 23. |
The value of enthalpy change(DeltaH ) for the reactionC_(2)H_(5)OH(l) +3O_(2)(g)rarr 2CO_(2)(g) +3H_(2)O(l) at 27^(@)C is -1366 kJ mol^(-1). The value of internal energy change for the above reaction at thistemperature will be |
|
Answer» `-1371.5 kJ ` `DeltaU = DeltaH -Deltan_(g)RT` `= - 1366.5 kJ mol^(-1) - ( -1) XX 8.314 xx 10^(-3) kJ K^(-1)mol^(-1) xx 300K` `= - 1366.5 + 0.8314 xx 3 kJ mol^(-1)` `=- 1366.5 +2.4942 = - 1364kJ mol^(-1)` |
|
| 24. |
Write preparation of diborane and give physical and chemical properties of it. |
|
Answer» Solution :Preparation : The simplest boron hydride known, is diborane. It is prepared by treating boron trifluoride with `LiAlH_4` in diethyl ETHER. `4BF_3 + 3LiAlH_4 to 2B_2H_6+ 3LiF + 3AlF_3` A convenient laboratory method for the preparation of diborane involves the oxidation of sodium borohydride with iodine. `I_2 + 2NaBH_4 to B_2H_6 + 2NaI + H_2` Diborane is produced on an industrial scale by the reaction of `BF_3` with sodium hydride. `2BF_3 + 6NaH overset(450 K)to B_2H_6+6NaF` Physical properties : Diborane is a colourless, highly toxic gas with a b.p. of 180 K. Diborane catches fire spontaneously uponexposure to air. It burns in oxygen releasing an enormousamount of energy. Chemical properties : Boranes are readily hydrolyzed by water to give boric acid . `B_2H_(6(g)) + 6H_2O_((l)) to 2B(OH)_(3(aq)) + 6H_(2(g))` Diborane undergoes CLEAVAGE reactions with Lewis bases (L) to give borane adducts, `BH_3 xx L`. `B_2H_6+2NMe_3 to 2BH_3 . NMe_3` `B_2H_6+ 2CO to 2BH_3 . CO` Reaction of ammonia with diborane gives initially `B_2H_6 .2NH_3` which is FORMULATED as `[BH_2(NH_3)_2]^(+) [BH_4]^-`further heating gives borazine, `B_3N_3H_6` known as "inorganic benzene" in view of its ring structure with alternate BH and NH GROUPS. `2B_2H_6+6NH_3 to 2[BH_2(NH_3)_2]^(+)[BH_4]^(-)oversetDeltato 2B_3N_3H_6+12H_2`  The four terminal hydrogen atoms and the two boron atoms lie in one plane. Above and below this plane, there are two bridging hydrogen atoms. The four terminal B-H bonds are regular two centre-two electron bonds while the two bridge (B-H-B) bonds are different and can be DESCRIBED in terms of three centre-two electron bonds shown in fig. Boron also forms a series of hydridoborates, the most important one is the tetrahedral `[BH_4]^-`ion. Tetrahydridoborates of several metals are known. Lithium and sodium tetrahydridoborates, also known as borohydrides, are prepared by the reaction of metal hydrides with `B_2H_6` in diethyl ether. `2MH+B_2H_6 to 2M^(+) [BH_4]^(-)`where , [M=Li,Na] Both `LiBH_4` and `NaBH_4` are used as reducing agents in organic synthesis. They are useful starting materials for preparing other metal borohydrides. |
|
| 25. |
What is the pH value of human blood ? |
|
Answer» 7.4 |
|
| 26. |
Which of the following compounds exhibits geometrical isomerism? |
|
Answer» 2,3-dimethyl 1-2-butene |
|
| 27. |
Which of following is correct for P_(4) molecule of white phosphorus ? |
|
Answer» It has 6 lone PAIRS of electrons |
|
| 28. |
Which of the following statements is not correct regarding purification of liquids by steam distillation ? |
|
Answer» Impurities MUST be non-volatile |
|
| 29. |
What is the state of hybridization of carbon atoms in diamond and graphite? |
| Answer» SOLUTION :`SP^(3) ` and `sp^(2)` | |
| 30. |
Torsion strain is the repulsive interaction between |
|
Answer» ELECTRON cloud of TWO bonds |
|
| 31. |
Which of the following orders of ionic radii is correct ? |
|
Answer» `H^(-)LGT H^(+)gt H` |
|
| 32. |
Which of the following statement about buckyball is incorrect ? |
|
Answer» It is `C_(60)` allotrope of carbon |
|
| 33. |
When the positive and negative charge present on carbon ? Give example |
|
Answer» Solution :If the carbon have three COVALENT bond and ONE vacant orbital then carbon contain (+1) charge e.g. All carbocation `H_(3) overset(+)(C )` means `H - UNDERSET(underset(H)(|))overset(+)(C )-H` If the carbon have three bond and one non bonding ELECTRON pair then carbon contain (-1) charge. e.g All carbanion `H_(3) bar(C )`: means `H- underset(underset(H)(|))overset(..)( C)-H` |
|
| 34. |
The stability of dihalides of Si, Ge, Sn and Pb increases steadily in the sequence, |
|
Answer» `GeX_(2)ltltSiX_(2)ltltSnX_(2)ltltPbX_(2)` |
|
| 35. |
When hydrogen gas at high pressure and room temperature expands adiabatically to a region of low pressure there is a decreases in temperature Hydrogen gas at room temperature is above its inversion temperature . |
| Answer» SOLUTION :HYDROGEN gas at high pressure and room TEMPERATURE expands adiabatically into a REGION of LOW pressure there is a increase in temperature . | |
| 36. |
The standard reduction potential of three metallic cations x,y and z are +0.52,-3.03 and -1.18V respectively .The order of reducing power is: |
|
Answer» `YgtZgtX` |
|
| 37. |
Which is the structural formula of 4-methyl hex-5-yne-2-one? |
|
Answer» `CH_(3)-CH_(2)-underset(underset(O)(||))(C )- underset(underset(CH_(3))(|))(C H)-C -= CH` |
|
| 38. |
Which of the following compounds has zero dipole moment? |
|
Answer» `BeCl_(2)` |
|
| 39. |
Which of the following ions is not isoelectronic with O^(2-) |
| Answer» Solution :`Ti^2`has more `e^-` than `O^(-2)` | |
| 40. |
Which of the following statements are correct. |
|
Answer» Tendency to form complex: `Sc^(+3) gt Y^(+3) gt LA^(+3)` |
|
| 41. |
Two gases have densities 0.388g/cc and 0.097gm/cc. If the second gas has the rate of diffusion 5cc/sec, find the rate of duffusion of the first gas. |
|
Answer» |
|
| 42. |
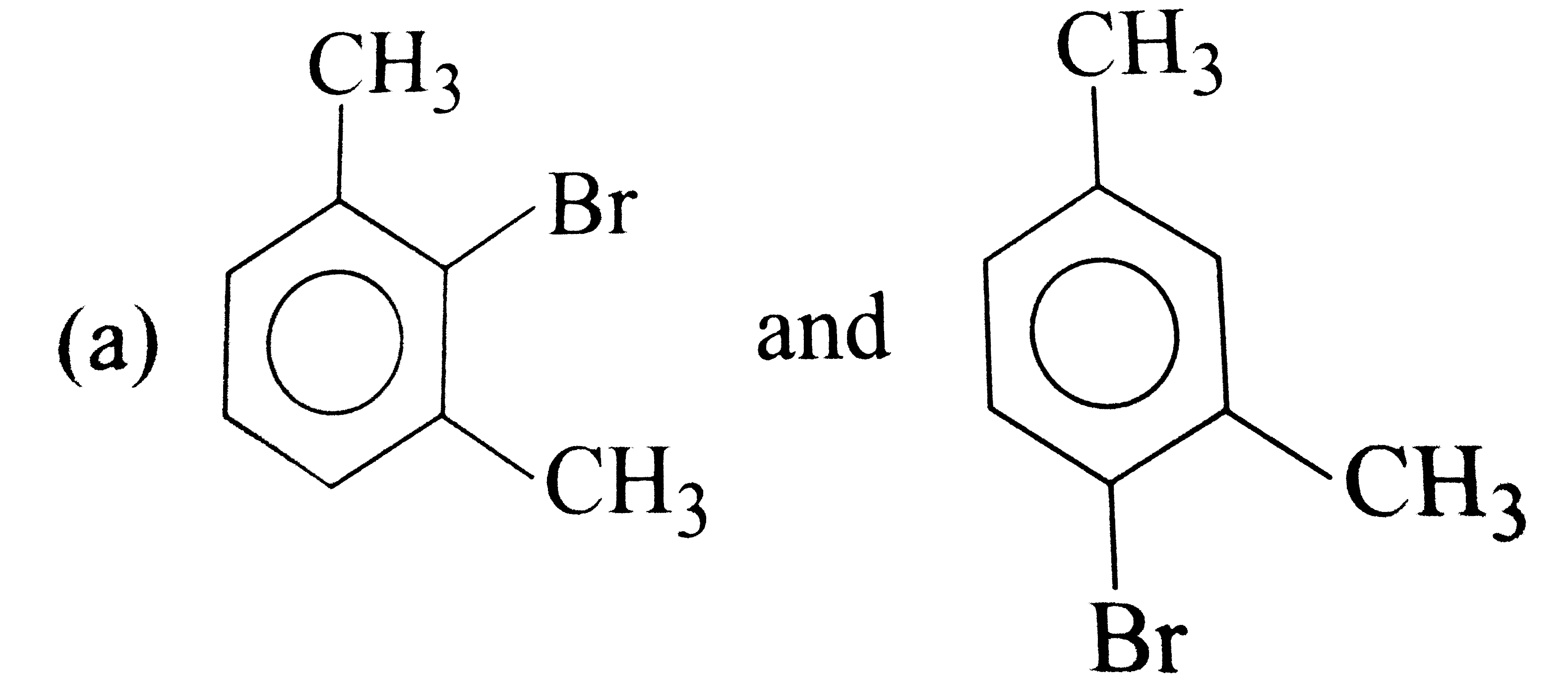
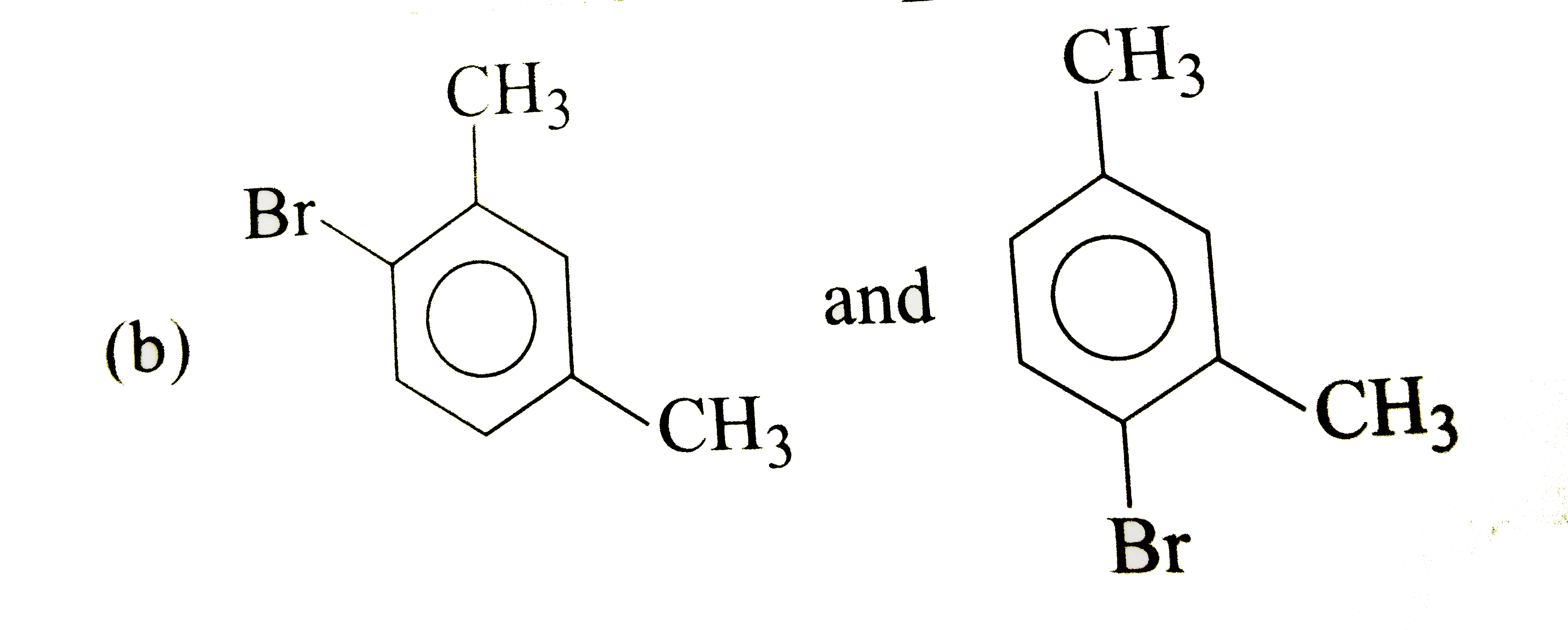
What products are formed when m-xylene is treated with Br_(2) in the presence of FeBr_(3) ? |
|
Answer»
|
|
| 43. |
What is the chemical compositon of zeolite ? |
| Answer» Solution :HYDRATED SODIUM ALUMINIUM silicate , `Na_(2), Al_(2), Si_(2)O_(8).xH_(2)O` | |
| 44. |
The RMS velocity of an ideal gas at 300K is 12240 cm/see, then its most probable velocity in cm/sec at the same temperature is |
|
Answer» 10000 `IMPLIES if C = 12240 cm//s ` then `C_p = 10000 cm//s`. |
|
| 45. |
Which of the following does not represent the correct resonance struture of carbon monoxide |
|
Answer» `: C: : : O:` |
|
| 46. |
Two liquids (A) and (B) can be separated by the method of fractional distillation. The boiling point of liquid (A) is less than boiling point of liquid (B). Which of the liquids do you expect to come out first in the distillate ? Explain. |
| Answer» Solution :Fractional distillation is used to SEPARATE the components of a MIXTURE if their boiling points differ by `20^(@)` or less. In this method, a fractionating column is used in between the flask and the condenser. The purpose of the fractionating column is to PROVIDE hurdles for the ascending vapours and to provide large surface area for condensing the high boiling liquid. Consequently, the vapours of the low boiling liquid (A) will move up while those of the high boiling liquid will condense and fall back into the flask. As a result of this FRACTIONATION, liquid A with lower boiling POINT will distil first and liquid B with higher boiling point afterwards. | |
| 47. |
Which of the following is an extensive property ? (a) Volume, (b) Surface tension, (c ) Viscosity , (d) Density. |
| Answer» SOLUTION :(a) VOLUME is an EXTENSIVE PROPERTY. | |
| 48. |
Which of the following do not sublime on heating ? |
|
Answer» `AlF_(3)` |
|
| 49. |
Which of the following contains maximum number of lone pairs on the central atom? |
| Answer» Solution :`1_(3)^(-)` | |